Advanced Solid Acid Catalysis for High-Purity Phenylene Sulfide Intermediates
Advanced Solid Acid Catalysis for High-Purity Phenylene Sulfide Intermediates
The pharmaceutical and agrochemical industries constantly seek robust synthetic routes for critical intermediates that balance efficiency with environmental compliance. Patent CN102516139A introduces a transformative methodology for the synthesis of phenylene sulfide compounds, utilizing a sophisticated combination of ionic liquid reaction media and transition metal-modified solid superacid catalysts. This technology represents a significant departure from traditional Friedel-Crafts thioalkylation processes, addressing long-standing issues regarding catalyst recovery and waste management. By employing specific ionic liquids such as 1-alkyl-3-methylimidazolium tetrafluoroborate alongside tin-based solid acids, the process achieves high conversion rates and exceptional selectivity. For R&D directors and procurement specialists, this patent outlines a pathway to producing high-purity phenylene sulfide intermediates that aligns with modern green chemistry principles while maintaining economic viability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of phenylene sulfide compounds has relied heavily on Method 3, which involves the Friedel-Crafts reaction of benzene and sulfur using large quantities of anhydrous aluminum trichloride (AlCl3) as a catalyst. While operationally simple, this conventional approach suffers from severe drawbacks that hinder modern industrial application. The primary issue is the generation of substantial amounts of acidic wastewater and solid waste during the quenching and workup phases, creating a heavy burden on environmental treatment facilities. Furthermore, the reaction often produces significant by-products such as thianthrene, which complicates purification and reduces the overall yield of the desired sulfide. The corrosive nature of AlCl3 also necessitates specialized equipment and strict safety protocols, driving up capital expenditure and operational costs for manufacturers seeking a reliable phenylene sulfide supplier.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a heterogeneous catalytic system within an ionic liquid medium to overcome these inefficiencies. The reaction proceeds by mixing benzene compounds and elemental sulfur in the presence of a solid superacid catalyst, such as SO4 2-/SnO2-Fe2O3, dispersed in an ionic liquid like 1-octyl-3-methylimidazolium hexafluorophosphate. This system eliminates the need for stoichiometric Lewis acids, thereby drastically reducing corrosive waste. The ionic liquid acts not only as a solvent but also stabilizes the reactive intermediates, enhancing selectivity towards the desired diaryl sulfide structure. As illustrated in the reaction scheme below, the transformation is direct and efficient, converting substituted benzenes into their corresponding sulfides with minimal side reactions.
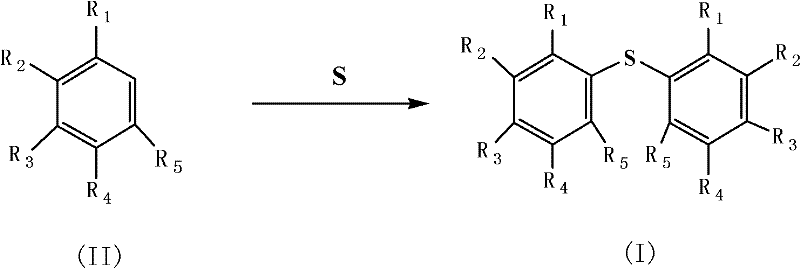
Mechanistic Insights into Solid Superacid-Catalyzed Thioalkylation
The core innovation lies in the synergistic interaction between the transition metal-modified tin oxide solid superacid and the ionic liquid medium. The solid acid catalysts, prepared via co-precipitation and calcination at temperatures between 500°C and 600°C, possess strong surface acidity capable of activating elemental sulfur. Unlike homogeneous catalysts, these solid acids provide active sites on their surface where the sulfur species can interact with the aromatic ring of the benzene substrate. The molar ratio of tin to iron or aluminum in the catalyst lattice, optimized between 2:1 and 20:1, is critical for tuning the acid strength and preventing over-oxidation or polymerization of the substrate. This precise control over the catalytic environment ensures that the electrophilic attack on the aromatic ring occurs selectively at the desired positions, minimizing the formation of polysulfides or other complex impurities.
Furthermore, the ionic liquid medium plays a pivotal role in impurity control and product isolation. Ionic liquids such as 1-alkyl-3-methylimidazolium salts have negligible vapor pressure and high thermal stability, allowing the reaction to proceed at moderate temperatures of 80°C to 110°C without solvent loss. More importantly, the unique solvation properties of the ionic liquid can suppress the formation of thianthrene, a common by-product in traditional methods, by stabilizing the monomeric sulfur species. Post-reaction, the product can be easily extracted using organic solvents like dichloromethane or toluene, leaving the catalyst and ionic liquid behind for potential recycling. This phase separation capability is essential for achieving the stringent purity specifications required for pharmaceutical intermediates, ensuring that heavy metal residues and acidic contaminants are kept to absolute minimums.
How to Synthesize Phenylene Sulfide Efficiently
Implementing this synthesis route requires careful attention to catalyst preparation and reaction parameters to maximize yield and purity. The process begins with the preparation of the solid superacid, followed by the precise dosing of reactants in the ionic liquid medium. The patent specifies a molar ratio of sulfur to benzene compound between 1:2 and 1:3, ensuring an excess of the aromatic substrate to drive the reaction to completion. Detailed standardized synthesis steps, including specific calcination times and extraction protocols, are outlined in the technical documentation to ensure reproducibility across different batch sizes.
- Prepare the solid superacid catalyst (e.g., SO4 2-/SnO2-Fe2O3) via co-precipitation and calcination at 500-600°C.
- Mix sulfur, ionic liquid (e.g., 1-octyl-3-methylimidazolium hexafluorophosphate), benzene derivative, and catalyst in a reactor.
- Heat the mixture to 80-110°C for 5-10 hours, then extract with organic solvent and distill to isolate the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this solid acid catalytic technology offers profound strategic advantages beyond mere technical performance. The shift away from hazardous and corrosive reagents like anhydrous aluminum trichloride fundamentally alters the cost structure of phenylene sulfide manufacturing. By eliminating the need for massive quantities of Lewis acid and the subsequent neutralization steps, the process significantly reduces the consumption of auxiliary chemicals and the volume of hazardous waste requiring disposal. This reduction in waste treatment complexity translates directly into lower operational expenditures and a reduced environmental footprint, making the supply chain more resilient against tightening environmental regulations.
- Cost Reduction in Manufacturing: The elimination of stoichiometric aluminum chloride catalysts removes a major cost driver associated with raw material procurement and waste neutralization. Traditional methods require expensive quenching agents and generate large volumes of saline wastewater, whereas this solid acid method allows for easier catalyst separation and potential reuse of the ionic liquid medium. Consequently, the overall cost of goods sold (COGS) is optimized through reduced material intensity and simplified downstream processing, providing a competitive edge in cost reduction in fine chemical manufacturing.
- Enhanced Supply Chain Reliability: The raw materials required for this process, including elemental sulfur and common benzene derivatives, are commodity chemicals with stable global supply chains. Unlike specialized organometallic catalysts that may face supply bottlenecks, the solid superacids described can be synthesized in-house or sourced reliably. Furthermore, the mild reaction conditions (50°C to 150°C) reduce the energy intensity of the process compared to high-temperature gas-phase methods, ensuring consistent production capabilities even during periods of energy volatility. This stability is crucial for reducing lead time for high-purity phenylene sulfides and maintaining continuous supply to downstream API manufacturers.
- Scalability and Environmental Compliance: The heterogeneous nature of the catalyst system facilitates straightforward scale-up from laboratory to commercial production without the engineering challenges associated with handling corrosive slurries. The process generates minimal acidic effluent, aligning with strict environmental compliance standards required by multinational corporations. This eco-friendly profile not only mitigates regulatory risk but also enhances the brand value of the final product as a sustainably sourced intermediate. The ability to scale complex phenylene sulfide derivatives efficiently ensures that supply can meet growing demand in the agrochemical and pharmaceutical sectors without compromising on quality or delivery schedules.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a clear understanding of the process capabilities and limitations for potential partners.
Q: What are the advantages of using solid superacids over traditional AlCl3 catalysts?
A: Solid superacids eliminate the need for stoichiometric amounts of corrosive anhydrous aluminum trichloride, significantly reducing acidic wastewater generation and simplifying the post-reaction workup process.
Q: Can this synthesis method be scaled for industrial production?
A: Yes, the patent explicitly states the method is beneficial for large-scale industrial production due to mild reaction conditions (80-110°C) and the reusability potential of the ionic liquid medium.
Q: What types of substituents are tolerated in this reaction?
A: The method tolerates various substituents including alkyl, hydroxyl, nitro, halogen, and amino groups, allowing for the synthesis of diverse derivatives like di-4'-hydroxyphenyl sulfide and di-4'-aminophenyl sulfide.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phenylene Sulfide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic pathways in the development of next-generation therapeutics and crop protection agents. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot scale to full manufacturing is seamless and efficient. We are committed to delivering phenylene sulfide intermediates that meet stringent purity specifications, utilizing rigorous QC labs to verify every batch against the highest industry standards. Our infrastructure is designed to handle complex chemistries safely, leveraging advanced catalytic technologies to optimize yield and minimize environmental impact.
We invite you to collaborate with us to leverage this advanced solid acid catalysis technology for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this greener route can improve your margin structure. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate why we are the preferred partner for high-quality chemical intermediates.
