Optimizing Tolvaptan Production: A Technical Analysis of Novel Synthetic Routes for Global Supply Chains
Optimizing Tolvaptan Production: A Technical Analysis of Novel Synthetic Routes for Global Supply Chains
The pharmaceutical landscape for vasopressin antagonists continues to evolve, driven by the demand for more efficient and environmentally sustainable manufacturing processes. Patent CN105753735B introduces a significant advancement in the preparation of Tolvaptan, a selective non-peptide Arginine Vasopressin (AVP) V2 receptor antagonist widely used for treating hyponatremia. This technical insight report analyzes the novel synthetic pathway disclosed in the patent, which offers a robust alternative to legacy methods by utilizing p-chloroaniline as a primary starting material. The innovation lies not merely in the chemical transformation but in the strategic elimination of costly heavy metal catalysts and the optimization of reaction conditions to enhance overall yield and purity. For R&D directors and procurement specialists, understanding these mechanistic shifts is critical for evaluating long-term supply chain stability and cost structures in API manufacturing.
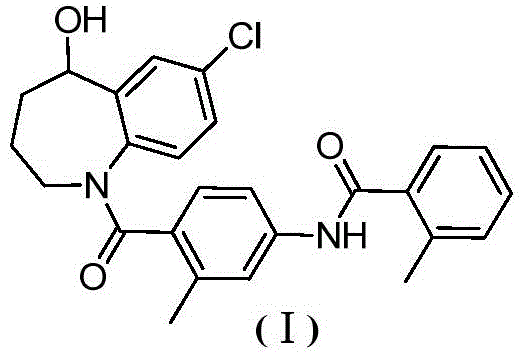
The target molecule, Tolvaptan (Formula I), possesses a complex 1-benzazepine core that has historically presented synthetic challenges regarding regioselectivity and functional group tolerance. The disclosed method addresses these challenges through a linear sequence that builds molecular complexity gradually, ensuring that each intermediate can be rigorously purified before proceeding to the next stage. This approach minimizes the propagation of impurities, a common pain point in multi-step syntheses of heterocyclic pharmaceuticals. By focusing on high atom economy and the use of commodity chemicals, the patent outlines a pathway that is inherently more scalable than previous iterations, positioning it as a viable candidate for commercial scale-up of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of the 7-chloro-5-oxo-2,3,4,5-tetrahydro-1H-1-benzazepine core, a pivotal precursor to Tolvaptan, has relied on methodologies that impose significant economic and environmental burdens. Prior art, including patents such as WO 2007/026971 and academic literature from Bioorganic & Medicinal Chemistry, often employs platinum oxide as a catalyst for key reduction steps. The reliance on platinum group metals (PGMs) introduces volatility into the supply chain due to fluctuating precious metal prices and necessitates stringent removal protocols to meet residual metal specifications in the final drug substance. Furthermore, alternative routes reported in the literature have utilized solvents like 1,2-dichloroethane, which poses severe environmental and safety hazards, complicating waste disposal and increasing the carbon footprint of the manufacturing process. These conventional methods also suffer from operational complexities, such as low yields in rearrangement reactions and difficult-to-source starting materials, which collectively hinder efficient industrial production.
The Novel Approach
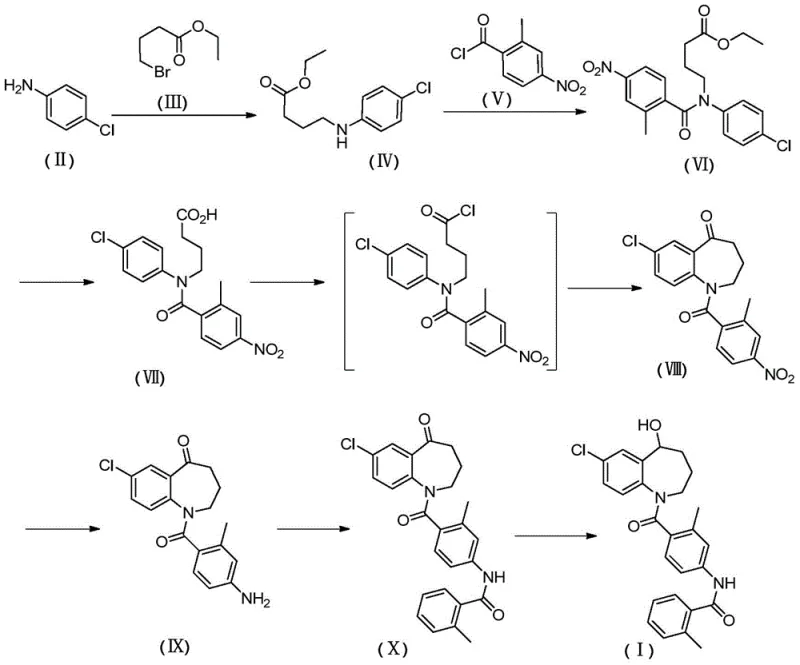
In stark contrast, the novel approach detailed in CN105753735B reconstructs the synthetic logic by initiating the sequence with p-chloroaniline and ethyl 4-bromobutyrate. This strategy bypasses the need for expensive noble metal catalysts entirely, replacing them with more accessible reagents like tin dichloride or sodium borohydride for reduction steps. The route is characterized by a convergent design where the side chain is assembled early, allowing for better control over the final coupling reactions. By shifting the cyclization strategy to a Friedel-Crafts acylation of a pre-functionalized linear precursor, the process achieves higher regioselectivity and reduces the formation of isomeric byproducts. This methodological shift not only simplifies the operational workflow but also aligns with green chemistry principles by reducing the reliance on toxic solvents and hazardous reagents, thereby offering substantial cost savings in pharmaceutical intermediate production.
Mechanistic Insights into Friedel-Crafts Cyclization and Reduction
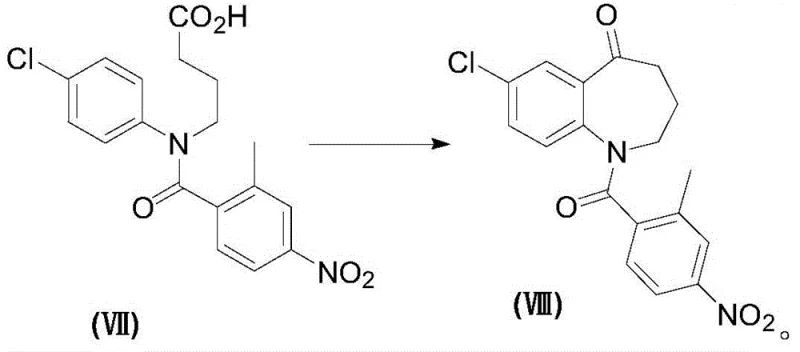
The cornerstone of this synthetic route is the formation of the seven-membered benzazepine ring, achieved through an intramolecular Friedel-Crafts acylation. In this critical transformation, the carboxylic acid intermediate (Formula VII) is first activated, typically using thionyl chloride or phosphorus pentachloride, to generate the corresponding acid chloride in situ. This activated species then undergoes cyclization in the presence of a Lewis acid catalyst, such as aluminum trichloride. The mechanism involves the generation of an acylium ion which attacks the electron-rich aromatic ring ortho to the amino substituent. The choice of aluminum trichloride is particularly advantageous as it provides the necessary Lewis acidity to drive the reaction to completion at moderate temperatures (20°C to 60°C), minimizing thermal degradation of the sensitive nitro group present on the pendant aromatic ring. This step is vital for establishing the core scaffold with high fidelity, ensuring that the subsequent reduction steps proceed without interference from structural impurities.
Following the ring closure, the process requires the selective reduction of the nitro group to an amine without affecting the ketone functionality of the benzazepine ring. The patent discloses the use of tin dichloride in acidic media or catalytic hydrogenation alternatives as effective strategies for this transformation. The chemoselectivity here is paramount; over-reduction of the ketone at this stage would lead to unwanted alcohol byproducts that complicate downstream purification. The subsequent reduction of the ketone to the secondary alcohol (Formula I) is performed in the final stages using sodium borohydride. This late-stage functionalization allows for precise stereochemical control and ensures that the hydroxyl group is introduced only after the more robust amide bonds have been formed. This sequential logic demonstrates a deep understanding of functional group compatibility, resulting in a cleaner impurity profile and higher overall process efficiency.
How to Synthesize Tolvaptan Efficiently
The synthesis of Tolvaptan via this novel route involves a carefully orchestrated sequence of seven distinct chemical transformations, beginning with the alkylation of p-chloroaniline. The process is designed to maximize yield at each stage, with experimental data indicating yields often exceeding 80% for individual steps when optimized conditions are applied. The initial alkylation sets the chain length, followed by acylation to introduce the substituted benzoyl moiety. Hydrolysis converts the ester to an acid, priming the molecule for the crucial cyclization event. Detailed standard operating procedures for each reaction condition, including solvent choices like acetonitrile and dichloromethane, are critical for reproducibility. For a comprehensive guide on executing these reactions with precise stoichiometry and temperature controls, refer to the standardized synthesis steps outlined below.
- React p-chloroaniline with ethyl 4-bromobutyrate to form the alkylated amine intermediate.
- Perform acylation with 2-methyl-4-nitrobenzoyl chloride followed by hydrolysis to generate the carboxylic acid precursor.
- Execute intramolecular Friedel-Crafts cyclization using aluminum trichloride to close the seven-membered ring.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the shift away from platinum-based catalysis represents a significant de-risking of the supply chain. Precious metal catalysts are subject to geopolitical supply constraints and price volatility, which can unpredictably impact the cost of goods sold (COGS). By substituting these with base metal catalysts or hydride reagents, manufacturers can achieve significant cost reduction in API manufacturing while simultaneously simplifying the regulatory filing process regarding elemental impurities. The use of p-chloroaniline as a starting material further enhances supply security, as it is a high-volume commodity chemical produced by multiple global vendors, ensuring that raw material availability remains stable even during market fluctuations. This diversification of the supply base is a key strategic advantage for long-term contract manufacturing agreements.
- Cost Reduction in Manufacturing: The elimination of expensive platinum oxide catalysts removes a major cost driver from the bill of materials. Additionally, the avoidance of specialized solvent recovery systems for 1,2-dichloroethane reduces capital expenditure on waste management infrastructure. The high yields reported in the patent examples suggest that less raw material is wasted per kilogram of finished product, directly improving the gross margin profile. Furthermore, the simplified workup procedures, which often involve standard aqueous extractions rather than complex chromatographic separations, lower the operational labor and utility costs associated with production batches.
- Enhanced Supply Chain Reliability: The reliance on commodity reagents such as ethyl 4-bromobutyrate and aluminum trichloride ensures that the production schedule is not held hostage by the lead times of exotic reagents. These materials are stocked by major chemical distributors worldwide, facilitating just-in-time inventory management and reducing the need for large safety stocks. The robustness of the reaction conditions, which tolerate moderate variations in temperature and stoichiometry, also contributes to consistent batch-to-batch quality, reducing the risk of production delays caused by out-of-specification results.
- Scalability and Environmental Compliance: The process is inherently scalable, having been designed with industrial constraints in mind. The solvents used, primarily dichloromethane and acetonitrile, are well-understood in large-scale reactor operations with established recovery and recycling protocols. By avoiding the generation of heavy metal waste streams, the facility's environmental compliance burden is significantly lightened, reducing the costs associated with hazardous waste disposal. This environmental friendliness aligns with the increasing regulatory pressure on pharmaceutical companies to adopt greener manufacturing practices, enhancing the brand value of the final product.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic route. They are derived from the specific advantages and operational parameters detailed in the patent documentation, providing clarity for technical teams evaluating feasibility. Understanding these nuances is essential for making informed decisions about technology transfer and process validation.
Q: How does this new route improve upon conventional Tolvaptan synthesis methods?
A: Unlike prior art methods that rely on expensive platinum oxide catalysts or environmentally hazardous solvents like 1,2-dichloroethane, this novel route utilizes readily available raw materials such as p-chloroaniline and avoids heavy metal contamination risks, significantly simplifying purification and waste treatment.
Q: What are the critical quality control points in the cyclization step?
A: The cyclization step (Formula VII to VIII) requires precise temperature control between 20°C and 60°C and the use of Lewis acids like aluminum trichloride. Maintaining strict anhydrous conditions during the activation of the acid chloride is essential to prevent hydrolysis and ensure high conversion rates.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process is designed for industrial scalability. It eliminates the need for specialized hydrogenation equipment required by platinum-catalyzed routes and uses standard organic solvents like dichloromethane and acetonitrile, which are easily managed in existing chemical infrastructure.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tolvaptan Intermediate Supplier
The technical advancements described in patent CN105753735B underscore the potential for a more efficient and sustainable supply of Tolvaptan intermediates. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent supply of high-purity intermediates. Our state-of-the-art facilities are equipped with rigorous QC labs capable of detecting trace impurities, guaranteeing that every batch meets stringent purity specifications required for global regulatory submissions. We understand that the transition to a new synthetic route requires confidence in the partner's ability to execute flawlessly at scale.
We invite you to engage with our technical procurement team to discuss how this optimized route can benefit your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to this platinum-free methodology. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments tailored to your volume requirements, ensuring a seamless integration of these high-quality intermediates into your supply chain.
