Advanced Synthesis of Polyhalogenated 2-Alkylthio-4-Amino Quinazolines for Oncology Drug Discovery
The pharmaceutical industry's relentless pursuit of potent anti-tumor agents has increasingly focused on cell cycle regulation mechanisms, specifically targeting the CDC25B phosphatase family. Patent CN107459489B introduces a groundbreaking class of polyhalogenated 2-alkylthio-4-aminoquinazoline compounds that exhibit exceptional inhibitory activity against CDC25B, a key regulator in the G2/M phase transition of the cell cycle. This technology represents a significant leap forward in oncology drug discovery, offering a versatile scaffold that can be finely tuned through various halogen and alkylthio substitutions. The core innovation lies not only in the biological efficacy of the resulting molecules but also in the robustness and flexibility of the synthetic methodology employed to construct them. As a leading entity in fine chemical manufacturing, we recognize the critical importance of such platforms in accelerating the pipeline from hit identification to lead optimization.
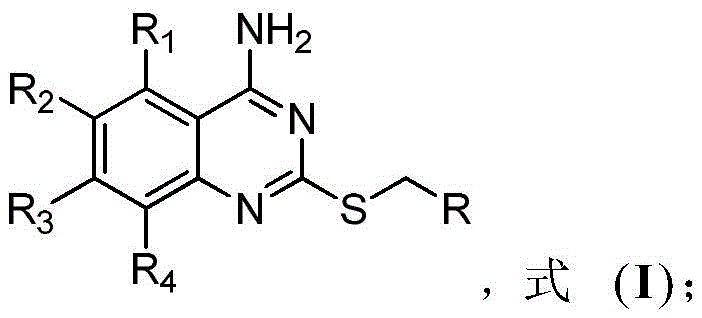
These compounds are defined by a quinazoline core substituted with multiple halogen atoms (fluorine or chlorine) and a diverse range of alkylthio groups at the 2-position. The structural versatility allows for extensive Structure-Activity Relationship (SAR) studies, enabling medicinal chemists to optimize potency, selectivity, and pharmacokinetic properties. The patent highlights that these derivatives serve as promising candidates for treating various malignancies, including colorectal, lung, liver, and gastric cancers, where CDC25B is often overexpressed. For R&D directors seeking reliable pharmaceutical intermediate suppliers, this chemistry offers a stable and well-characterized entry point into next-generation kinase and phosphatase inhibitor programs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of complex quinazoline derivatives has been fraught with challenges that hinder rapid drug development. Conventional routes often involve multi-step sequences requiring harsh reaction conditions, such as high temperatures and strong acidic or basic environments, which can degrade sensitive functional groups. Furthermore, many established methods rely on expensive transition metal catalysts that necessitate rigorous removal steps to meet residual metal specifications for API manufacturing. These legacy processes frequently suffer from poor atom economy, generating substantial chemical waste and increasing the environmental footprint of production. Additionally, the purification of intermediates in traditional syntheses can be labor-intensive, often requiring repeated recrystallizations or complex chromatographic separations that lower overall yields and extend lead times.
The Novel Approach
In stark contrast, the methodology disclosed in CN107459489B offers a streamlined, one-pot solution that addresses these historical bottlenecks. The invention utilizes readily available polyhalogenated benzonitriles and S-substituted isothiourea hydrogen halide salts as starting materials, reacting them under either microwave irradiation with a solid support or conventional heating with an inorganic base. This approach eliminates the need for precious metal catalysts, thereby simplifying the downstream purification process and significantly reducing raw material costs. The reaction conditions are remarkably mild, typically ranging from 60°C to 120°C, which preserves the integrity of sensitive substituents. By achieving high conversion rates in a single step, this novel approach drastically improves atom economy and operational efficiency, making it an ideal candidate for cost reduction in API manufacturing.

Mechanistic Insights into Base-Promoted Cyclization
The chemical transformation relies on a nucleophilic substitution followed by an intramolecular cyclization mechanism. In the microwave-assisted pathway, basic alumina acts not merely as a support but as a heterogeneous base catalyst that activates the isothiourea salt. The amino group of the isothiourea attacks the electron-deficient carbon of the nitrile group on the polyhalogenated benzene ring. This initial addition is facilitated by the electron-withdrawing nature of the halogen substituents, which increase the electrophilicity of the nitrile carbon. Subsequent cyclization occurs through the attack of the ring nitrogen onto the thiourea carbon, closing the quinazoline ring system. The use of microwave energy provides rapid, uniform heating that accelerates molecular motion and collision frequency, driving the reaction to completion in minutes rather than hours.
Alternatively, the homogeneous pathway utilizes inorganic bases like cesium carbonate or potassium carbonate in aprotic solvents such as 1,4-dioxane. Here, the base deprotonates the isothiourea salt, generating a highly reactive nucleophile that attacks the benzonitrile substrate. This mechanism allows for precise control over reaction kinetics and is particularly advantageous for scaling up reactions where microwave penetration depth might be a limiting factor. The choice of base and solvent system is critical for minimizing side reactions, such as hydrolysis of the nitrile group or displacement of the halogen atoms, ensuring a clean impurity profile. This mechanistic understanding is vital for process chemists aiming to ensure commercial scale-up of complex pharmaceutical intermediates.

How to Synthesize Polyhalogenated Quinazolines Efficiently
The synthesis protocol outlined in the patent provides two distinct yet complementary pathways to access the target library, catering to both rapid screening and bulk production needs. The process begins with the precise stoichiometric mixing of the polyhalogenated benzonitrile and the S-substituted isothiourea salt, typically in a molar ratio of 1:1.2 to 1:1.5 to ensure complete consumption of the limiting reagent. Depending on the desired throughput, the reaction mixture is either adsorbed onto basic alumina for microwave irradiation or dissolved in 1,4-dioxane with an inorganic base for thermal reflux. Following the reaction, the workup is straightforward, involving either direct column chromatography for the solid-phase method or water precipitation followed by filtration for the homogeneous method. The detailed standardized synthesis steps for implementing this technology in your laboratory are provided below.
- Dissolve polyhalogenated benzonitrile and S-substituted isothiourea hydrogen halide in ethanol or 1,4-dioxane.
- Add basic alumina for solid-phase microwave reaction OR add inorganic base (Cs2CO3/K2CO3) for homogeneous heating.
- Monitor by TLC, purify via column chromatography or water precipitation, and dry to obtain the target quinazoline derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this synthetic technology offers compelling advantages that directly impact the bottom line and operational resilience. The reliance on commercially available starting materials, such as tetrafluoroisophthalonitrile and various isothiourea salts, ensures a stable and secure supply chain, mitigating the risks associated with custom synthesis of exotic precursors. The elimination of transition metal catalysts removes a significant cost center related to both the purchase of expensive metals and the implementation of scavenging technologies required for their removal. Furthermore, the high atom economy and simplified purification protocols translate to reduced solvent consumption and waste disposal costs, aligning with modern green chemistry initiatives and regulatory expectations.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by utilizing inexpensive inorganic bases like potassium carbonate or solid supports like alumina instead of costly organometallic catalysts. The one-pot nature of the reaction reduces labor hours and equipment usage time, while the high yields reported (ranging from 55% to 93%) maximize the output per unit of raw material input. This efficiency is crucial for maintaining competitive pricing in the generic and specialty pharmaceutical markets.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions, which tolerate a wide range of functional groups and substituents, ensures consistent batch-to-batch quality. The availability of multiple synthetic routes (microwave vs. thermal) provides flexibility in manufacturing planning, allowing production to continue even if specific equipment is unavailable. This redundancy is a key factor in reducing lead time for high-purity pharmaceutical intermediates and ensuring uninterrupted supply to downstream drug manufacturers.
- Scalability and Environmental Compliance: The homogeneous route using 1,4-dioxane and inorganic bases is inherently scalable to industrial reactors, facilitating the transition from gram-scale research to kilogram-scale production. The workup procedures, which often involve simple precipitation or standard chromatography, generate less hazardous waste compared to traditional methods involving heavy metals. This alignment with environmental compliance standards reduces the regulatory burden and potential liabilities associated with chemical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these polyhalogenated quinazoline derivatives. These answers are derived directly from the experimental data and technical specifications provided in the patent literature, ensuring accuracy and relevance for decision-makers. Understanding these details is essential for evaluating the feasibility of integrating this chemistry into your existing drug discovery or manufacturing workflows.
Q: What are the primary advantages of the microwave-assisted synthesis route?
A: The microwave-assisted route utilizing basic alumina significantly reduces reaction time to 5-30 minutes compared to traditional reflux methods, offering higher energy efficiency and rapid library generation for SAR studies.
Q: How does this method improve impurity profiles for API intermediates?
A: By employing a one-pot cyclization strategy with high atom economy, the process minimizes side reactions and byproduct formation, resulting in crude products that are easier to purify to stringent pharmaceutical specifications.
Q: Is this synthesis scalable for commercial production?
A: Yes, the patent discloses a parallel homogeneous route using inorganic bases like cesium carbonate in 1,4-dioxane, which is highly amenable to scale-up from gram to kilogram quantities without requiring specialized microwave equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polyhalogenated Quinazoline Supplier
At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from the bench to the plant. Our commitment to quality is underscored by our stringent purity specifications and rigorous QC labs, which utilize advanced analytical techniques to verify the identity and purity of every batch. We understand that in the competitive landscape of oncology drug development, time and quality are paramount, and our infrastructure is designed to meet these demands with precision and reliability.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next-generation anti-tumor programs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities can accelerate your timeline and optimize your budget for high-value pharmaceutical intermediates.
