Advanced Camphorylimidazole Ionic Liquids for Efficient Ester Manufacturing and Scale-Up
Advanced Camphorylimidazole Ionic Liquids for Efficient Ester Manufacturing and Scale-Up
The chemical industry is currently witnessing a significant paradigm shift towards greener, more efficient catalytic systems, particularly in the synthesis of high-value ester compounds. Patent CN110272344B introduces a groundbreaking class of camphorylimidazole type ionic liquids that serve as highly effective catalysts for aldehyde-alcohol oxidation esterification reactions. This technology leverages the unique structural properties of natural camphor derivatives combined with imidazolium cations to create a robust catalytic environment. Unlike conventional methods that often rely on corrosive strong acids or toxic heavy metals, this novel approach utilizes renewable resources to achieve superior reaction kinetics. The general structural formula of these ionic liquids, featuring a rigid camphor backbone linked to an imidazolium ring, provides exceptional stability and tunable solubility profiles.
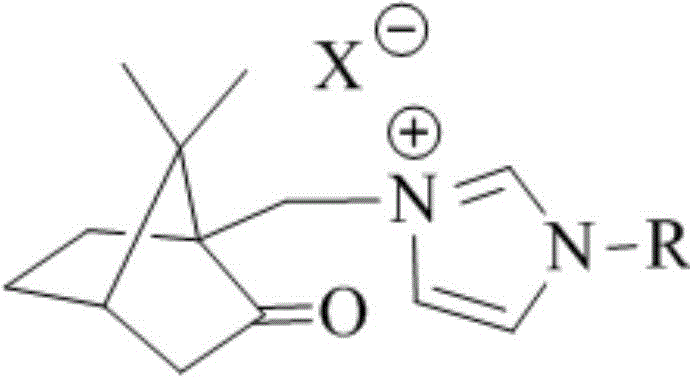
For procurement managers and supply chain directors, the implications of this technology are profound. By transitioning to this bio-based catalytic system, manufacturers can significantly reduce their reliance on petrochemical-derived catalysts and mitigate the environmental hazards associated with traditional esterification processes. The ability to tune the anion component (such as PF6-, BF4-, or N(Tf)2-) allows for precise optimization of the catalyst for specific solvent systems and reaction conditions. This flexibility ensures that the technology can be seamlessly integrated into existing production lines for pharmaceutical intermediates and fine chemicals, offering a reliable pathway to cost reduction in fine chemical manufacturing without compromising on product purity or yield.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of ester compounds has been plagued by significant technical and economic inefficiencies inherent in traditional methodologies. The most common approach involves the direct dehydration and esterification of carboxylic acids and alcohols under the action of strong mineral acids. This method frequently suffers from low reaction efficiency, requiring harsh conditions that can degrade sensitive functional groups, leading to complex impurity profiles that are difficult and expensive to remove. Furthermore, alternative pathways such as activating carboxylic acids to acyl halides or anhydrides prior to nucleophilic attack by alcohols exhibit poor atom economy. These processes generate substantial amounts of stoichiometric waste, such as hydrochloric acid or carboxylic acid byproducts, necessitating extensive downstream processing and waste treatment infrastructure.
Another prevalent method utilizes condensation reagents like DCC (dicyclohexylcarbodiimide), which, while effective for small-scale laboratory synthesis, presents severe challenges for commercial scale-up. The formation of insoluble urea byproducts complicates filtration and purification, drastically increasing operational costs and extending production lead times. Additionally, the use of transition metal catalysts in oxidative esterification often raises concerns regarding residual metal contamination in the final product, a critical issue for pharmaceutical applications where strict regulatory limits apply. These cumulative drawbacks highlight the urgent need for a more sustainable, efficient, and economically viable catalytic system that can overcome the limitations of atom economy and functional group tolerance.
The Novel Approach
The camphorylimidazole type ionic liquid represents a transformative solution to these longstanding challenges by introducing a metal-free, organocatalytic system derived from renewable resources. The synthetic route begins with the conversion of camphorsulfonic acid into 10-iodocamphor, followed by a quaternization reaction with aryl imidazoles to form the core ionic liquid structure. This process is depicted in the comprehensive reaction scheme below, illustrating the straightforward transformation from natural precursors to high-performance catalysts.
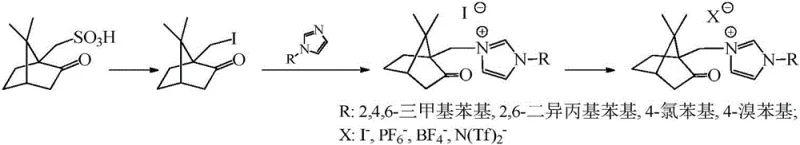
This novel approach offers distinct advantages in terms of reaction selectivity and operational simplicity. The bulky camphor skeleton provides a unique steric environment that enhances the selectivity of the oxidation esterification reaction, minimizing side reactions and byproduct formation. Consequently, the reaction proceeds under remarkably mild conditions, typically requiring temperatures between 50°C and 70°C and atmospheric pressure, which significantly reduces energy consumption compared to traditional high-temperature reflux methods. The high catalytic efficiency observed in patent examples, with yields reaching up to 98% for various substrates, demonstrates the robustness of this system. Moreover, the ionic nature of the catalyst facilitates easy separation from organic products, potentially allowing for catalyst recycling and further enhancing the overall economic viability of the process for large-scale industrial applications.
Mechanistic Insights into Camphorylimidazole-Catalyzed Oxidation Esterification
The exceptional performance of camphorylimidazole ionic liquids in catalyzing aldehyde-alcohol oxidation esterification can be attributed to the synergistic interaction between the imidazolium cation and the specific anion within the reaction medium. The general reaction mechanism involves the activation of the aldehyde substrate by the ionic liquid, facilitating the nucleophilic attack by the alcohol molecule in the presence of an oxidant (typically air or oxygen supplied via an air balloon in the described examples). The rigid bicyclic structure of the camphor moiety imposes conformational constraints that likely stabilize the transition state of the reaction, thereby lowering the activation energy required for the esterification process. This structural rigidity also prevents the catalyst from undergoing degradation under the oxidative conditions, ensuring consistent performance throughout the reaction duration.
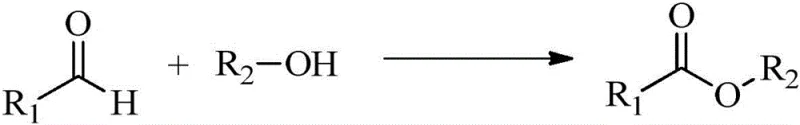
Furthermore, the tunability of the anion plays a crucial role in modulating the solubility and polarity of the catalyst, which directly influences the reaction kinetics and substrate scope. For instance, the use of hydrophobic anions like hexafluorophosphate (PF6-) or bis(trifluoromethanesulfonyl)imide (N(Tf)2-) enhances the compatibility of the catalyst with organic solvents such as toluene, creating a homogeneous reaction phase that maximizes contact between reactants. This homogeneity is critical for achieving the high conversion rates reported in the patent data. Additionally, the absence of transition metals eliminates the risk of metal-catalyzed side reactions, such as over-oxidation or polymerization, which are common pitfalls in traditional oxidative esterification. The result is a clean reaction profile with minimal impurity generation, simplifying the downstream purification process and ensuring the production of high-purity ester intermediates suitable for sensitive pharmaceutical applications.
How to Synthesize Camphorylimidazole Ionic Liquid Efficiently
The preparation of these advanced catalysts follows a logical three-step sequence that is amenable to standard chemical manufacturing equipment. The process begins with the iodination of camphorsulfonic acid, a reaction that requires careful control of temperature and stoichiometry to ensure high purity of the 10-iodocamphor intermediate. Following isolation and purification, the iodide undergoes quaternization with various substituted imidazoles to generate the precursor salt. The final step involves an ion exchange reaction to introduce the desired counter-anion, completing the synthesis of the target ionic liquid. Detailed standardized synthesis steps see the guide below.
- React camphorsulfonic acid with iodine and triphenyl phosphine in toluene at 90-110°C to synthesize 10-iodocamphor.
- Perform a quaternization reaction between the synthesized 10-iodocamphor and aryl imidazole in DMF at 130-150°C to form the iodide salt.
- Conduct an ion exchange reaction using an anion exchanger (e.g., sodium hexafluorophosphate) in dichloromethane to obtain the final ionic liquid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of camphorylimidazole ionic liquid technology offers a strategic advantage in terms of cost stability and operational efficiency. The reliance on camphor, a naturally occurring and renewable resource, mitigates the volatility associated with petrochemical feedstocks, providing a more predictable cost structure for long-term production planning. The elimination of expensive transition metal catalysts and the reduction in waste generation translate directly into lower raw material costs and reduced expenditure on waste disposal and environmental compliance. This shift towards a greener manufacturing process not only aligns with corporate sustainability goals but also reduces the regulatory burden associated with handling hazardous chemicals.
- Cost Reduction in Manufacturing: The implementation of this catalytic system significantly lowers the overall cost of goods sold by streamlining the production workflow. By operating under mild conditions, the process reduces energy consumption associated with heating and cooling, while the high selectivity minimizes the loss of valuable starting materials to byproducts. The simplified purification process, driven by the high purity of the crude reaction mixture, reduces the requirement for extensive chromatography or recrystallization steps, thereby saving on solvent usage and labor costs. Furthermore, the potential for catalyst recovery and reuse, inherent to many ionic liquid systems, offers an additional avenue for long-term cost optimization that is not available with single-use homogeneous acid catalysts.
- Enhanced Supply Chain Reliability: Sourcing raw materials from renewable biological sources like camphor diversifies the supply chain and reduces dependency on fluctuating oil markets. The synthetic route is robust and utilizes readily available reagents such as iodine, triphenylphosphine, and common aryl imidazoles, ensuring that production is not bottlenecked by scarce specialty chemicals. This reliability is crucial for maintaining continuous manufacturing operations and meeting tight delivery schedules for downstream customers in the pharmaceutical and agrochemical sectors. The stability of the final ionic liquid products also simplifies storage and transportation logistics, as they do not require the stringent handling precautions necessary for corrosive acids or pyrophoric reagents.
- Scalability and Environmental Compliance: The process described in the patent is inherently scalable, utilizing standard reactor configurations and solvent systems that are familiar to chemical engineers. The absence of heavy metals simplifies the environmental impact assessment and facilitates easier permitting for new production facilities. Waste streams are less toxic and easier to treat, reducing the liability and cost associated with environmental remediation. As global regulations on chemical manufacturing become increasingly stringent, adopting this green chemistry technology positions manufacturers as leaders in sustainability, enhancing their brand reputation and market competitiveness while ensuring compliance with future regulatory frameworks.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of camphorylimidazole ionic liquids in industrial settings. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing a clear understanding of the technology's capabilities and limitations. Understanding these details is essential for R&D teams evaluating the feasibility of integrating this catalyst into their existing synthetic routes.
Q: What are the primary advantages of using camphorylimidazole ionic liquids over traditional acid catalysts?
A: Unlike traditional strong acids which suffer from low efficiency and poor functional group tolerance, camphorylimidazole ionic liquids offer high catalytic activity under mild conditions (50-70°C) with excellent selectivity and yields up to 98%.
Q: Can this catalyst be used for both aromatic and aliphatic aldehydes?
A: Yes, the patent data demonstrates broad substrate scope, successfully catalyzing the oxidation esterification of various aromatic aldehydes (e.g., benzaldehyde, nitrobenzaldehyde) and aliphatic aldehydes (e.g., n-propionaldehyde).
Q: Is the raw material source for this catalyst sustainable?
A: Yes, the synthesis begins with camphorsulfonic acid, which is derived from natural renewable resources (camphor), aligning with green chemistry principles and ensuring a stable supply chain.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Camphorylimidazole Ionic Liquid Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the camphorylimidazole ionic liquid system in driving innovation within the fine chemical and pharmaceutical industries. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to industrial reality is seamless and efficient. Our state-of-the-art facilities are equipped to handle the synthesis of complex ionic liquids with stringent purity specifications, supported by rigorous QC labs that guarantee every batch meets the highest international standards. We are committed to delivering not just a product, but a comprehensive solution that enhances your manufacturing efficiency and product quality.
We invite you to collaborate with our technical team to explore how this novel catalyst can optimize your specific esterification processes. Whether you require a Customized Cost-Saving Analysis for your current production line or need to evaluate the feasibility of this route for a new API intermediate, our experts are ready to assist. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and sample quantities for your evaluation. Let us help you leverage this cutting-edge technology to achieve superior economic and environmental outcomes in your chemical manufacturing operations.
