Advanced Pyrazole-Linked Triazole Phosphine Ligands for High-Efficiency Coupling Reactions
Introduction to Patent CN111116653A Technology
The landscape of transition metal-catalyzed cross-coupling reactions has been revolutionized by the development of specialized ligands that enhance both the efficiency and selectivity of bond formation. Patent CN111116653A discloses a groundbreaking preparation method for pyrazole-linked triazole phosphine compounds, representing a significant leap forward in ligand design for fine chemical synthesis. These novel compounds address critical limitations found in earlier generations of phosphine ligands, offering a robust platform for constructing complex molecular architectures essential in the pharmaceutical and agrochemical sectors. The technology described herein provides a reliable pathway to synthesize ligands that maintain stability under ambient conditions while delivering exceptional catalytic performance in demanding coupling environments.
For R&D directors and process chemists, the implications of this patent are profound, as it offers a solution to the persistent challenges of catalyst deactivation and limited substrate scope. The core innovation lies in the unique structural motif combining a pyrazole ring with a triazole moiety, linked to a phosphine group. This architecture not only stabilizes the active catalytic species but also facilitates the oxidative addition and reductive elimination steps crucial for catalytic cycles. By leveraging this technology, manufacturers can achieve higher purity profiles and more consistent batch-to-batch reproducibility, which are paramount metrics for any reliable pharmaceutical intermediates supplier aiming to meet stringent regulatory standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the field of cross-coupling catalysis has relied heavily on ligands such as Buchwald ligands, biphenyl monophosphines, and general dialkyl phosphines. While effective in many contexts, these traditional ligands suffer from inherent drawbacks that hinder large-scale industrial adoption. A primary concern is their susceptibility to oxidation; many dialkyl phosphines are highly air-sensitive, requiring rigorous exclusion of oxygen and moisture throughout the synthesis and storage phases, which drastically increases operational complexity and cost. Furthermore, the synthesis of sophisticated biaryl phosphine ligands often involves multi-step sequences with low overall yields, making them economically prohibitive for cost-sensitive applications like generic drug manufacturing.
Another significant limitation is the narrow substrate scope associated with older ligand classes. In complex molecule synthesis, particularly when dealing with sterically hindered amines or electron-deficient aryl halides, conventional ligands often fail to drive the reaction to completion, resulting in residual starting materials and difficult-to-remove impurities. This inefficiency necessitates extensive downstream purification, consuming valuable time and resources. Additionally, the inability to produce certain high-performance ligands on a large scale has created supply chain bottlenecks, forcing procurement teams to rely on limited sources with long lead times. These factors collectively contribute to elevated production costs and reduced agility in responding to market demands for new therapeutic candidates.
The Novel Approach
The novel approach detailed in the patent introduces a pyrazole-triazole phosphine scaffold that effectively overcomes these historical barriers. By integrating a heterocyclic triazole ring directly onto the pyrazole core, the resulting ligand exhibits enhanced electronic and steric properties that stabilize the transition metal center. This structural modification significantly reduces the tendency for phosphine oxidation, allowing for easier handling and storage without the need for extreme inert atmosphere conditions typically required for sensitive phosphines. The synthesis route is designed for scalability, utilizing readily available starting materials and straightforward reaction conditions that are amenable to kilogram-scale production.
Moreover, this new class of ligands demonstrates remarkable versatility across a broad spectrum of coupling reactions. As illustrated in the reaction schemes, these compounds facilitate efficient C-N bond formation in Buchwald-Hartwig aminations, even with challenging substrates like secondary amines and sterically crowded aryl halides. The improved catalytic activity means that lower loadings of expensive palladium catalysts can be employed without sacrificing yield or reaction rate. This translates directly into cost reduction in pharmaceutical intermediates manufacturing, as the expense associated with precious metal recovery and removal is minimized. The ability to achieve high conversion rates under milder conditions also supports the synthesis of thermally sensitive compounds, expanding the utility of this technology in the production of complex active pharmaceutical ingredients.
Mechanistic Insights into Phosphinylation and Catalytic Stability
The synthesis of the target pyrazole-triazole phosphine compounds involves a critical phosphinylation step that defines the final structure and performance of the ligand. This transformation typically proceeds via the deprotonation of the pyrazole-triazole precursor using a strong base such as n-butyllithium at cryogenic temperatures, specifically around -78°C in tetrahydrofuran (THF). This low-temperature lithiation generates a reactive nucleophilic species at the triazole ring, which subsequently attacks a chlorophosphine electrophile, such as di-tert-butylphosphine chloride or dicyclohexylphosphine chloride. The precision required in this step underscores the importance of strict temperature control and anhydrous conditions to prevent side reactions and ensure high regioselectivity.
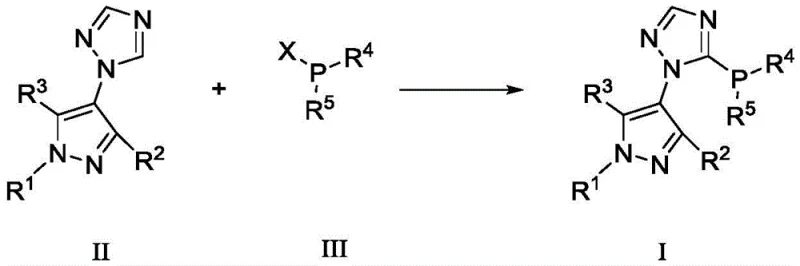
Once formed, the phosphine moiety benefits from the steric bulk provided by the adjacent substituents, such as tert-butyl or cyclohexyl groups. This steric shielding protects the phosphorus atom from aerial oxidation, a common degradation pathway for many phosphine ligands. Mechanistically, during the catalytic cycle, the ligand coordinates to the palladium center, modulating its electron density to facilitate the oxidative addition of the aryl halide. The electron-rich nature of the triazole-pyrazole system enhances the nucleophilicity of the metal center, accelerating the rate-determining steps of the cycle. Furthermore, the rigid structure of the ligand helps maintain the coordination geometry required for efficient reductive elimination, thereby minimizing the formation of homocoupling by-products. This mechanistic robustness ensures that the catalyst remains active for longer periods, supporting higher turnover numbers and improved process economics.
How to Synthesize Pyrazole-Linked Triazole Phosphine Efficiently
The preparation of these high-value ligands follows a logical sequence of organic transformations that can be optimized for industrial throughput. The process begins with the halogenation of a diketone precursor, followed by substitution with a triazole salt and subsequent cyclization to form the core heterocyclic structure. Alkylation of the pyrazole nitrogen allows for fine-tuning of the ligand's solubility and steric profile. The final and most critical step involves the introduction of the phosphine group under strictly controlled inert conditions. Detailed standardized synthetic steps for the preparation of Ligand L1 and L2 are provided in the guide below, ensuring reproducibility and safety during scale-up operations.
- Perform halogenation of dibenzoylmethane derivatives with bromine in NMP at room temperature to obtain alpha-bromo intermediates.
- Execute triazolyl substitution using 1,2,4-triazole sodium salt in DMF at 45°C, followed by cyclization with hydrazine hydrate.
- Conduct alkylation of the pyrazole-triazole core with alkyl halides using potassium carbonate in NMP.
- Complete the synthesis via phosphinylation using n-butyllithium at -78°C in THF, followed by reaction with chlorophosphines.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel ligand technology offers tangible strategic benefits beyond mere technical performance. The streamlined synthesis route reduces the number of unit operations required compared to traditional biaryl phosphine ligands, leading to a significantly simplified manufacturing process. This simplification directly correlates with lower capital expenditure for equipment and reduced labor costs per kilogram of product. Furthermore, the enhanced stability of the final ligand product minimizes losses due to degradation during storage and transportation, ensuring that the material received at the manufacturing site retains its full catalytic potency. This reliability is crucial for maintaining continuous production schedules and avoiding costly downtime caused by reagent failure.
- Cost Reduction in Manufacturing: The implementation of these ligands enables a substantial decrease in the consumption of expensive palladium catalysts. Due to their high activity and selectivity, the catalyst loading can be reduced while maintaining excellent reaction yields, which drastically lowers the raw material cost per batch. Additionally, the reduced need for rigorous exclusion of air and moisture during handling translates to lower operational costs associated with inert gas consumption and specialized glovebox equipment. The overall process efficiency is further enhanced by the ease of purification, as the high selectivity minimizes the formation of difficult-to-separate impurities, reducing solvent usage and waste disposal costs associated with chromatography or recrystallization steps.
- Enhanced Supply Chain Reliability: The synthetic route relies on commercially available starting materials such as dibenzoylmethane derivatives and common chlorophosphines, mitigating the risk of supply disruptions associated with exotic or proprietary reagents. The robustness of the synthesis allows for flexible production scheduling, as the intermediates are stable and can be stockpiled if necessary. This flexibility empowers supply chain planners to respond more agilely to fluctuations in demand for downstream pharmaceutical intermediates. Moreover, the scalability of the process from gram to multi-kilogram scales ensures that suppliers can meet increasing volume requirements without the need for extensive process re-engineering, guaranteeing a steady flow of critical materials to the production line.
- Scalability and Environmental Compliance: The preparation method is designed with environmental sustainability in mind, utilizing solvents and reagents that are manageable within standard waste treatment protocols. The high atom economy of the coupling reactions facilitated by these ligands results in less chemical waste generation per unit of product, aligning with green chemistry principles and reducing the environmental footprint of the manufacturing facility. The ability to operate at moderate temperatures and pressures further enhances safety profiles, lowering the risk of thermal runaways and ensuring compliance with stringent occupational health and safety regulations. This combination of scalability and environmental responsibility makes the technology an attractive option for companies aiming to optimize their ESG (Environmental, Social, and Governance) metrics while maintaining competitive production costs.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the application and handling of pyrazole-triazole phosphine ligands. These answers are derived directly from the experimental data and specifications outlined in the patent documentation, providing clarity on performance expectations and operational parameters. Understanding these details is essential for process engineers and quality assurance teams to integrate this technology effectively into existing workflows.
Q: How does the stability of pyrazole-triazole phosphine ligands compare to traditional Buchwald ligands?
A: The pyrazole-triazole phosphine compounds exhibit superior stability due to the steric hindrance provided by the aryl groups below the phosphorus atom, making them less susceptible to air oxidation compared to conventional dialkyl phosphine ligands.
Q: What types of coupling reactions are these ligands suitable for?
A: These ligands are highly versatile and effective for C-N, C-C, C-S, and C-B bond formation, specifically excelling in Buchwald-Hartwig amination, Suzuki coupling, Heck reactions, and Negishi coupling.
Q: Can these ligands reduce palladium catalyst loading in industrial processes?
A: Yes, the high selectivity and catalytic efficiency of these ligands allow for significantly reduced usage amounts of transition metal catalysts while maintaining high reaction rates and conversion yields.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrazole-Triazole Phosphine Supplier
At NINGBO INNO PHARMCHEM, we recognize the pivotal role that advanced ligands play in modern synthetic chemistry. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We are committed to delivering high-purity pyrazole-triazole phosphine compounds that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our dedication to quality ensures that every batch of ligand supplied performs consistently, enabling our partners to achieve reproducible results in their critical coupling reactions.
We invite you to collaborate with us to explore how this innovative ligand technology can optimize your specific synthesis routes. Our technical sales team is ready to provide a Customized Cost-Saving Analysis tailored to your current manufacturing processes, highlighting potential efficiencies and economic benefits. Please contact our technical procurement team to request specific COA data for our available ligand grades and to discuss route feasibility assessments for your target molecules. Together, we can drive innovation and efficiency in the production of next-generation pharmaceutical and agrochemical intermediates.
