Advanced Synthesis of Cefotiam Hydrochloride: A Technical Breakthrough for Industrial Scale-Up
Advanced Synthesis of Cefotiam Hydrochloride: A Technical Breakthrough for Industrial Scale-Up
The pharmaceutical industry continuously seeks robust synthetic routes for second-generation cephalosporins, particularly cefotiam hydrochloride, due to its broad-spectrum antibacterial efficacy. A pivotal advancement in this domain is detailed in patent CN109517002B, which discloses a highly efficient synthesis method that addresses longstanding challenges in yield and purity. This technical insight report analyzes the proprietary methodology, focusing on the strategic use of a high-concentration boron trifluoride dimethyl carbonate complex as a catalyst. By optimizing the condensation reaction between 7-aminocephalosporanic acid (7-ACA) and 1-(2-dimethylaminoethyl)-1H-5-mercapto-tetrazole (DMMT), the process achieves a stable, mild reaction environment that significantly minimizes by-product generation. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediates supplier, understanding these mechanistic improvements is crucial for securing a consistent supply chain of high-quality active pharmaceutical ingredients.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of cefotiam hydrochloride has been plagued by inefficiencies inherent in traditional catalytic systems. Prior art methods often relied on sodium bicarbonate or simple boron trifluoride catalysis for the 3-position condensation, which frequently resulted in suboptimal total yields and unsatisfactory product purity profiles. Alternative approaches, such as those utilizing methanesulfonic acid, managed to improve yield but at the cost of significantly prolonged reaction times, creating bottlenecks in production throughput. Furthermore, other established routes involving 4-chloro-3-oxobutanoyl chloride (COBC) presented severe logistical hurdles due to transportation difficulties, while mixed anhydride methods necessitated ultra-low temperature conditions that are energy-intensive and difficult to maintain on a commercial scale. These limitations collectively hindered the ability to achieve cost reduction in pharmaceutical intermediates manufacturing while maintaining rigorous quality standards.
The Novel Approach
The methodology outlined in patent CN109517002B introduces a paradigm shift by employing a sophisticated solvent and catalyst system that overcomes these historical barriers. The core innovation lies in the use of a mixed solvent system comprising dimethyl carbonate and specific organic acids, such as citric or tartaric acid, paired with a boron trifluoride-dimethyl carbonate complex. This combination not only adjusts the polarity and proton concentration of the reaction medium to enhance solubility but also exerts a controlled inhibition effect that stabilizes the reaction kinetics. Consequently, the process operates under mild conditions of 20-30°C, drastically shortening the overall reaction time compared to acidic alternatives. This novel approach ensures that the resulting 3-position intermediate and the final cefotiam hydrochloride possess higher purity and stable quality, making it an ideal candidate for the commercial scale-up of complex antibiotics.
Mechanistic Insights into BF3-DMC Catalyzed Condensation and Acylation
The chemical elegance of this synthesis is rooted in the precise manipulation of reaction environments to favor nucleophilic substitution while suppressing degradation pathways. In the initial step, the boron trifluoride-dimethyl carbonate complex acts as a potent Lewis acid catalyst, activating the 3-position of the 7-ACA nucleus for substitution by the mercapto-tetrazole moiety. Unlike traditional strong acids that might promote ring opening or degradation of the beta-lactam structure, this complex facilitates a homogeneous reaction with high selectivity. The addition of organic acid co-solvents further modulates the proton activity, ensuring that the reaction proceeds smoothly without the violent exotherms often associated with cephalosporin modifications. This controlled environment is critical for minimizing the formation of polymeric impurities and isomers that typically complicate downstream purification efforts.
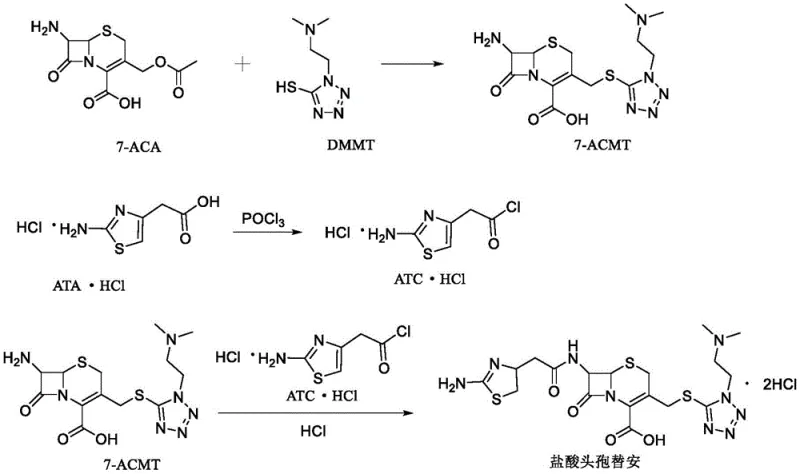
Furthermore, the final acylation step demonstrates exceptional process control through a multi-stage pH adjustment strategy. The reaction between the 7-ACMT intermediate and aminothiazole acetyl chloride hydrochloride is conducted in a polar mixed solvent of acetonitrile and water. Crucially, the acid-binding agent is added in three distinct batches to adjust the pH sequentially to ranges of 6.0-7.0, 5.5-6.5, and 4.5-5.0. This graduated approach neutralizes the hydrochloric acid generated during amide bond formation incrementally, preventing local acidity spikes that could degrade the sensitive beta-lactam ring. By maintaining the reaction temperature between -10°C and 0°C and utilizing this stepped pH control, the process ensures thorough conversion and superior impurity profiles, with maximum single impurity contents reported as low as 0.04%. This level of control is essential for producing high-purity API intermediates that meet stringent global regulatory requirements.
How to Synthesize Cefotiam Hydrochloride Efficiently
Implementing this synthesis route requires strict adherence to the specified stoichiometric ratios and temperature controls to replicate the high yields observed in the patent examples. The process begins with the preparation of the 7-ACMT intermediate, followed by the in-situ or separate generation of the acyl chloride, and concludes with the coupling reaction. Operators must pay particular attention to the solvent ratios, specifically the 6-8:1 molar ratio of the boron trifluoride complex to 7-ACA, and the precise timing of the reducing agent addition to quench residual oxidants. The following guide outlines the standardized operational procedure derived from the patent data, serving as a foundational reference for process engineers aiming to optimize their production lines.
- Condense 7-ACA and DMMT in dimethyl carbonate with an organic acid co-solvent and boron trifluoride-dimethyl carbonate complex catalyst at 20-30°C to form 7-ACMT.
- React aminothiazole acetic acid hydrochloride with phosphorus oxychloride in dichloromethane using amine and iodide catalysts to generate aminothiazole acetyl chloride hydrochloride.
- Couple 7-ACMT with the acyl chloride in an acetonitrile-water system, adjusting pH in three stages at -10 to 0°C, followed by crystallization to obtain the final product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthesis method offers substantial strategic benefits for procurement managers and supply chain heads focused on efficiency and reliability. The elimination of ultra-low temperature requirements, which are necessary in competing mixed anhydride methods, translates directly into reduced energy consumption and lower capital expenditure on specialized cryogenic equipment. Additionally, the use of stable, commercially available solvents like dimethyl carbonate and dichloromethane simplifies raw material sourcing and inventory management. The robust nature of the reaction conditions means that the process is less susceptible to batch-to-batch variability, thereby enhancing supply chain continuity and reducing the risk of production delays caused by failed batches or extensive rework.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by streamlining the purification workflow. Because the reaction generates fewer by-products and achieves higher crude purity (up to 99.8% in examples), the need for extensive recrystallization or chromatographic purification is minimized. This reduction in downstream processing steps lowers solvent consumption and waste disposal costs. Furthermore, the high total molar yield of approximately 83-84% based on 7-ACA ensures better utilization of expensive starting materials, directly improving the cost-of-goods-sold (COGS) profile for the final active ingredient.
- Enhanced Supply Chain Reliability: The reliance on common industrial reagents rather than unstable or hazardous intermediates like COBC enhances the resilience of the supply chain. The method avoids the use of liquid chlorinating agents that require difficult handling protocols, substituting them with solid or stable liquid alternatives that are easier to transport and store. This stability allows for more flexible production scheduling and reduces the lead time associated with procuring specialty chemicals, ensuring that manufacturing timelines are met consistently even in volatile market conditions.
- Scalability and Environmental Compliance: The mild reaction conditions and the absence of heavy metal catalysts make this route highly scalable and environmentally compliant. The simplified workup procedures, which involve straightforward extraction and crystallization, reduce the volume of organic waste generated per kilogram of product. This aligns with modern green chemistry principles and facilitates easier compliance with increasingly stringent environmental regulations regarding solvent emissions and waste treatment, positioning manufacturers as responsible partners in the global pharmaceutical supply network.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this synthesis technology. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on how this method outperforms legacy processes in terms of yield, purity, and operational safety. Understanding these nuances is vital for technical teams evaluating the feasibility of technology transfer.
Q: What are the key advantages of using boron trifluoride-dimethyl carbonate complex in this synthesis?
A: According to patent CN109517002B, using this complex catalyst instead of traditional acids allows for milder reaction conditions (20-30°C), significantly reduces by-product formation, and improves the overall yield and purity of the 7-ACMT intermediate.
Q: How does the new method improve impurity control compared to prior art?
A: The method employs a specific three-stage pH adjustment strategy during the final acylation step and utilizes a polar mixed solvent system. This controls the proton concentration effectively, preventing violent reactions and ensuring a maximum single impurity content as low as 0.04-0.08%.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the process avoids the need for special equipment or ultra-low temperatures required by mixed anhydride methods. The use of stable solvents like dimethyl carbonate and dichloromethane, along with mild temperature ranges, makes it highly scalable and safe for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefotiam Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global antibiotic market. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patent CN109517002B are fully realized in practical manufacturing settings. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch against the highest industry standards.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic advantages specific to your production volume. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your project moves forward with the most efficient and reliable chemical solution available.
