Scalable Asymmetric Synthesis of 2(S)-Glutaric Acid Derivatives for Cardiovascular Drug Manufacturing
Scalable Asymmetric Synthesis of 2(S)-Glutaric Acid Derivatives for Cardiovascular Drug Manufacturing
The pharmaceutical industry's relentless pursuit of safer and more effective cardiovascular therapies has placed a premium on high-purity chiral intermediates. Patent CN1028361C represents a significant technological leap in this domain, detailing a robust process for the preparation of substantially optically pure 2(S)-stereoisomers of glutaric acid derivatives. These compounds serve as critical precursors for a class of glutaramide derivatives known for their potent antihypertensive properties, as previously disclosed in European patent application 89308740.3. Unlike traditional methods that rely on the inefficient fractional crystallization of racemic mixtures, this invention leverages advanced asymmetric synthesis techniques to establish the required stereochemistry early in the synthetic sequence. By utilizing chiral auxiliaries and controlled lithiation strategies, the process avoids the generation of unwanted 2(R)-stereoisomers, thereby streamlining the production workflow. For R&D directors and process chemists, this patent offers a blueprint for achieving superior enantiomeric excess without the yield penalties associated with classical resolution techniques.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the manufacture of chiral glutaric acid derivatives relied heavily on the synthesis of racemic mixtures followed by resolution. This conventional approach is inherently flawed from both an economic and environmental perspective, as it theoretically caps the maximum yield of the desired active isomer at 50%. The remaining 50%, consisting of the inactive or less active 2(R)-stereoisomer, constitutes waste that must be separated and disposed of, often requiring multiple recrystallization steps that consume vast quantities of solvents and energy. Furthermore, fractional crystallization is notoriously difficult to control on a large industrial scale, where slight variations in cooling rates or solvent composition can lead to significant fluctuations in optical purity. For supply chain managers, this unpredictability translates into inconsistent batch quality and extended lead times, as additional purification cycles are frequently needed to meet the stringent specifications required for API intermediates. The reliance on racemic precursors also complicates the regulatory landscape, as impurities profiles become more complex with the presence of enantiomeric contaminants.
The Novel Approach
The methodology outlined in the patent data introduces a paradigm shift by employing asymmetric synthesis to construct the chiral center directly. This novel route utilizes (S,S)-α,α'-dimethyldibenzylamine as a chiral auxiliary, which is attached to an acrylate backbone to create a chiral electrophile. This strategic modification ensures that subsequent carbon-carbon bond-forming reactions occur with high facial selectivity. 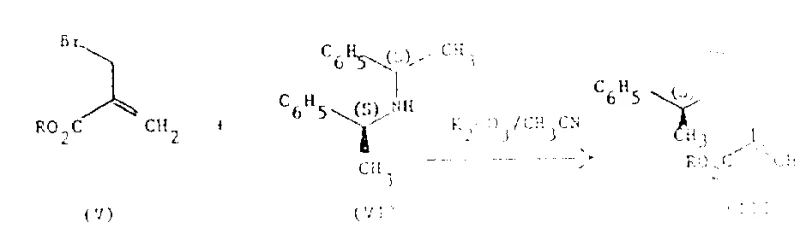 As illustrated in the preparation of the starting material, the chiral amine reacts with 2-bromoethyl acrylate under basic conditions to form the requisite chiral acrylate derivative. This pre-installation of chirality dictates the stereochemical outcome of the subsequent alkylation step, effectively bypassing the need for post-synthesis resolution. The result is a process that not only improves overall throughput by avoiding the 50% yield loss of racemic methods but also simplifies the purification protocol, as the crude product already possesses high optical purity. This approach aligns perfectly with the goals of cost reduction in pharmaceutical intermediate manufacturing by maximizing atom economy and minimizing waste generation.
As illustrated in the preparation of the starting material, the chiral amine reacts with 2-bromoethyl acrylate under basic conditions to form the requisite chiral acrylate derivative. This pre-installation of chirality dictates the stereochemical outcome of the subsequent alkylation step, effectively bypassing the need for post-synthesis resolution. The result is a process that not only improves overall throughput by avoiding the 50% yield loss of racemic methods but also simplifies the purification protocol, as the crude product already possesses high optical purity. This approach aligns perfectly with the goals of cost reduction in pharmaceutical intermediate manufacturing by maximizing atom economy and minimizing waste generation.
Mechanistic Insights into Lithium-Mediated Asymmetric Alkylation
The core of this synthetic strategy lies in the generation and reaction of a lithiated cyclopentanecarboxylate species. The mechanism involves the deprotonation of cyclopentanecarboxylic acid using a strong base, typically lithium diisopropylamide (LDA), to form a dianion intermediate. This step is critical and must be conducted under strictly controlled inert atmosphere and cryogenic conditions, typically ranging from -70°C to room temperature, to prevent side reactions and ensure the stability of the reactive species. The use of lithium cations is particularly advantageous as they coordinate with the carboxylate oxygen atoms, organizing the transition state and enhancing the nucleophilicity of the alpha-carbon. When this dianion reacts with the chiral acrylate electrophile, the bulky chiral auxiliary shields one face of the double bond, forcing the nucleophilic attack to occur from the less hindered side. This steric guidance is the fundamental driver of the observed 2(S)-stereoselectivity. 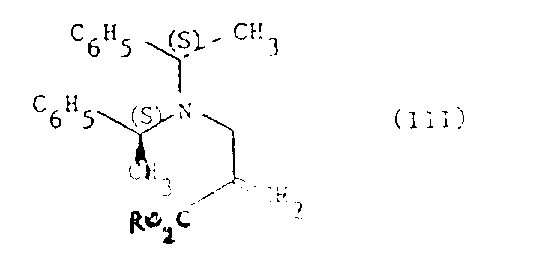 The reaction mixture is subsequently acidified at low temperatures, preferably below -10°C, to quench the reaction and protonate the carboxylate groups without compromising the newly formed stereocenter. This precise control over reaction parameters ensures that the optical purity of the product remains consistently high, usually containing less than 5% of the undesired 2(R)-stereoisomer.
The reaction mixture is subsequently acidified at low temperatures, preferably below -10°C, to quench the reaction and protonate the carboxylate groups without compromising the newly formed stereocenter. This precise control over reaction parameters ensures that the optical purity of the product remains consistently high, usually containing less than 5% of the undesired 2(R)-stereoisomer.
Impurity control in this process is managed through the inherent selectivity of the asymmetric step and the subsequent removal of the chiral auxiliary. Since the initial alkylation produces predominantly the desired isomer, the burden on downstream purification is significantly reduced. However, the patent notes that standard purification techniques such as extraction and silica gel chromatography can be employed to isolate the intermediate if necessary. The final step involves catalytic hydrogenation, typically using palladium hydroxide on carbon, to cleave the benzyl groups from the chiral auxiliary. This hydrogenolysis step is highly chemoselective, removing the protecting groups to reveal the primary amine while leaving the ester and carboxylic acid functionalities intact. The ability to perform this deprotection under mild conditions (room temperature, atmospheric pressure) further minimizes the risk of racemization or degradation, ensuring that the high optical purity established in the earlier steps is preserved in the final glutaric acid derivative. This mechanistic robustness makes the process highly attractive for the commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize 2(S)-Aminomethyl-3-(1-carboxycyclopentyl)propionic Acid Efficiently
The synthesis of these valuable intermediates follows a logical three-stage progression designed to maximize yield and stereochemical integrity. The process begins with the preparation of the chiral acrylate building block, followed by the critical asymmetric alkylation with a lithiated cyclopentane species, and concludes with the removal of the chiral auxiliary to yield the free amine. Each stage requires careful attention to temperature control and stoichiometry to ensure optimal results. The detailed standardized synthesis steps for implementing this route are provided in the guide below.
- Preparation of chiral acrylate by reacting 2-bromoethyl acrylate with (S,S)-alpha,alpha'-dimethyldibenzylamine in acetonitrile.
- Generation of cyclopentane carboxylate dianion using lithium diisopropylamide (LDA) at cryogenic temperatures (-70°C).
- Coupling the dianion with the chiral acrylate, followed by acidic workup and catalytic hydrogenation to remove the chiral auxiliary.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this asymmetric synthesis route offers tangible benefits that extend beyond simple chemical elegance. The primary advantage lies in the drastic simplification of the manufacturing workflow. By eliminating the need for racemic resolution, the process removes an entire unit operation that typically consumes significant resources in terms of solvents, filtration equipment, and time. This streamlining leads to a substantial reduction in manufacturing costs, as the theoretical yield of the active isomer is effectively doubled compared to traditional resolution methods. Furthermore, the reagents used in this process, such as cyclopentanecarboxylic acid and acrylates, are commercially available commodity chemicals, ensuring a stable and reliable supply chain. The avoidance of exotic or hard-to-source chiral catalysts reduces the risk of supply disruptions and price volatility, making the production of these intermediates more predictable and cost-effective.
- Cost Reduction in Manufacturing: The elimination of fractional crystallization steps significantly lowers solvent consumption and waste disposal costs. Additionally, the chiral auxiliary used in the process can potentially be recovered and recycled after the hydrogenation step, further driving down the cost of goods sold. The higher overall yield means that less raw material is required to produce the same amount of API intermediate, directly impacting the bottom line. This efficiency makes the process economically viable even for large-scale production runs where marginal savings per kilogram translate into significant total cost reductions.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials mitigates the risk of bottlenecks often associated with specialized chiral reagents. The robustness of the reaction conditions, which tolerate standard industrial equipment and do not require extreme pressures or temperatures beyond standard cryogenic capabilities, ensures that the process can be easily transferred between manufacturing sites. This flexibility allows for multi-sourcing strategies, reducing dependency on a single supplier and enhancing the resilience of the supply chain against unforeseen disruptions. The consistent quality of the output also reduces the likelihood of batch rejections, ensuring a steady flow of material to downstream API manufacturers.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing reaction conditions that are manageable in large-scale reactors. The reduction in solvent usage and waste generation aligns with increasingly stringent environmental regulations, reducing the burden of effluent treatment. The use of catalytic hydrogenation for deprotection is a clean technology that produces minimal byproducts compared to stoichiometric chemical reductions. This environmental friendliness not only lowers compliance costs but also enhances the sustainability profile of the final pharmaceutical product, a factor that is becoming increasingly important to stakeholders and regulators alike.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of these glutaric acid derivatives. The answers are derived directly from the technical specifications and experimental data provided in the patent literature, ensuring accuracy and relevance for industry professionals. Understanding these details is crucial for evaluating the feasibility of integrating this technology into existing production pipelines.
Q: What is the optical purity achieved in this asymmetric synthesis route?
A: The process described in patent CN1028361C consistently achieves an optical purity of approximately 95% or higher for the desired 2(S)-stereoisomer, significantly reducing the need for downstream resolution steps compared to racemic methods.
Q: Why is the 2(S)-configuration critical for antihypertensive agents?
A: Biological activity in glutaramide derivatives used for treating hypertension and heart failure is highly stereospecific. The 2(S)-configuration aligns with the receptor binding sites, whereas the 2(R)-isomer is often inactive or less potent, making enantiomeric purity essential for efficacy.
Q: Can the chiral auxiliary be recovered after the reaction?
A: Yes, the chiral auxiliary ((S,S)-alpha,alpha'-dimethyldibenzylamine) is removed via catalytic hydrogenation. This step not only reveals the primary amine required for the final API but also allows for the potential recovery and recycling of the amine auxiliary, enhancing process economics.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Glutaric Acid Derivatives Supplier
The technical potential of the asymmetric synthesis route described in patent CN1028361C is immense, offering a clear path to high-purity intermediates for the next generation of antihypertensive drugs. At NINGBO INNO PHARMCHEM, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this chemistry from the laboratory to the marketplace. Our facility is equipped with state-of-the-art cryogenic reactors and hydrogenation units capable of handling the specific requirements of this lithiation-based process. We maintain stringent purity specifications and operate rigorous QC labs to ensure that every batch of 2(S)-glutaric acid derivatives meets the exacting standards of the global pharmaceutical industry. Our team of expert process chemists is ready to optimize this route for your specific needs, ensuring maximum efficiency and yield.
We invite you to initiate a conversation about optimizing your supply chain for cardiovascular intermediates. Our technical procurement team is prepared to provide a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this asymmetric route. We encourage you to contact us to request specific COA data and route feasibility assessments tailored to your project timelines. By partnering with us, you gain access to a reliable source of high-quality intermediates that can accelerate your drug development programs and reduce your overall manufacturing costs.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
