Advanced Manufacturing of Symmetric Pyrazine Derivatives for Pharmaceutical Applications
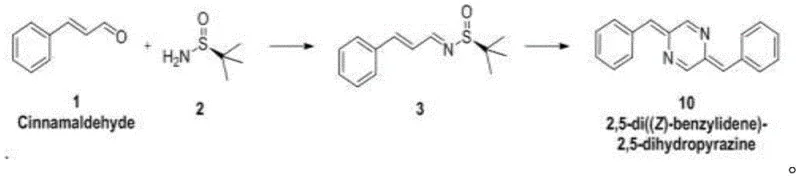
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for constructing heterocyclic scaffolds, particularly pyrazine derivatives which serve as critical backbones in numerous bioactive molecules including antibiotics and antihypertensives. A significant technological breakthrough in this domain is documented in Chinese Patent CN110128357B, which discloses a novel intermolecular [3+3] dimerization strategy for chiral imine molecules. This patent introduces a streamlined synthetic route that transforms readily available cinnamaldehyde and chiral tert-butylsulfinamide into high-value symmetric pyrazine derivatives, specifically 2,5-di((Z)-benzylidene)-2,5-dihydropyrazine. Unlike traditional methods that often rely on harsh conditions, this innovation leverages a mild base-mediated radical mechanism, offering a compelling alternative for manufacturers aiming to optimize their production of complex pharmaceutical intermediates. The strategic importance of this technology lies in its ability to bypass the limitations of conventional catalysis, providing a cleaner, more efficient pathway that aligns with modern green chemistry principles while maintaining high stereochemical integrity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of pyrazine compounds has been fraught with significant technical and operational challenges that hinder efficient commercial manufacturing. Traditional routes frequently necessitate the use of aggressive reagents such as strong acids or bases, which not only complicate downstream processing but also pose severe corrosion risks to standard reactor vessels, requiring expensive specialized equipment capable of withstanding extreme pH levels. Furthermore, many established protocols depend heavily on transition metal catalysts including nickel, copper, chromium, platinum, and cobalt to drive dehydration or dehydrogenation reactions. The reliance on these heavy metals introduces a critical bottleneck in the supply chain, as their removal to meet stringent pharmaceutical purity standards requires additional, costly purification steps such as scavenging or extensive chromatography. Additionally, conventional processes often operate under high temperature and high pressure conditions to overcome kinetic barriers, increasing energy consumption and safety risks associated with thermal runaway or pressurized vessel failure. These factors collectively inflate the cost of goods sold (COGS) and extend lead times, making traditional pyrazine synthesis less attractive for large-scale procurement strategies focused on cost reduction in API manufacturing.
The Novel Approach
In stark contrast to these legacy methods, the technology described in patent CN110128357B presents a paradigm shift by utilizing a chiral tert-butylsulfinamide-induced radical dimerization under remarkably mild conditions. This innovative approach replaces hazardous transition metals with cesium carbonate (Cs2CO3), a weak inorganic base that effectively promotes the reaction without the need for extreme thermal or pressure inputs. The process begins with the condensation of cinnamaldehyde and the chiral auxiliary to form a stable imine intermediate, which then undergoes a controlled radical dissociation upon heating. This methodology not only simplifies the reaction setup by eliminating the need for high-pressure autoclaves but also drastically reduces the complexity of the workup procedure, as there are no heavy metal residues to remove. The result is a highly selective transformation that yields symmetric dimeric structures with excellent efficiency, providing a new strategic avenue for the synthesis of symmetrical dimer molecules. By adopting this route, manufacturers can achieve significant operational simplifications and enhance the overall sustainability of their production lines.
Mechanistic Insights into Cs2CO3-Mediated Radical Dimerization
The core of this technological advancement lies in its unique mechanistic pathway, which diverges from standard ionic condensations to exploit radical chemistry for bond formation. The reaction initiates with the base-mediated homolytic cleavage of the sulfur-nitrogen bond in the chiral sulfinylimine intermediate, generating a reactive nitrogen-centered radical species. This radical intermediate is stabilized by the adjacent conjugated system, allowing for a subsequent 1,3-hydrogen migration that shifts the radical character to the alpha-position of the imine. This rearrangement is crucial as it generates an allylic-type radical that is primed for dimerization. The process avoids the formation of unstable carbocation intermediates often seen in acid-catalyzed routes, thereby minimizing side reactions such as polymerization or hydrolysis that typically plague pyrazine synthesis. The use of cesium carbonate is particularly effective due to its solubility profile in organic solvents like acetonitrile, ensuring homogeneous reaction conditions that facilitate consistent radical generation throughout the reaction mixture. This precise control over radical kinetics is what enables the high selectivity observed in the formation of the target pyrazine ring system.
Following the generation of the alpha-amino radical monomers, the mechanism proceeds through a concerted intermolecular [3+3] dimerization event. Two radical monomers couple to form a transient dimeric intermediate, which subsequently undergoes a 1,2-hydrogen migration to restore aromaticity and stabilize the pyrazine core. This final aromatization step is thermodynamically driven and irreversible, pushing the equilibrium towards the product and ensuring high conversion rates. The stereochemical outcome is dictated by the initial chiral induction from the tert-butylsulfinamide, although the final symmetric product results from the loss of the chiral auxiliary during the radical fragmentation. Understanding this mechanism is vital for R&D directors aiming to replicate or modify the process for analogous substrates, as it highlights the importance of maintaining anhydrous conditions and inert atmosphere to prevent radical quenching by oxygen or moisture. The robustness of this radical cascade ensures that impurity profiles remain clean, significantly reducing the burden on quality control laboratories during batch release testing.
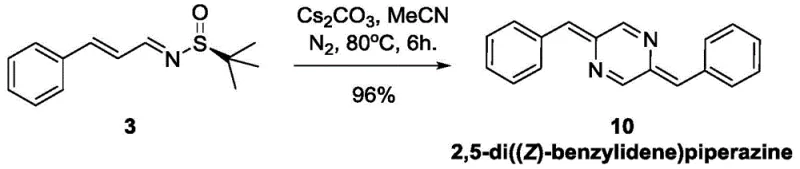
How to Synthesize 2,5-di((Z)-benzylidene)piperazine Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires strict adherence to the optimized parameters defined in the patent to ensure maximum yield and purity. The process is divided into two distinct stages: the formation of the chiral imine precursor and the subsequent base-mediated cyclization. Operators must ensure that all solvents, particularly tetrahydrofuran and acetonitrile, are thoroughly dried to prevent hydrolysis of the sensitive imine intermediates. The use of an inert gas blanket, such as nitrogen, is mandatory during the heating phases to protect the radical species from atmospheric oxygen, which could otherwise terminate the chain reaction and lead to oxidative byproducts. Temperature control is also critical, with the initial condensation requiring moderate heating around 40-60°C, while the dimerization step necessitates a slightly higher temperature of 80°C to activate the radical dissociation. Detailed standardized operating procedures for each step, including precise molar ratios and workup protocols, are essential for reproducible results.
- Condense cinnamaldehyde with chiral tert-butylsulfinamide using tetraethyl titanate in THF at 40-60°C to form the chiral imine intermediate.
- Treat the isolated chiral imine with cesium carbonate (Cs2CO3) in acetonitrile at 80°C under nitrogen protection.
- Monitor the radical dimerization via TLC, then perform aqueous workup and column chromatography to isolate the symmetric pyrazine derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented methodology offers tangible strategic benefits that extend beyond mere chemical efficiency. The primary advantage lies in the drastic simplification of the raw material portfolio; by eliminating the need for scarce and price-volatile transition metal catalysts, companies can insulate their supply chains from geopolitical disruptions affecting the mining and refining sectors. Furthermore, the substitution of strong acids and bases with mild cesium carbonate reduces the regulatory burden associated with the handling, storage, and disposal of hazardous corrosive materials. This shift not only lowers compliance costs but also enhances workplace safety, reducing the risk of accidents that could disrupt production schedules. The simplified downstream processing, characterized by the absence of heavy metal scavenging steps, translates directly into shorter cycle times and faster time-to-market for finished intermediates. These operational efficiencies collectively contribute to a more resilient and cost-effective supply network capable of responding agilely to market demands.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts such as palladium, platinum, or rhodium removes a significant variable cost component from the bill of materials. Moreover, the absence of these metals negates the need for specialized purification resins or activated carbon treatments typically required to meet residual metal specifications in pharmaceutical grades. This reduction in processing steps leads to substantial savings in both consumable materials and labor hours, directly improving the gross margin of the final product. Additionally, the mild reaction conditions reduce energy consumption compared to high-pressure hydrogenation or high-temperature dehydration processes, further lowering the utility overheads associated with production. The cumulative effect of these factors is a significantly reduced cost of goods sold, allowing for more competitive pricing strategies in the global marketplace.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals like cinnamaldehyde and cesium carbonate ensures a stable and diversified supply base, mitigating the risk of single-source bottlenecks. Unlike specialized chiral ligands or organometallic complexes which may have long lead times and limited suppliers, the reagents used in this process are widely available from multiple global vendors. This abundance allows procurement teams to negotiate better terms and maintain safety stocks without tying up excessive working capital. The robustness of the reaction also means that slight variations in raw material quality are less likely to cause batch failures, enhancing the overall reliability of supply. Consequently, manufacturers can promise more consistent delivery schedules to their downstream clients, strengthening long-term partnerships and contractual agreements.
- Scalability and Environmental Compliance: From a scale-up perspective, the mild nature of this chemistry allows for the use of standard glass-lined or stainless steel reactors without the need for exotic alloys resistant to strong corrosion. This compatibility with existing infrastructure accelerates the technology transfer from lab to plant, reducing capital expenditure requirements for new facilities. Environmentally, the process generates less hazardous waste, as the byproducts are primarily organic and easily treatable, unlike heavy metal sludge which requires specialized disposal protocols. This alignment with green chemistry principles facilitates easier permitting and regulatory approval in jurisdictions with strict environmental laws. The combination of easy scalability and reduced environmental footprint makes this technology an ideal candidate for sustainable manufacturing initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this dimerization technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a reliable basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this route into their existing portfolios. The answers reflect the practical realities of scaling radical chemistry and managing chiral auxiliaries in an industrial setting.
Q: What are the advantages of using Cs2CO3 over transition metal catalysts for pyrazine synthesis?
A: Using Cs2CO3 eliminates the need for expensive and toxic transition metals like nickel or copper, simplifying purification and reducing environmental hazards associated with heavy metal waste disposal.
Q: How does the chiral tert-butylsulfinamide function in this reaction pathway?
A: It acts as a chiral inducing group that facilitates the formation of the imine intermediate and subsequently undergoes radical dissociation under heating to drive the [3+3] dimerization mechanism.
Q: Is this synthesis method suitable for large-scale commercial production?
A: Yes, the mild reaction conditions (80°C, atmospheric pressure) and avoidance of specialized high-pressure equipment make this route highly adaptable for commercial scale-up from kilograms to metric tons.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,5-di((Z)-benzylidene)piperazine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced synthetic methodologies like the one described in CN110128357B for producing high-value pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of critical materials. Our state-of-the-art facilities are equipped to handle sensitive radical chemistries under strictly controlled inert atmospheres, guaranteeing the stringent purity specifications required for drug substance manufacturing. With our rigorous QC labs and commitment to process optimization, we are uniquely positioned to deliver this complex pyrazine derivative with the highest levels of quality and efficiency, supporting your drug development timelines from preclinical studies through to commercial launch.
We invite you to collaborate with us to leverage this innovative synthesis route for your specific project needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this metal-free approach can optimize your budget. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments. Let us help you secure a sustainable and cost-effective supply chain for your next-generation therapeutic candidates.
