Advanced Synthesis of Mirabegron Intermediates: Scaling Green Chemistry for Global Pharma Supply Chains
The pharmaceutical landscape for treating overactive bladder has been significantly transformed by the introduction of Mirabegron, a beta-3 adrenergic agonist. As global demand for this active pharmaceutical ingredient surges, the efficiency and safety of its supply chain have become paramount concerns for multinational drug developers. Patent CN110872234B introduces a groundbreaking synthesis method for (1R)-2-[[[2-(4-nitrophenyl)ethyl]amino]methyl]benzyl alcohol hydrochloride, a critical chiral intermediate in the Mirabegron production pathway. This innovation addresses long-standing bottlenecks in traditional manufacturing by replacing hazardous reduction steps with a mild, direct nucleophilic substitution. For R&D directors and procurement strategists, this patent represents a pivotal shift towards greener, more cost-effective chemistry that maintains rigorous stereochemical integrity while drastically simplifying the operational workflow.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art, specifically documented in patent EP1440969, outlines a synthetic route that has plagued manufacturers with safety hazards and economic inefficiencies for nearly two decades. The conventional process initiates with the condensation of D-mandelic acid and p-nitrophenylethylamine hydrochloride to form an amide intermediate, which subsequently requires reduction using Borane Dimethyl Sulfide. This reducing agent is notoriously dangerous; it is pyrophoric, toxic, and demands stringent safety protocols that inflate operational costs and complicate regulatory compliance. Furthermore, the reliance on solvents such as tetrahydrofuran, chloroform, or dichloromethane introduces significant environmental liabilities and waste disposal challenges. The multi-step nature of this legacy route inherently accumulates yield losses at each stage, resulting in a final product that is not only expensive to produce but also carries a higher risk of impurity profiles that are difficult to purge.
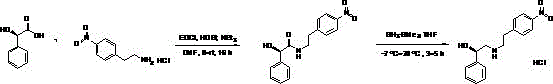
The Novel Approach
In stark contrast, the methodology disclosed in CN110872234B streamlines the synthesis into a highly efficient, single-step transformation that bypasses the need for dangerous reducing agents entirely. By utilizing 1R-2-chloro-1-phenylethyl alcohol and p-nitrophenethylamine as starting materials, the process leverages a direct substitution reaction in a methanol medium. This approach eliminates the formation of the amide precursor and the subsequent reduction step, collapsing the synthetic timeline and reducing the overall material footprint. The reaction conditions are remarkably mild, operating at temperatures between 60°C and 65°C, which minimizes thermal degradation and energy consumption. The result is a robust process that delivers high yields—exemplified by Example 1 achieving 98.4%—while utilizing methanol, a solvent known for its low toxicity and ease of recycling, thereby aligning perfectly with modern green chemistry principles.
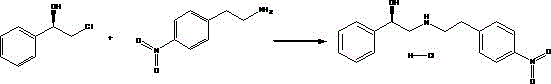
Mechanistic Insights into Nucleophilic Substitution and Stereochemical Retention
From a mechanistic perspective, the elegance of this new route lies in the chemoselectivity of the nucleophilic attack. The reaction involves the secondary amine nitrogen of p-nitrophenethylamine attacking the benzylic carbon of the 1R-2-chloro-1-phenylethyl alcohol. Crucially, the chiral center of the molecule resides at the C1 position, bearing the hydroxyl group, whereas the substitution occurs at the adjacent C2 methylene group where the chlorine leaving group is situated. Because the reaction center is distinct from the stereocenter, the (1R) configuration is inherently preserved throughout the transformation without the risk of racemization that often plagues reactions directly involving chiral carbons. This spatial separation ensures that the optical purity of the starting material is faithfully transferred to the final intermediate, a critical quality attribute for the biological activity of the downstream Mirabegron API.
Furthermore, the choice of methanol as the solvent plays a dual role in both kinetics and purification. Methanol effectively solvates the polar amine and the chlorohydrin, facilitating the collision frequency required for the substitution to proceed efficiently at moderate temperatures. The protocol specifies a precise solubility range for the p-nitrophenethylamine methanol solution (25-28%), which optimizes the reaction homogeneity and prevents localized concentration gradients that could lead to side reactions. Post-reaction, the addition of anti-solvents such as ethyl acetate or acetone induces rapid crystallization of the hydrochloride salt. This crystallization step acts as a powerful purification mechanism, excluding non-polar impurities and unreacted starting materials, thereby yielding a product with purity levels exceeding 99% as confirmed by HPLC analysis, ensuring it meets the stringent specifications required for pharmaceutical intermediates.
How to Synthesize (1R)-2-[[[2-(4-Nitrophenyl)Ethyl]Amino]Methyl]Benzyl Alcohol Hydrochloride Efficiently
The implementation of this synthesis protocol requires precise control over stoichiometry and thermal parameters to maximize yield and purity. The process begins with the preparation of a methanol solution of p-nitrophenethylamine, ensuring the concentration falls within the optimal 25-28% range to maintain solubility without excessive solvent volume. This solution is then combined with 1R-2-chloro-1-phenylethyl alcohol at room temperature before being subjected to controlled heating. The reaction mixture is maintained at a reflux temperature of 60-65°C for a duration of 8 to 12 hours, a window that allows for complete conversion while preventing thermal stress on the product. Following the reaction, the mixture is cooled, and a precipitation solvent is introduced to isolate the solid product. For a comprehensive, step-by-step technical guide including exact reagent grades and equipment specifications, please refer to the standardized protocol below.
- Mix 1R-2-chloro-1-phenylethyl alcohol and p-nitrophenethylamine in methanol at room temperature with a molar ratio of 1: 1 to 1:20.
- Heat the solution to 60-65°C and reflux with stirring for 8-12 hours to ensure complete conversion.
- Cool the reaction mixture, add a precipitation solvent like ethyl acetate or acetone, and filter to isolate the hydrochloride salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthesis route offers profound strategic advantages that extend far beyond simple chemical efficiency. The elimination of Borane Dimethyl Sulfide removes a major bottleneck associated with the sourcing, storage, and handling of hazardous materials, which often require specialized logistics and insurance premiums. By shifting to a process driven by stable, commodity-grade chemicals like methanol and simple amines, the supply chain becomes significantly more resilient and less susceptible to disruptions caused by regulatory crackdowns on dangerous goods. This stability translates directly into more predictable lead times and a reduced risk of production stoppages, ensuring a continuous flow of critical intermediates to downstream API manufacturing facilities.
- Cost Reduction in Manufacturing: The economic implications of removing the reduction step are substantial. Borane Dimethyl Sulfide is an expensive reagent that adds significant variable costs to every batch produced. By replacing this with a direct substitution using cheaper starting materials, the raw material cost base is drastically lowered. Additionally, the simplified workup procedure, which relies on filtration rather than complex extractions or distillations, reduces labor hours and utility consumption. The ability to use methanol, a low-cost solvent that is easily recovered and recycled, further diminishes the operational expenditure, creating a leaner cost structure that enhances competitiveness in the global generic drug market.
- Enhanced Supply Chain Reliability: The reliance on widely available starting materials mitigates the risk of supply shortages. Unlike specialized reducing agents that may have limited suppliers and long lead times, 1R-2-chloro-1-phenylethyl alcohol and p-nitrophenethylamine are accessible from multiple global sources. This diversification of the supply base empowers procurement teams to negotiate better terms and secure backup options, ensuring that production schedules remain uninterrupted. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, providing an additional layer of security against supply chain volatility.
- Scalability and Environmental Compliance: Scaling this process from pilot plant to commercial production is straightforward due to the absence of exothermic hazards associated with borane reductions. The mild thermal profile allows for the use of standard glass-lined or stainless steel reactors without the need for specialized cryogenic or high-pressure equipment. From an environmental standpoint, the process generates significantly less hazardous waste, simplifying effluent treatment and reducing the carbon footprint of the manufacturing site. This alignment with environmental, social, and governance (ESG) goals is increasingly becoming a prerequisite for partnerships with top-tier pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. These insights are derived directly from the experimental data and comparative analysis presented in patent CN110872234B, providing a clear understanding of the process capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for large-scale production.
Q: Why is the new synthesis method safer than the conventional Borane Dimethyl Sulfide route?
A: The conventional method relies on Borane Dimethyl Sulfide, a pyrophoric and toxic reducing agent that requires specialized handling and increases safety risks. The new patent CN110872234B utilizes a direct nucleophilic substitution in methanol, eliminating hazardous reagents entirely and significantly improving operational safety.
Q: How does this process impact the optical purity of the final intermediate?
A: The reaction targets the benzylic chlorine at the C2 position, while the chiral center containing the hydroxyl group is located at the C1 position. Since the reaction site is distinct from the stereocenter, the (1R) configuration is preserved throughout the substitution, ensuring high optical purity without the need for complex resolution steps.
Q: What are the scalability advantages of using methanol as the primary solvent?
A: Methanol is a widely available, low-cost, and environmentally friendly solvent with excellent solubility properties for the reactants. Its use simplifies the workup process, allows for easy recovery and recycling, and removes the need for exotic or difficult-to-dispose organic solvents, facilitating seamless commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (1R)-2-[[[2-(4-Nitrophenyl)Ethyl]Amino]Methyl]Benzyl Alcohol Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex pharmaceuticals like Mirabegron depends on a partner who can bridge the gap between innovative patent chemistry and industrial reality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of green chemistry are fully realized in your supply chain. We operate stringent purity specifications and maintain rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of intermediate meets the highest international standards for chirality and chemical purity.
We invite you to collaborate with us to optimize your Mirabegron supply chain through this superior synthesis technology. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data from our pilot runs and detailed route feasibility assessments to demonstrate how our manufacturing expertise can drive down your costs while securing your supply of this critical pharmaceutical intermediate.
