Advanced Butylene Liquid Crystal Compounds for High Performance Display Manufacturing
Advanced Butylene Liquid Crystal Compounds for High Performance Display Manufacturing
The rapid evolution of flat panel display technology, specifically MVA, IPS, and LCOS architectures, has created an urgent demand for negative liquid crystal materials that offer superior response times and environmental stability. Patent CN103539640A introduces a groundbreaking class of butylene type liquid crystal compounds designed to address these critical performance bottlenecks. These novel materials are characterized by their unique molecular architecture, which incorporates fluorine-substituted benzene rings and cyclohexyl linkages to achieve a low viscosity profile while maintaining a high absolute value of dielectric constant. This combination is essential for reducing the rotational viscosity of the liquid crystal mixture, thereby eliminating motion blur in high-speed video applications. Furthermore, the strong electron-withdrawing effect of the carbon-fluorine bonds imparts exceptional chemical inertness and resistance to UV degradation, ensuring long-term reliability in consumer electronics. As a leading provider of advanced electronic chemicals, we recognize the strategic importance of this intellectual property in next-generation display manufacturing.
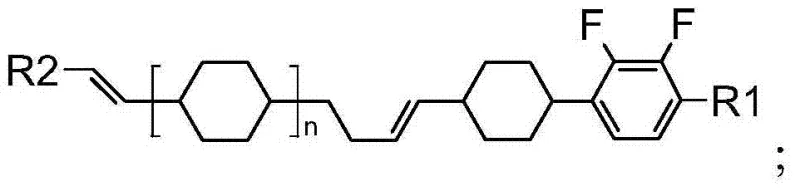
The structural versatility of these compounds allows for fine-tuning of physical properties by varying the R1 and R2 groups, which can be linear alkyl or alkenyl chains with varying degrees of fluorination. This modularity enables material scientists to optimize clearing points and melting ranges for specific display operating conditions. The patent details a robust synthetic pathway that avoids the use of scarce transition metal catalysts, relying instead on well-established organic transformations that are amenable to strict quality control protocols. By focusing on the precise control of stereochemistry, particularly the E/Z configuration of the olefinic bridges, the invention ensures that the final product possesses the optimal molecular shape for nematic phase alignment. This level of molecular precision is what distinguishes high-performance electronic materials from commodity chemicals, driving value for downstream panel manufacturers seeking competitive differentiation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional liquid crystal materials often suffer from high rotational viscosity, which directly correlates to slow response times and the dreaded streaking effect observed in moving images on LCD screens. Conventional synthesis routes for alkenyl-based liquid crystals frequently struggle with stereocontrol, resulting in mixtures of cis and trans isomers that degrade the overall electro-optical performance of the display mixture. Many prior art methods rely on harsh reaction conditions or expensive palladium-catalyzed cross-couplings that introduce trace metal impurities, which can adversely affect the voltage holding ratio (VHR) of the liquid crystal cell. Additionally, the thermal and photochemical stability of older generation compounds is often insufficient for the rigorous demands of large-screen, high-brightness displays used in outdoor or automotive applications. The inability to consistently produce high-purity trans-isomers without complex and costly purification steps has been a persistent barrier to achieving the desired balance of low viscosity and high dielectric anisotropy in negative liquid crystal formulations.
The Novel Approach
The methodology disclosed in CN103539640A overcomes these historical challenges through a strategically designed sequence of Wittig reactions and isomerization steps that prioritize stereochemical purity and process efficiency. By utilizing a specific phosphonium salt intermediate and controlling the reaction temperature within a narrow window of -40 to 0°C, the process effectively minimizes the formation of unwanted Z-isomers during the initial carbon-carbon bond formation. A key innovation lies in the dedicated isomerization step where the crude aldehyde intermediate is treated with a base, such as aqueous sodium hydroxide, at low temperatures to enrich the transoid conformation before the final coupling. This proactive approach to impurity management significantly reduces the burden on downstream purification, allowing for higher overall yields and lower production costs. The final acid-catalyzed double bond isomerization ensures that the terminal olefin adopts the thermodynamically stable E-configuration, which is critical for maintaining the linearity of the molecular core and optimizing the birefringence properties required for fast switching speeds.
Mechanistic Insights into Wittig Coupling and Isomerization
The core of this synthesis relies on the precise execution of the Wittig reaction, a powerful tool for constructing carbon-carbon double bonds with defined geometry. In the first stage, the reaction between the ketone precursor and the methoxymethyl triphenylphosphonium chloride generates an enol ether intermediate, which is subsequently hydrolyzed to reveal the aldehyde functionality. The choice of base, preferably potassium tert-butoxide in tetrahydrofuran, is critical for generating the reactive ylide species while minimizing side reactions such as aldol condensation. The subsequent isomerization of the resulting aldehyde mixture is a subtle yet vital step; under basic conditions, the equilibrium shifts towards the more stable trans-isomer due to reduced steric hindrance between the cyclohexyl ring and the aromatic system. This mechanistic understanding allows process chemists to manipulate reaction parameters, such as solvent polarity and temperature, to drive the equilibrium towards the desired product, achieving trans-isomer ratios exceeding 86% as demonstrated in the experimental examples.
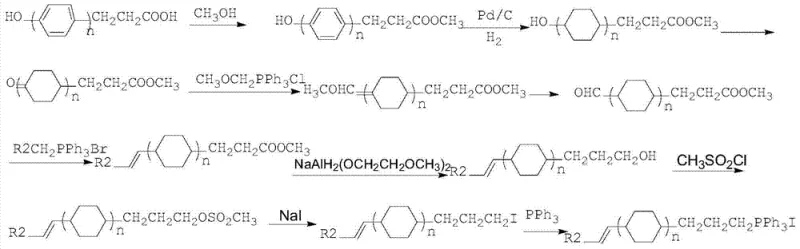
Furthermore, the synthesis of the phosphonium salt side chain, as illustrated in the provided reaction schemes, involves a multi-step sequence starting from readily available hydroxy-phenylpropionic acid derivatives. The oxidation of the cyclohexanol moiety to the ketone using sodium hypochlorite offers a greener alternative to chromium-based oxidants, aligning with modern environmental compliance standards. The reduction of the ester group to the corresponding alcohol using red aluminum (sodium bis(2-methoxyethoxy)aluminum hydride) provides excellent chemoselectivity, leaving the vinyl group intact for subsequent functionalization. The final quaternization with triphenylphosphine is conducted at elevated temperatures in ethylene glycol, a high-boiling solvent that facilitates the nucleophilic substitution without decomposing the sensitive vinyl functionality. This detailed mechanistic pathway highlights the robustness of the chemistry, ensuring that each intermediate can be isolated and characterized to meet stringent purity specifications before proceeding to the next step, thereby guaranteeing the quality of the final liquid crystal active ingredient.
How to Synthesize Butylene Liquid Crystal Efficiently
The synthesis of these high-value electronic materials requires a disciplined approach to process control, leveraging the specific reaction conditions outlined in the patent to maximize yield and purity. The following guide summarizes the critical operational parameters derived from the experimental embodiments, focusing on the preparation of the representative compound VC2VCW01. Operators must pay close attention to temperature gradients during the addition of reagents, particularly during the exothermic Wittig coupling, to prevent thermal runaway and the formation of byproducts. The use of anhydrous solvents and inert atmosphere techniques is mandatory to protect the reactive organometallic intermediates from moisture and oxygen degradation. For a comprehensive breakdown of the standardized operating procedures, including exact stoichiometric ratios and workup protocols, please refer to the detailed synthesis steps provided below.
- Perform Wittig reaction on starting compound I with CH3OCH2PPh3Cl using base catalysis to obtain Compound II.
- Hydrolyze Compound II under acid catalysis to generate Compound III (aldehyde intermediate).
- Conduct isomerization of Compound III under base catalysis to secure the transoid conformation of Compound IV.
- React Compound IV with phosphonium salt Compound V via Wittig reaction to form the alkene linkage in Compound VI.
- Finalize the synthesis by double bond isomerization of Compound VI under acid catalysis to yield the E-formula Compound VII.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the adoption of this synthetic route offers significant strategic advantages by simplifying the supply chain and reducing dependency on exotic reagents. The reliance on bulk commodity chemicals such as cyclohexanone derivatives, triphenylphosphine, and common mineral acids means that raw material sourcing is stable and less susceptible to geopolitical disruptions. The elimination of precious metal catalysts like palladium not only lowers the direct material cost but also removes the need for expensive metal scavenging steps, which are often required to meet the ultra-low conductivity specifications of liquid crystal displays. This streamlined process flow translates directly into improved manufacturing efficiency and a reduced carbon footprint, aligning with the sustainability goals of major electronics manufacturers. Furthermore, the high selectivity of the isomerization steps minimizes waste generation, as fewer byproducts need to be separated and disposed of, contributing to a more environmentally responsible production lifecycle.

- Cost Reduction in Manufacturing: The process achieves substantial cost savings by utilizing cost-effective oxidants like sodium hypochlorite instead of heavy metal reagents and by avoiding complex chromatographic purifications through high-selectivity isomerization. The ability to recrystallize the final product to achieve 99.9% GC purity using standard solvents like ethanol and toluene further reduces processing costs compared to methods requiring preparative HPLC. By optimizing the stoichiometry of the phosphonium salt and base, the reaction minimizes excess reagent consumption, leading to a more economical use of raw materials per kilogram of finished product.
- Enhanced Supply Chain Reliability: The synthetic pathway is built upon a foundation of widely available industrial intermediates, ensuring that production schedules are not held hostage by the lead times of specialized custom synthesis providers. The robustness of the chemistry allows for flexible manufacturing campaigns, where the same equipment train can be utilized for different analogues within the compound family by simply swapping the R1 or R2 side chain precursors. This flexibility enhances supply continuity, enabling manufacturers to respond rapidly to fluctuations in market demand for specific display grades without the need for extensive retooling or new vendor qualification processes.
- Scalability and Environmental Compliance: The reaction conditions are mild and operate at near-ambient pressures, making the technology easily scalable from pilot plant to multi-ton commercial production without significant engineering hurdles. The use of aqueous workups and recyclable organic solvents simplifies waste treatment and solvent recovery systems, ensuring compliance with increasingly stringent environmental regulations in key manufacturing regions. The high thermal stability of the intermediates reduces the risk of safety incidents during scale-up, providing a safer working environment and lowering insurance and liability costs associated with hazardous chemical processing.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and performance of this butylene liquid crystal technology. These answers are derived directly from the experimental data and technical specifications contained within the patent documentation, providing a reliable basis for feasibility assessments. Understanding these nuances is crucial for R&D teams evaluating the material for integration into new display formulations and for supply chain managers planning long-term procurement strategies.
Q: What are the key performance advantages of this butylene liquid crystal compound?
A: The compound exhibits low viscosity and a high absolute value of dielectric constant, which significantly improves response speed in LCD panels. It also demonstrates superior chemical, light, and thermal stability compared to conventional materials.
Q: How does the synthesis method ensure high stereochemical purity?
A: The process utilizes specific isomerization steps under controlled temperature conditions (e.g., -10 to 0°C) and acid/base catalysis to convert cis-isomers into the desired transoid conformation, achieving GC purity levels up to 99.9%.
Q: Is this manufacturing process suitable for large-scale industrial production?
A: Yes, the synthesis relies on standard organic reactions such as Wittig coupling and ester reduction using commercially available reagents like sodium borohydride or red aluminum, making it highly scalable for commercial supply chains.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Butylene Liquid Crystal Supplier
At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure necessary to bring complex electronic chemical pathways like this from the laboratory bench to full-scale commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. We understand that the performance of a liquid crystal display is only as good as the purity of its components; therefore, our stringent purity specifications and rigorous QC labs are designed to detect and eliminate trace impurities that could compromise device performance. Whether you require custom synthesis of specific R1/R2 analogues or large-volume supply of the standard VC2VCW01 grade, our facility is equipped to handle the unique challenges of fluorinated fine chemical manufacturing.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be adapted to your specific production requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how optimizing this supply chain can reduce your overall cost of goods sold. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments tailored to your project timeline, ensuring a seamless transition from development to mass production.
