Advancing Supramolecular Architecture: Template-Free Synthesis of Double-Layer Eight-Membered Rings
Introduction to Breakthrough Supramolecular Architecture
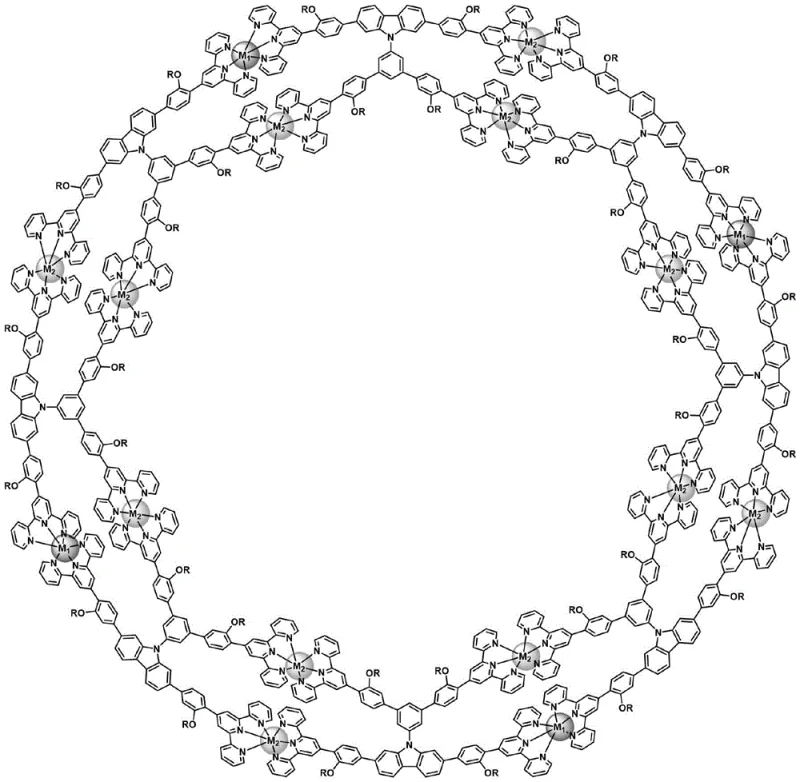
The field of supramolecular chemistry has long sought efficient methods to construct large, well-defined macrocyclic structures without relying on complex templating agents. Patent CN115181282B introduces a significant advancement in this domain by disclosing a double-layer eight-membered hollow ring metal-organic supramolecule and its novel synthesis method. This technology leverages coordination-driven self-assembly of dimeric K-type six-arm terpyridine metal-organic ligands with transition metal ions to create rigid, stable hollow ring structures. Unlike traditional approaches that depend on temporary bonding forces which often require harsh conditions to remove, this invention achieves precise structural control under mild reaction conditions. The resulting supramolecule features a unique double-layer eight-membered ring topology, offering immense potential for applications in host-guest chemistry, catalysis, and advanced functional materials where structural integrity and specific cavity sizes are paramount for performance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of large supramolecular macrocycles such as crown ethers, cyclodextrins, and pillararenes has heavily relied on template effects to guide the assembly process. These templates utilize temporary interactions like metal-ligand coordination, hydrogen bonding, or covalent bonding to connect substrates and promote ring closure. However, this dependency introduces significant inefficiencies in the manufacturing workflow. The removal of these templates often necessitates additional chemical steps, extensive purification protocols, and the use of specific reagents that can degrade the final product or leave behind impurities. Furthermore, the flexibility of organic ligands in conventional systems can lead to thermodynamic or kinetic instability, resulting in mixed product distributions rather than a single, well-defined supramolecular species. This lack of precision complicates the scale-up process and increases the overall cost of goods, making it difficult to achieve the high purity levels required for sensitive applications in pharmaceuticals or electronics.
The Novel Approach
The methodology described in the patent represents a paradigm shift by enabling the template-free self-assembly of metal-organic supramolecular macrocycles. By utilizing a specifically designed dimeric K-type six-arm terpyridine ligand, the system directs the formation of the eight-membered hollow ring structure purely through robust coordination bonds with transition metal ions. This approach eliminates the need for sacrificial templates, thereby streamlining the synthesis workflow and reducing the generation of chemical waste. The rigid nature of the terpyridine-metal complexes ensures that the resulting supramolecular architecture maintains its shape and stability without external support. Additionally, the modular design of the ligand allows for the incorporation of different metal ions, such as Ruthenium, Zinc, Iron, or Copper, providing a versatile platform for tuning the electronic and catalytic properties of the final material. This level of control over shape and size addresses a critical challenge in supramolecular chemistry, paving the way for more reliable and scalable production of complex functional materials.
Mechanistic Insights into Coordination-Driven Self-Assembly
The core of this technology lies in the precise engineering of the organic ligand and its subsequent interaction with metal centers. The synthesis begins with the construction of a dimeric K-type six-arm terpyridine metal-organic ligand, which serves as the fundamental building block for the supramolecular assembly. This ligand is synthesized through a series of sophisticated organic transformations, including cyclization reactions to form the terpyridine core, alkylation to introduce solubilizing ether or alkyl chains, and multiple Suzuki coupling reactions to link the aromatic units. The strategic placement of bromine and boronic ester functional groups allows for the stepwise assembly of the large molecular framework with high regioselectivity. Once the ligand is fully constructed, it acts as a multidentate chelator that coordinates with transition metal ions. The geometry of the terpyridine units dictates the angle and orientation of the metal-ligand bonds, effectively programming the system to close into an eight-membered ring rather than forming linear polymers or other oligomeric by-products.
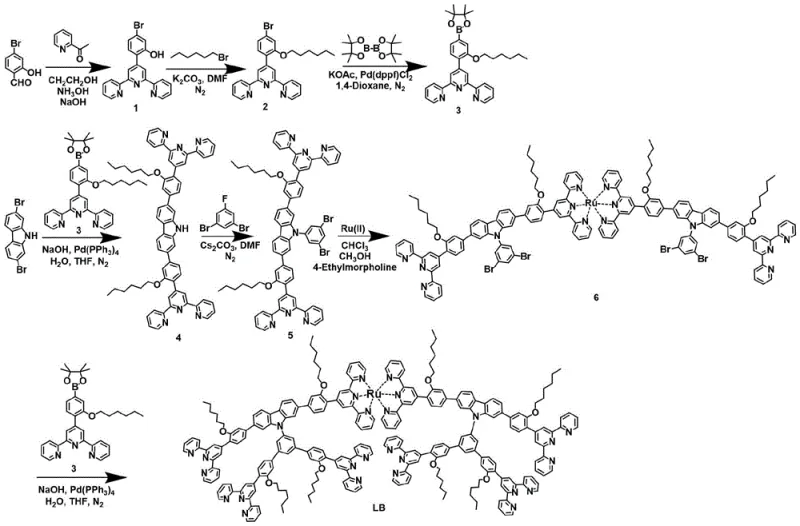
Impurity control is inherently built into the molecular design through the rigidity of the coordination complexes. The use of Ruthenium (RuII) ions at specific nodes (M1) and Zinc (ZnII), Iron (FeII), or Copper (CuII) ions at others (M2) creates a thermodynamically stable network that resists rearrangement. During the self-assembly process, the reaction conditions are carefully optimized, typically involving heating at 60-70°C in a mixed solvent system of methanol, chloroform, and acetonitrile. This specific solvent ratio is crucial for maintaining the solubility of the growing oligomers while promoting the correct intermolecular interactions. Following the assembly, an anion exchange step using agents like lithium bistrifluoromethanesulfonimide or ammonium hexafluorophosphate is employed. This step replaces weaker coordinating anions introduced during the metal salt addition with bulky, non-coordinating anions, which facilitates the precipitation of the supramolecular product from the solution. This purification strategy ensures that the final isolated material is free from unreacted ligands or incorrect oligomeric species, delivering a product with high structural fidelity.
How to Synthesize Double-Layer Eight-Membered Hollow Ring Supramolecules Efficiently
The synthesis of these complex architectures requires a disciplined approach to organic synthesis and coordination chemistry. The process is divided into two main stages: the preparation of the specialized ligand and the subsequent self-assembly with metal ions. The ligand synthesis involves multiple steps including the formation of terpyridine derivatives, protection and deprotection strategies, and palladium-catalyzed cross-coupling reactions. Each step must be monitored closely to ensure high conversion and minimize side reactions that could hinder the final assembly. The detailed standardized synthesis steps for producing the dimeric K-type six-arm terpyridine metal-organic ligand and its subsequent conversion into the final supramolecular structure are outlined below.
- Preparation of the dimeric K-type six-arm terpyridine metal-organic ligand (Formula I) through multi-step organic synthesis including cyclization, alkylation, and Suzuki coupling.
- Dissolution of the ligand in a mixed solvent system of methanol, chloroform, and acetonitrile followed by the addition of transition metal salt solutions.
- Heating the reaction mixture at 60-70°C for 6-12 hours to facilitate self-assembly, followed by anion exchange and filtration to isolate the final supramolecular product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this template-free synthesis route offers substantial strategic benefits regarding cost efficiency and operational reliability. Traditional methods for creating large macrocycles often involve expensive templating agents and complex purification sequences that drive up the cost of goods sold. By eliminating the template requirement, this new process removes an entire category of raw material costs and reduces the consumption of solvents and reagents associated with template removal. Furthermore, the use of robust Suzuki coupling reactions and standard coordination chemistry means that the synthesis can be performed using widely available catalysts and reagents, reducing dependency on exotic or supply-constrained chemicals. This simplification of the bill of materials directly translates to improved margin potential and greater resilience against supply chain disruptions.
- Cost Reduction in Manufacturing: The elimination of template agents significantly lowers raw material expenses and simplifies the downstream processing workflow. Without the need for template removal, the number of unit operations such as extraction, washing, and chromatography is drastically reduced. This reduction in processing steps not only saves on labor and energy costs but also minimizes solvent waste disposal fees. The high selectivity of the coordination-driven assembly ensures that the yield of the desired product is maximized relative to by-products, further enhancing the overall economic efficiency of the manufacturing process.
- Enhanced Supply Chain Reliability: The synthetic route relies on commodity chemicals and established reaction types like Suzuki coupling and nucleophilic substitution. This ensures that the raw materials required for production are readily available from multiple global suppliers, mitigating the risk of single-source bottlenecks. The robustness of the reaction conditions, which operate at moderate temperatures and pressures, allows for flexible manufacturing scheduling and reduces the likelihood of batch failures due to sensitive process parameters. This reliability is critical for maintaining consistent supply to downstream customers in industries where continuity is essential.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing reaction conditions that are easily transferable from laboratory to pilot and commercial scales. The absence of harsh template removal steps reduces the generation of hazardous waste streams, aligning with increasingly stringent environmental regulations. The ability to precipitate the final product through simple anion exchange and filtration offers a green chemistry advantage by minimizing the volume of organic solvents required for isolation. This environmental profile not only reduces compliance costs but also enhances the sustainability credentials of the final product, which is a growing priority for end-users in the pharmaceutical and electronic sectors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of these double-layer eight-membered hollow ring supramolecules. The answers are derived directly from the technical specifications and experimental data provided in the patent documentation to ensure accuracy and relevance for potential partners.
Q: What is the primary advantage of this template-free synthesis method?
A: The primary advantage is the elimination of template removal steps, which simplifies purification and reduces waste. The rigid structure formed by coordination bonds ensures stability without needing temporary bonding forces like hydrogen bonds that require harsh conditions to break.
Q: Which metal ions are compatible with this supramolecular assembly?
A: The system is versatile and supports various transition metal ions. Specifically, M1 positions are occupied by Ruthenium (RuII) ions, while M2 positions can accommodate Zinc (ZnII), Iron (FeII), or Copper (CuII) ions, allowing for tunable electronic and catalytic properties.
Q: How does the alkyl chain length affect the solubility of the ligand?
A: The introduction of ether or alkyl chains (R groups) at specific positions on the terpyridine ligand significantly enhances solubility in organic solvents like chloroform and methanol. This modification is critical for facilitating the homogeneous mixing required for effective self-assembly.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Double-Layer Eight-Membered Hollow Ring Metal-Organic Supramolecule Supplier
The development of template-free supramolecular synthesis represents a significant leap forward in materials science, offering a pathway to highly structured and functional macrocycles. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate this innovative patent technology into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex synthetic routes like the multi-step ligand assembly and coordination process can be executed with precision and consistency. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to verify the structural integrity and composition of every batch, guaranteeing that our clients receive materials that meet the highest standards of quality.
We invite you to collaborate with us to optimize your supply chain for advanced functional materials. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and application needs. By leveraging our process optimization capabilities, we can help you reduce lead times and secure a stable supply of high-purity supramolecular intermediates. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our manufacturing excellence can support your next generation of products.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
