Advanced Manufacturing of Methyl 3-Methyl-2-Amino-5-Chlorobenzoate for Diamide Insecticide Production
The global demand for high-efficiency diamide insecticides, such as chlorantraniliprole and cyantraniliprole, has placed immense pressure on the supply chains of their critical precursors. Patent CN115806497A introduces a transformative methodology for the preparation of anthranilate compounds, specifically targeting the synthesis of methyl 3-methyl-2-amino-5-chlorobenzoate, a pivotal building block in modern agrochemistry. This intellectual property addresses long-standing inefficiencies in traditional synthetic routes by integrating a streamlined sequence that eliminates cumbersome isolation steps and mitigates significant safety hazards associated with chlorination reactions. For R&D directors and process chemists, this patent represents a shift towards greener, more robust manufacturing protocols that prioritize both operational safety and economic viability without compromising on chemical purity or yield.
Furthermore, the strategic implementation of recyclable catalytic systems within this framework underscores a commitment to sustainable chemical manufacturing. By leveraging a platinum-based catalyst capable of extensive reuse, the process drastically reduces the environmental footprint typically associated with noble metal waste in pharmaceutical and agrochemical synthesis. This innovation not only aligns with increasingly stringent global environmental regulations but also offers a compelling value proposition for procurement managers seeking to stabilize raw material costs against volatile market fluctuations. The technical depth of this disclosure provides a clear roadmap for scaling production from laboratory benchtops to multi-ton commercial facilities, ensuring a reliable supply of high-quality intermediates for the next generation of crop protection agents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of anthranilate intermediates has been plagued by significant operational challenges that hinder efficient commercial scale-up. Prior art methods, such as those disclosed in WO2006062978, often rely on the use of acetonitrile as a solvent for chlorination reactions involving sulfuryl chloride. Acetonitrile is inherently hygroscopic, meaning it readily absorbs moisture from the environment, which poses a severe risk when reacting with sulfuryl chloride. The interaction between water and sulfuryl chloride generates gaseous by-products, including sulfur oxides, which can lead to unpredictable and violent gas evolution during the reaction. On a large industrial scale, this phenomenon manifests as sudden surges or runaway reactions, creating dangerous conditions where reactants can be ejected from the reactor, posing threats to personnel and equipment integrity.
In addition to safety concerns, conventional processes frequently employ expensive chlorinating agents such as sodium hypochlorite solutions or N-chlorosuccinimide, which are economically unfeasible for high-volume production. These reagents not only drive up the direct material costs but also complicate the downstream purification processes due to the formation of diverse impurity profiles. Furthermore, traditional routes often necessitate the isolation of intermediate compounds through crystallization or precipitation, introducing additional unit operations that increase processing time, solvent consumption, and overall yield loss. The cumulative effect of these limitations results in a manufacturing process that is cumbersome, inefficient, and fraught with safety risks, making it unsuitable for the rigorous demands of modern agrochemical supply chains.
The Novel Approach
The methodology outlined in patent CN115806497A presents a sophisticated solution to these entrenched problems by re-engineering the solvent system and reaction sequence. A cornerstone of this novel approach is the substitution of hygroscopic solvents with chlorine-containing organic solvents, such as dichloroethane, which do not absorb moisture to the same extent. This strategic solvent selection allows for precise control over the water content in the reaction mixture, enabling operators to reduce moisture levels to less than 0.5% w/w prior to the introduction of sulfuryl chloride. By maintaining such low moisture levels, the formation of hazardous gaseous by-products is effectively suppressed, transforming a potentially dangerous exothermic process into a safe, controllable, and scalable operation suitable for continuous or batch manufacturing.
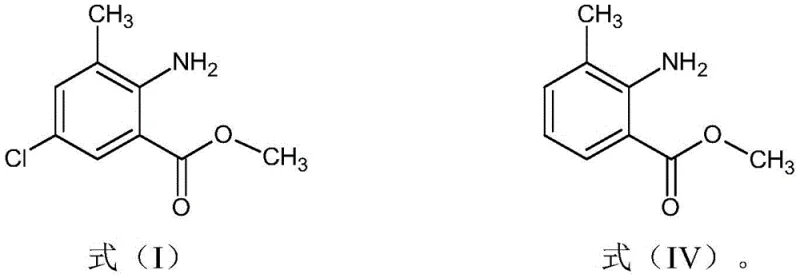
Moreover, this innovative process eliminates the need to isolate the intermediate methyl 3-methyl-2-aminobenzoate (Formula IV) before proceeding to the chlorination step. Instead, the organic layer containing the intermediate is directly treated with sulfuryl chloride after moisture adjustment. This telescoping of steps significantly reduces the number of unit operations, minimizes solvent usage, and prevents yield losses associated with solid handling techniques like filtration and drying. The use of cost-effective reagents like sulfuryl chloride, combined with the ability to recycle the platinum catalyst used in the preceding reduction step more than 20 times, creates a highly economical pathway. This holistic optimization ensures that the production of anthranilate compounds is not only safer and environmentally friendlier but also substantially more cost-competitive for large-scale industrial applications.
Mechanistic Insights into Catalytic Reduction and Electrophilic Chlorination
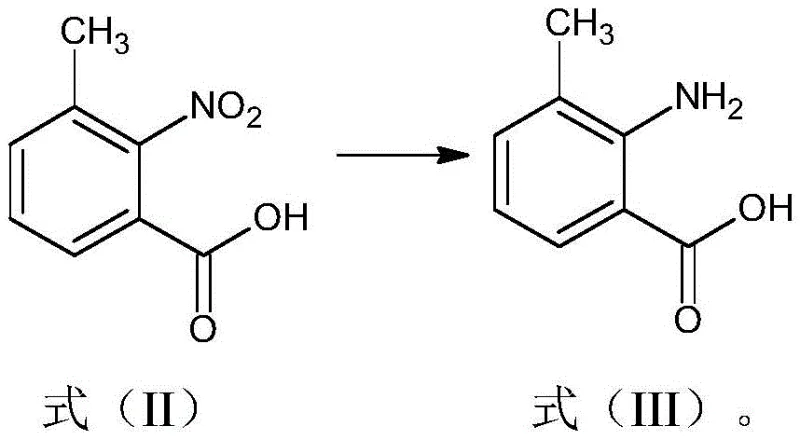
The success of this synthetic route relies heavily on the efficiency of the initial reduction step, where 2-nitro-3-methylbenzoic acid (Formula II) is converted into 2-amino-3-methylbenzoic acid (Formula III). This transformation is achieved through catalytic hydrogenation using a platinum catalyst, preferably platinum on carbon (Pt/C), under hydrogen pressure. The choice of platinum over other noble metals like palladium is critical, as the patent data indicates that the platinum catalyst maintains its activity and structural integrity over multiple cycles, allowing for reuse more than 20 times without significant degradation. This recyclability is a key mechanistic advantage, as it prevents the leaching of metal contaminants into the product stream and reduces the total catalyst loading required per kilogram of final product, directly impacting the cost of goods sold.
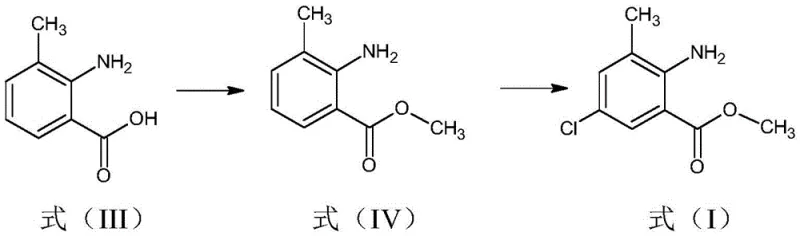
Following the reduction, the methylation of the carboxylic acid group to form the ester (Formula IV) sets the stage for the final chlorination. The subsequent electrophilic aromatic substitution involves the reaction of the amino-ester with sulfuryl chloride. Mechanistically, the strict control of moisture is paramount here; water acts as a nucleophile that competes with the aromatic ring for the electrophilic chlorine species, leading to hydrolysis of the sulfuryl chloride and the generation of acidic gases. By ensuring the organic layer is dried to below 0.5% moisture via azeotropic distillation, the reaction kinetics are directed exclusively towards the desired chlorination of the aromatic ring at the 5-position. This precision prevents the formation of side products and ensures a high-purity profile for the final anthranilate compound, which is essential for downstream coupling reactions in diamide insecticide synthesis.
How to Synthesize Methyl 3-Methyl-2-Amino-5-Chlorobenzoate Efficiently
Implementing this improved process requires careful attention to solvent selection and moisture management to replicate the safety and efficiency benefits described in the patent. The synthesis begins with the hydrogenation of the nitro precursor, followed by a one-pot methylation and extraction sequence that avoids isolating the sensitive amino-ester intermediate. Operators must utilize chlorine-containing solvents for the extraction phase to facilitate effective moisture removal via azeotropic distillation before introducing the chlorinating agent. This specific sequence of operations is designed to maximize throughput while minimizing waste generation, making it an ideal protocol for facilities aiming to enhance their cost reduction in agrochemical intermediate manufacturing capabilities.
- Reduce 2-nitro-3-methylbenzoic acid (Formula II) using a recyclable platinum catalyst under hydrogen pressure to obtain 2-amino-3-methylbenzoic acid (Formula III).
- Methylate Formula III with dimethyl sulfate in the presence of a base and solvent to generate methyl 3-methyl-2-aminobenzoate (Formula IV).
- Extract Formula IV with a chlorine-containing solvent, reduce moisture to <0.5%, and react directly with sulfuryl chloride to yield the final anthranilate (Formula I).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented process offers tangible strategic advantages that extend beyond simple chemical yield improvements. The primary value driver lies in the significant simplification of the manufacturing workflow, which directly translates to reduced operational expenditures and enhanced supply reliability. By eliminating the isolation of intermediate compounds, the process removes entire stages of production such as filtration, drying, and re-dissolution, which are often bottlenecks in batch processing. This streamlining not only accelerates the overall production cycle time but also reduces the consumption of utilities and auxiliary materials, resulting in substantial cost savings that can be passed down through the supply chain to benefit end-users in the agrochemical sector.
- Cost Reduction in Manufacturing: The economic viability of this process is heavily bolstered by the use of inexpensive and readily available reagents such as sulfuryl chloride, replacing costly alternatives like N-chlorosuccinimide found in older methods. Additionally, the ability to recycle the platinum catalyst used in the reduction step more than 20 times dramatically lowers the amortized cost of noble metals, which are typically a major expense in fine chemical synthesis. The elimination of intermediate isolation further reduces labor costs and equipment occupancy time, allowing facilities to produce higher volumes with the same infrastructure. These factors combine to create a highly competitive cost structure that insulates buyers from raw material price volatility.
- Enhanced Supply Chain Reliability: Safety is a critical component of supply chain continuity, and this process significantly mitigates the risk of production shutdowns due to safety incidents. By avoiding the use of hygroscopic solvents that lead to runaway gas evolution, the process ensures stable and predictable reaction profiles even at large scales. This stability reduces the likelihood of unplanned maintenance or regulatory interventions caused by safety breaches. Furthermore, the robustness of the solvent system and the recyclability of the catalyst mean that the supply of key inputs is less susceptible to disruptions, ensuring a consistent flow of high-purity agrochemical intermediates to downstream manufacturers.
- Scalability and Environmental Compliance: The design of this synthetic route is inherently scalable, having been developed with industrial feasibility as a core objective. The reduction in effluent generation, achieved by telescoping steps and minimizing solvent swaps, aligns perfectly with modern environmental, health, and safety (EHS) standards. Facilities adopting this method can expect lower waste disposal costs and a reduced environmental footprint, which is increasingly important for maintaining social license to operate in the chemical industry. The process avoids extreme temperature conditions and complex post-treatment procedures, making it easier to transfer from pilot plants to full-scale commercial production units without extensive re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this improved anthranilate synthesis process. These insights are derived directly from the experimental data and embodiments disclosed in the patent documentation, providing clarity on how this technology can be integrated into existing manufacturing frameworks. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this route for their specific production needs.
Q: How does this process improve safety compared to prior art chlorination methods?
A: Unlike prior art using acetonitrile which is hygroscopic and leads to unpredictable gas surges with sulfuryl chloride, this method uses non-hygroscopic chlorine-containing solvents and strictly controls moisture below 0.5%, preventing runaway reactions.
Q: What represents the primary cost-saving driver in this synthetic route?
A: The primary cost drivers are the elimination of intermediate isolation steps for Formula IV and the ability to recycle the platinum reduction catalyst more than 20 times, significantly lowering catalyst consumption costs.
Q: Is this process suitable for large-scale commercial production of diamide insecticides?
A: Yes, the process is designed for industrial feasibility by avoiding extreme temperature conditions, utilizing cost-effective reagents like sulfuryl chloride, and simplifying post-treatment procedures to enhance throughput.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Methyl 3-Methyl-2-Amino-5-Chlorobenzoate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful formulation of next-generation diamide insecticides. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of methyl 3-methyl-2-amino-5-chlorobenzoate performs consistently in your downstream synthesis. Our capability to implement advanced catalytic technologies, such as the recyclable platinum systems described in recent patents, allows us to offer superior value without compromising on quality or safety standards.
We invite global partners to collaborate with us to optimize their supply chains and achieve significant competitive advantages in the agrochemical market. By leveraging our expertise in process development and manufacturing, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring that your project moves forward with the most efficient and reliable sourcing strategy available in the industry.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
