Revolutionizing Halogenated Acid Production: A Green, Scalable Route to 8-Bromooctanoic Acid
The landscape of fine chemical manufacturing is undergoing a significant transformation, driven by the urgent need for greener, more efficient synthetic pathways that do not compromise on yield or purity. A pivotal advancement in this domain is detailed in patent CN114230451A, which discloses a novel preparation method for halogenated acid compounds, specifically focusing on the synthesis of 8-Bromooctanoic acid. This compound serves as a critical building block in the development of various pharmaceutical agents and agrochemical formulations, necessitating a supply chain that is both robust and environmentally sustainable. The innovation lies in the strategic replacement of hazardous oxidants and harsh pH controllers with a catalytic TEMPO system regulated by carbon dioxide, marking a departure from legacy methods that have long plagued the industry with toxicity and waste management challenges.
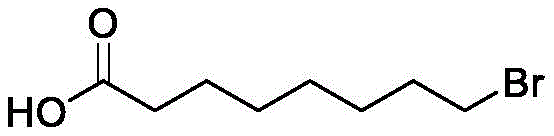
For R&D directors and process chemists evaluating potential partners, the structural integrity and purity of intermediates like 8-Bromooctanoic acid are paramount. The molecule features a long aliphatic chain terminated by a reactive carboxylic acid group and a bromine atom, a combination that requires precise handling to avoid unwanted side reactions such as elimination or nucleophilic substitution during synthesis. The method outlined in the patent addresses these sensitivities directly, offering a pathway that preserves the halogen functionality while efficiently oxidizing the primary alcohol. This level of chemical precision is essential for downstream applications where impurity profiles can dictate the success of final drug substance registration and regulatory approval.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
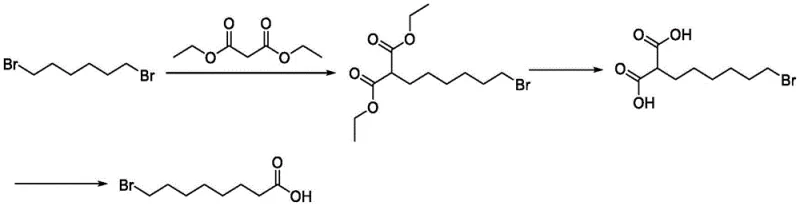
Historically, the synthesis of 8-Bromooctanoic acid has been fraught with inefficiencies and environmental hazards that hinder large-scale commercial viability. One conventional approach, referenced in patent CN113087623, relies on the substitution reaction of 1,6-dibromohexane with diethyl malonate, followed by hydrolysis and decarboxylation. While chemically feasible, this route suffers from inherently low conversion rates in the initial substitution step, necessitating a large excess of the dibromo raw material to drive the reaction forward. Furthermore, the subsequent decarboxylation requires high temperatures, which not only consumes significant energy but also promotes the formation of numerous by-products, complicating the purification process and reducing overall yield.
Another widely cited method involves the oxidation of 1,8-octanediol derivatives using Jones reagent (chromium trioxide in sulfuric acid). Although this route offers a shorter synthetic sequence, it introduces severe environmental and safety liabilities. The use of hexavalent chromium generates substantial amounts of toxic heavy metal waste, imposing rigorous and costly disposal requirements on manufacturers. Additionally, the harsh acidic conditions can be detrimental to sensitive functional groups, and the removal of residual chromium to meet pharmaceutical standards is a technically demanding and expensive endeavor. These factors collectively render traditional methods suboptimal for modern, sustainability-focused supply chains.
The Novel Approach
In stark contrast, the methodology presented in CN114230451A leverages a biomimetic oxidation strategy that circumvents the pitfalls of its predecessors. By utilizing a catalytic amount of TEMPO (2,2,6,6-tetramethylpiperidine-1-oxyl) in conjunction with sodium hypochlorite and sodium chlorite, the process achieves high-efficiency oxidation under mild conditions. The true breakthrough, however, is the use of carbon dioxide gas to regulate the reaction pH. Unlike hydrochloric acid, which can induce chlorination side reactions or elimination of the bromine atom, or phosphate buffers which create significant wastewater treatment burdens, CO2 provides a gentle, self-regulating acidic environment. This ensures that the intermediate aldehyde is smoothly converted to the carboxylic acid without compromising the integrity of the bromo-alkyl chain, resulting in a cleaner reaction profile and superior product quality.
Mechanistic Insights into TEMPO-Catalyzed Oxidation with CO2 Regulation
The core of this technological advancement rests on the synergistic interaction between the nitroxyl radical catalyst and the stoichiometric oxidants within a carefully controlled pH window. In this mechanism, TEMPO acts as the active oxidant, cycling between its oxoammonium cation form and the hydroxylamine species. Sodium hypochlorite serves to regenerate the active oxoammonium species from the reduced hydroxylamine, while sodium chlorite acts as the terminal oxidant to convert the intermediate aldehyde into the final carboxylic acid. This dual-oxidant system is highly effective but notoriously sensitive to pH; the oxidation of the aldehyde to the acid by chlorite proceeds optimally only in a slightly acidic environment, typically between pH 5 and 7.

Maintaining this specific pH range is critical for minimizing impurities. If the environment becomes too acidic (as with HCl), the risk of nucleophilic attack on the alkyl bromide increases, leading to chlorinated impurities or alkene formation via elimination. Conversely, if the pH is too high, the oxidation rate drops, and the reaction may stall at the aldehyde stage. The introduction of carbon dioxide gas allows for dynamic, real-time adjustment of the acidity. As CO2 dissolves in the aqueous phase, it forms carbonic acid, buffering the system precisely within the required 6-7 range. This elegant solution eliminates the need for strong mineral acids or bulky buffer salts, thereby simplifying the workup procedure and significantly reducing the inorganic salt load in the effluent, which is a major advantage for industrial waste management.
How to Synthesize 8-Bromooctanoic Acid Efficiently
Implementing this synthesis requires careful attention to reagent addition rates and temperature control to maximize the benefits of the catalytic cycle. The process begins with the dissolution of the starting material, 8-bromooctanol, in a mixed solvent system of acetonitrile and water, which provides the necessary solubility for both organic substrates and inorganic oxidants. The reaction is initiated at moderate temperatures, typically below 35°C, to prevent thermal degradation of the reagents and ensure selectivity. The sequential addition of sodium hypochlorite and sodium chlorite, coupled with the continuous sparging of CO2, drives the conversion to completion within a few hours. For a detailed, step-by-step operational guide including specific molar ratios and quenching protocols, please refer to the standardized synthesis instructions below.
- Prepare the reaction mixture by dissolving 8-bromooctanol in an acetonitrile-water solvent system with catalytic TEMPO.
- Maintain the reaction pH between 5 and 7 by continuously sparging carbon dioxide gas into the system while adding oxidants.
- Quench the reaction with sodium sulfite, extract the product, and purify via crystallization or vacuum distillation to achieve >99% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers tangible strategic benefits beyond mere chemical elegance. The primary advantage lies in the drastic simplification of the supply chain for raw materials. The reagents employed—TEMPO, sodium hypochlorite (bleach), sodium chlorite, and carbon dioxide—are commodity chemicals available globally in bulk quantities. This contrasts sharply with specialized oxidants or exotic catalysts that may suffer from supply bottlenecks or volatile pricing. By relying on widely accessible inputs, manufacturers can secure a more stable and predictable supply of 8-Bromooctanoic acid, mitigating the risk of production delays caused by raw material shortages.
- Cost Reduction in Manufacturing: The economic implications of this process are profound, primarily driven by the elimination of costly purification steps associated with traditional methods. By avoiding the use of chromium-based oxidants, the expense of heavy metal scavenging and hazardous waste disposal is completely removed from the cost structure. Furthermore, the high selectivity of the TEMPO/CO2 system minimizes the formation of difficult-to-remove by-products, leading to higher crude yields and reducing the loss of valuable material during recrystallization or distillation. The use of cheap CO2 for pH control also negates the need for expensive buffer salts and the subsequent wastewater treatment costs associated with high-salt effluents, contributing to substantial overall cost savings in pharmaceutical intermediate manufacturing.
- Enhanced Supply Chain Reliability: The robustness of this chemistry translates directly into improved delivery performance. Because the reaction conditions are mild and the reagents are stable, the process is less prone to batch failures or deviations that can disrupt production schedules. The ability to run the reaction at near-ambient temperatures (20-35°C) reduces the energy load on manufacturing facilities and lowers the risk of thermal runaway incidents, ensuring consistent uptime. This reliability allows suppliers to offer shorter lead times for high-purity intermediates, enabling downstream pharmaceutical clients to maintain leaner inventory levels and respond more agilely to market demands.
- Scalability and Environmental Compliance: From a regulatory and sustainability perspective, this method positions the supply chain for long-term viability. The absence of heavy metals and the reduction in saline wastewater align perfectly with increasingly stringent global environmental regulations, such as REACH in Europe and various EPA guidelines in the US. This compliance reduces the administrative burden on suppliers and minimizes the risk of shutdowns due to environmental violations. Moreover, the process is inherently scalable; the use of gas-liquid mass transfer (CO2 sparging) is a unit operation that scales linearly from pilot plants to multi-ton reactors, facilitating the commercial scale-up of complex intermediates without the need for significant process re-engineering.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this new preparation method is essential for stakeholders evaluating its adoption. The following questions address common inquiries regarding the safety, efficiency, and purity profiles of the CO2-regulated TEMPO oxidation process. These answers are derived directly from the experimental data and technical specifications provided in the patent literature, ensuring that decision-makers have accurate information for their risk assessments and vendor qualification processes.
Q: Why is CO2 used instead of HCl for pH control in this oxidation?
A: Using hydrochloric acid can lead to the formation of chloride impurities or elimination by-products due to the acidic environment reacting with the halogenated chain. CO2 provides a mild, buffered acidic environment (pH 6-7) that prevents these side reactions while ensuring efficient oxidation.
Q: How does this method compare to traditional Jones oxidation?
A: Traditional Jones oxidation utilizes toxic chromium reagents which generate hazardous heavy metal waste. The TEMPO/NaClO/NaClO2 method described in CN114230451A eliminates heavy metals entirely, offering a significantly safer and more environmentally compliant process for industrial scale-up.
Q: What purity levels can be achieved with this synthetic route?
A: The patented process demonstrates the capability to achieve purity levels exceeding 99.7% through standard workup procedures such as crystallization or vacuum distillation, making it suitable for high-specification pharmaceutical applications without extensive additional purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 8-Bromooctanoic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the adoption of advanced synthetic methodologies is key to maintaining a competitive edge in the global pharmaceutical market. Our team of expert process chemists has thoroughly analyzed the pathway described in CN114230451A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are equipped with state-of-the-art reactors capable of handling gas-liquid reactions and stringent purity specifications, supported by rigorous QC labs that ensure every batch meets the highest international standards. Our commitment to green chemistry aligns with the industry's shift towards sustainable manufacturing, allowing us to deliver high-quality intermediates with a minimized environmental footprint.
We invite you to collaborate with us to leverage this innovative technology for your specific project needs. Whether you require custom synthesis services or bulk supply of 8-Bromooctanoic acid, our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our optimized supply chain can enhance the efficiency and profitability of your drug development pipeline.
