Advanced Phase Transfer Catalysis for High-Purity Bis-isobutyronitrile Hydrazine Manufacturing
The chemical industry is constantly evolving, driven by the need for more efficient, sustainable, and high-yielding synthetic routes for critical intermediates. A significant breakthrough in this domain is documented in patent CN101445471B, which details a novel method for synthesizing bis-isobutyronitrile hydrazine, a pivotal precursor in the production of Diisopropyl azodicarboxylate (DIAD). This patent introduces a sophisticated application of phase transfer catalysis that fundamentally alters the reaction kinetics and thermodynamics of the condensation between acetone cyanohydrin and hydrazine hydrate. By leveraging quaternary ammonium compounds, the process achieves a remarkable synthesis yield ranging from 98.23% to 98.60%, coupled with a product purity exceeding 98%. For R&D directors and process engineers, this represents a shift from traditional, water-intensive batch processes to a streamlined, high-efficiency protocol that minimizes waste and maximizes reactor throughput. The implications for large-scale manufacturing are profound, offering a pathway to reduce both operational expenditures and environmental footprint while ensuring the stringent quality standards required by the pharmaceutical and fine chemical sectors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of bis-isobutyronitrile hydrazine has been plagued by inefficiencies inherent in classical condensation protocols. Traditional methods often rely on the mechanical recycling of mother liquor water to improve yield, a practice that introduces significant variability and quality control challenges. As noted in prior art such as CN1785963, reusing mother liquor requires precise pH adjustments and leads to the gradual accumulation of foreign matter and impurities. Over repeated cycles, this accumulation causes the product color to degrade, shifting from desirable white crystals to an unacceptable yellow hue, which can compromise the quality of downstream derivatives like DIAD. Furthermore, conventional techniques necessitate the addition of substantial volumes of water—typically 7 to 10 times the molar amount of acetone cyanohydrin—to act as a dispersant. This excessive water usage drastically enlarges the effective volume of the reaction unit, forcing manufacturers to utilize larger reactors to maintain the same output, thereby inflating capital expenditure and energy costs associated with heating and subsequent solvent removal.
The Novel Approach
The methodology outlined in patent CN101445471B offers a transformative solution to these entrenched problems by integrating phase transfer catalysts (PTC) into the reaction matrix. Instead of relying on massive volumes of water for dispersion, this approach utilizes quaternary ammonium salts to facilitate the interaction between the organic acetone cyanohydrin and the aqueous hydrazine hydrate phases. This catalytic intervention allows for a dramatic reduction in water consumption, lowering the water-to-reactant ratio to a mere 1:1.5 to 1:5. The result is a highly concentrated reaction environment that not only accelerates the reaction rate but also significantly enhances the separation efficiency during the cooling crystallization phase. By eliminating the need for mother liquor recycling, the process ensures the production of stable, high-quality white crystals with consistent purity profiles. This shift from a dilution-dependent mechanism to a catalysis-driven interface optimization represents a major leap forward in process intensification, enabling manufacturers to achieve higher throughput within existing infrastructure.
Mechanistic Insights into Quaternary Ammonium-Catalyzed Condensation
The core innovation of this synthesis lies in the mechanistic role of the phase transfer catalyst, typically a quaternary ammonium salt with the general formula R4N+X-. In the biphasic system formed by acetone cyanohydrin and hydrazine hydrate, the reactants reside in different phases, creating a kinetic barrier to reaction. The phase transfer catalyst acts as a molecular shuttle, transporting the nucleophilic hydrazine species into the organic phase or facilitating the encounter at the interphase boundary. This dramatically increases the frequency of effective collisions between reactant molecules, thereby propelling the reaction towards the formation of bis-isobutyronitrile hydrazine with exceptional speed and efficiency. The catalyst also serves a dual function as a dispersant, stabilizing the emulsion and ensuring uniform heat distribution throughout the reaction vessel, which is critical for preventing localized hot spots that could lead to side reactions or decomposition.
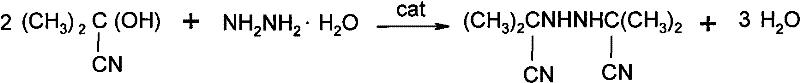
Furthermore, the mechanistic pathway inherently supports superior impurity control. In traditional water-heavy systems, the solubility of the product can lead to losses in the mother liquor, necessitating recycling strategies that introduce contaminants. In the PTC-mediated process, the reduced water content and the specific interaction of the catalyst promote the precipitation of the product upon cooling to 5°C. This crystallization behavior is highly selective, excluding many polar impurities that might otherwise co-precipitate in a less optimized system. The result is a crude product with purity greater than 98% directly after filtration and drying, minimizing the need for extensive recrystallization or purification steps. This mechanistic elegance translates directly to commercial value, as it simplifies the downstream processing train and reduces the generation of hazardous waste streams associated with purification solvents.
How to Synthesize Bis-isobutyronitrile Hydrazine Efficiently
Implementing this advanced synthesis route requires precise control over reaction parameters to fully realize the benefits of phase transfer catalysis. The process begins with the charging of acetone cyanohydrin, a selected quaternary ammonium catalyst (such as tetrabutylammonium bromide or tetramethyl ammonium chloride), and a minimized quantity of dilution water into a stirred reactor at ambient temperature. Hydrazine hydrate is then added dropwise to manage the exotherm, followed by heating the mixture to a controlled range of 55°C to 85°C. Maintaining this temperature for 2.5 to 5 hours ensures complete conversion, after which the mixture is cooled to induce crystallization. The detailed standardized operating procedures, including specific molar ratios and safety protocols for handling hydrazine derivatives, are critical for safe and reproducible manufacturing.
- Combine acetone cyanohydrin, a quaternary ammonium phase transfer catalyst, and a controlled amount of dilution water in a reactor under stirring at ambient temperature.
- Add hydrazine hydrate dropwise and heat the mixture to a temperature range of 55°C to 85°C, maintaining this condition for 2.5 to 5 hours to ensure complete condensation.
- Cool the reaction mixture to approximately 5°C to induce crystallization, then filter the solid product and dry under reduced pressure to obtain high-purity white crystals.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of the technology described in CN101445471B offers compelling economic and logistical advantages. The primary driver of value is the substantial reduction in production costs derived from process intensification. By eliminating the need for excessive water volumes, the process significantly reduces the energy load required for heating large masses of solvent and subsequently removing it during the drying phase. This efficiency gain translates directly into lower utility costs per kilogram of product. Additionally, the high yield of over 98% means that raw material utilization is maximized, reducing the cost of goods sold (COGS) by minimizing waste of expensive precursors like acetone cyanohydrin. The elimination of complex mother liquor recycling loops further simplifies the operational workflow, reducing labor costs and the potential for human error in pH adjustment and filtration steps.
- Cost Reduction in Manufacturing: The implementation of phase transfer catalysis fundamentally alters the cost structure of bis-isobutyronitrile hydrazine production. By drastically reducing the volume of water required, the process allows for a higher concentration of reactants, which means that existing reactor vessels can produce significantly more product per batch without capital expansion. This intensification leads to a decrease in the fixed cost allocation per unit. Moreover, the high selectivity and yield minimize the generation of by-products and waste, thereby lowering the costs associated with waste treatment and disposal. The qualitative improvement in process efficiency ensures that the manufacturing line operates with maximum asset utilization, providing a robust buffer against fluctuating raw material prices.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by complex processes that are prone to failure or quality deviations. The novel synthesis method enhances reliability by producing a consistent, high-purity product that meets stringent specifications without the need for rework. The stability of the white crystalline product ensures longer shelf life and easier handling during logistics and storage. Furthermore, the simplified process flow, which avoids the complexities of mother liquor management, reduces the risk of batch failures due to impurity buildup. This reliability allows suppliers to commit to tighter delivery schedules and maintain steady inventory levels, crucial for downstream pharmaceutical manufacturers who depend on just-in-time delivery of critical intermediates.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new challenges, but this PTC-mediated route is inherently designed for industrial viability. The reduction in solvent volume means that scaling up does not require a proportional increase in reactor size, making the transition from pilot plant to commercial production smoother and less capital intensive. From an environmental perspective, the process aligns with green chemistry principles by reducing water consumption and waste generation. The lower volume of aqueous waste simplifies effluent treatment, helping manufacturers comply with increasingly strict environmental regulations. This sustainability profile not only mitigates regulatory risk but also enhances the brand value of the supply chain partners by supporting eco-friendly manufacturing practices.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis method is essential for stakeholders evaluating its adoption. The following questions address common inquiries regarding the catalyst selection, reaction safety, and product specifications based on the patent data. These insights are derived directly from the experimental embodiments and comparative examples provided in the intellectual property documentation, ensuring that the information is grounded in verified scientific results.
Q: What are the primary advantages of using a phase transfer catalyst in this synthesis?
A: The use of a quaternary ammonium phase transfer catalyst significantly accelerates the reaction speed by improving the interface contact between the organic and aqueous phases. This innovation allows for a drastic reduction in the amount of dilution water required, thereby increasing throughput and reducing energy consumption for solvent removal, while boosting yields to over 98%.
Q: How does this method improve product quality compared to traditional recycling methods?
A: Traditional methods that mechanically recycle mother liquor often lead to the accumulation of impurities, resulting in yellowish products that compromise downstream quality. The novel phase transfer catalytic method produces stable, white crystalline bis-isobutyronitrile hydrazine with purity exceeding 98%, ensuring consistent quality for sensitive applications like DIAD production.
Q: What represent the optimal reaction conditions for industrial scale-up?
A: For optimal industrial results, the reaction should be conducted at temperatures between 55°C and 85°C for a duration of 2.5 to 5 hours. The molar ratio of acetone cyanohydrin to hydrazine hydrate is maintained around 1:0.5 to 0.6, utilizing minimal water (1:1.5 to 5 ratio) to maximize reactor efficiency and minimize waste treatment loads.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bis-isobutyronitrile Hydrazine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your final pharmaceutical products. Our technical team has extensively analyzed the advancements presented in patent CN101445471B and possesses the expertise to implement this phase transfer catalytic technology at scale. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of high yield and purity are realized in every batch we deliver. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that the bis-isobutyronitrile hydrazine supplied meets the exacting standards required for the synthesis of DIAD and other sensitive fine chemicals.
We invite you to collaborate with us to optimize your supply chain for this critical intermediate. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how our advanced manufacturing capabilities can reduce your overall procurement costs. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us partner with you to secure a reliable, cost-effective, and high-quality supply of bis-isobutyronitrile hydrazine for your global operations.
