Scalable Manufacturing of Key Androgen Receptor Antagonist Intermediate via Optimized Suzuki Coupling
Scalable Manufacturing of Key Androgen Receptor Antagonist Intermediate via Optimized Suzuki Coupling
The pharmaceutical industry's relentless pursuit of potent oncology therapeutics has placed significant focus on Androgen Receptor (AR) antagonists, particularly for the treatment of prostate cancer. A pivotal molecule in this therapeutic class is N-((S)-1-(3-(3-chloro-4-cyanophenyl)-1H-pyrazole-1-yl)-propyl-2-yl)-5-(1-hydroxyethyl)-1H-pyrazole-3-carboxamide, designated as compound (1A). The commercial viability of such complex small molecules often hinges on the efficiency of synthesizing their key building blocks. Patent CN107428695B discloses a transformative process for preparing the critical intermediate 2-chloro-4-(1H-pyrazol-3-yl)benzonitrile, known as compound (V). This technical breakthrough addresses long-standing bottlenecks in cross-coupling chemistry, offering a pathway that is not only chemically robust but also economically superior for large-scale production. By re-engineering the solvent system and catalyst loading, this innovation provides a reliable pharmaceutical intermediate supplier with the tools to deliver high-purity materials while drastically simplifying the manufacturing workflow.
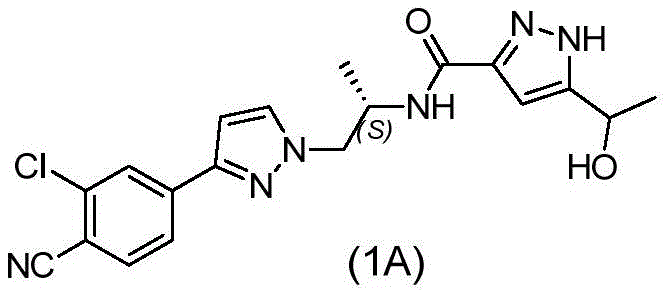
The structural complexity of compound (1A) necessitates a highly convergent synthetic strategy where the integrity of the pyrazole-benzonitrile core is paramount. The intermediate (V) serves as the foundational scaffold upon which the rest of the pharmacophore is built. Traditional methods for constructing this biaryl linkage often suffer from poor atom economy and cumbersome purification steps that erode overall yield. The methodology outlined in CN107428695B represents a paradigm shift, moving away from high-loading precious metal catalysis towards a more sustainable, low-loading regime. For R&D directors evaluating process feasibility, this patent offers a compelling case study in how subtle changes in reaction conditions—specifically the switch to an acetonitrile-water biphasic system—can yield disproportionate benefits in downstream processing and cost reduction in API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art processes, such as those disclosed in WO2011/051540 and WO2012/143599, rely heavily on traditional Suzuki-Miyaura coupling conditions that are increasingly viewed as inefficient for modern commercial scale-up. These legacy methods typically employ a THF-water or THF-toluene-water solvent mixture in conjunction with bis(triphenylphosphine)palladium(II) chloride. A critical drawback of these established routes is the requirement for high catalyst loading, often up to 5 mol-%, to drive the reaction to completion. This excessive use of palladium not only inflates raw material costs but also creates significant challenges in meeting stringent residual metal specifications in the final active pharmaceutical ingredient. Furthermore, the workup procedures associated with these older methods are operationally intensive, frequently requiring distillation of solvents to near dryness to isolate the protected intermediate (III).
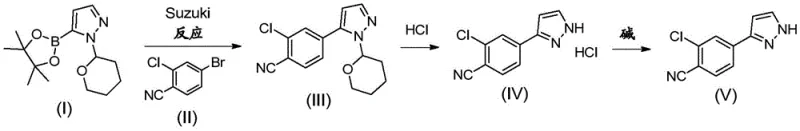
The reliance on ethanolic HCl for the subsequent deprotection step further complicates the manufacturing process. Handling large volumes of ethanolic HCl on an industrial scale introduces safety hazards and logistical burdens related to solvent recovery and waste disposal. The cumulative effect of multiple distillation steps, phase transfers, and crystallizations in the conventional route results in a fragmented process flow that is prone to yield losses. For supply chain heads, these inefficiencies translate into longer lead times and higher variability in production batches. The need to distill solvents to dryness is particularly energy-intensive and poses a risk of thermal degradation for sensitive intermediates. Consequently, there has been a persistent demand for a more practical and economical process that eliminates these unit operations while maintaining high chemical purity.
The Novel Approach
The inventive process described in CN107428695B fundamentally reimagines the synthesis of compound (V) by optimizing both the catalytic system and the solvent environment. Instead of the traditional THF-based systems, the new method utilizes an acetonitrile-water solvent mixture. This seemingly simple substitution unlocks a powerful advantage: upon completion of the Suzuki reaction, the reaction mixture spontaneously separates into two distinct liquid phases. This phase separation allows for the direct removal of the aqueous layer containing inorganic salts and base residues without the need for energy-intensive distillation. The organic acetonitrile phase, containing the desired product, can then be treated directly to induce crystallization. This streamlined isolation technique significantly reduces the processing time and energy consumption associated with the manufacture of this key pharmaceutical intermediate.
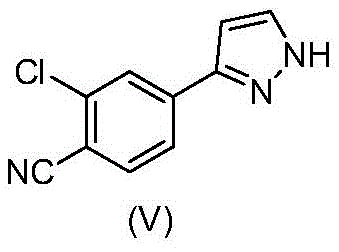
Moreover, the novel approach achieves high conversion rates with a dramatically reduced amount of palladium catalyst. By employing palladium(II) acetate [Pd(OAc)2] in combination with triphenylphosphine, the reaction proceeds efficiently with catalyst loadings as low as 0.5 to 2 mol-%, with a preferred range of 0.6 to 0.8 mol-%. This represents a substantial reduction compared to the 5 mol-% required in prior art, leading to direct cost savings and easier purification. The deprotection step is similarly optimized; the process replaces ethanolic HCl with a catalytic amount of 30% aqueous HCl in methanol. This modification not only simplifies reagent handling but also facilitates a one-pot conversion from the protected intermediate (III) to the final product (V) without isolating the hydrochloride salt (IV). The result is a cohesive, continuous process that enhances overall yield and operational safety, making it ideally suited for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Pd(OAc)2-Catalyzed Suzuki Coupling
The success of the optimized process lies in the nuanced interplay between the catalyst precursor and the solvent system. In the conventional route, the use of pre-formed Pd(PPh3)2Cl2 in THF often requires higher loadings to overcome deactivation pathways or solubility issues. In contrast, the in situ generation of the active catalytic species from Pd(OAc)2 and triphenylphosphine in an acetonitrile-water medium appears to create a more robust catalytic cycle. The polar nature of acetonitrile facilitates the dissolution of the inorganic base, typically potassium carbonate, ensuring efficient transmetallation. Simultaneously, the presence of water aids in the hydrolysis of the boronic ester pinacol group, generating the reactive boronate species necessary for the coupling. The specific molar ratio of Pd(OAc)2 to triphenylphosphine, maintained at approximately 1:3, ensures that the palladium center remains sufficiently ligated to prevent precipitation of palladium black while remaining active for oxidative addition into the aryl bromide bond of compound (II).
Impurity control is another critical aspect where the new mechanism excels. High levels of residual palladium are a common regulatory hurdle in API synthesis. By reducing the initial catalyst loading to less than 1 mol-%, the burden on downstream scavenging technologies is significantly alleviated. Furthermore, the phase separation step acts as an inherent purification stage. Inorganic impurities and polar byproducts preferentially partition into the aqueous phase, which is discarded prior to product isolation. The subsequent crystallization of compound (III) from the acetonitrile phase by the addition of water further purifies the material, excluding non-polar organic impurities. This multi-stage purification inherent in the reaction design ensures that the final intermediate (V) meets stringent purity specifications, often exceeding 99% HPLC purity as demonstrated in the patent examples, thereby reducing the risk of carryover impurities into the final drug substance.
How to Synthesize 2-chloro-4-(1H-pyrazol-3-yl)benzonitrile Efficiently
The execution of this improved synthesis requires precise control over temperature and addition rates to maximize yield and minimize side reactions. The process begins with the preparation of the reaction mixture under an inert nitrogen atmosphere to prevent oxidation of the phosphine ligand and the palladium catalyst. The careful balance of acetonitrile and water, typically in a ratio ranging from 40:60 to 60:40 by volume, is crucial for achieving the desired phase separation post-reaction. The reaction is heated to a moderate temperature of 70 ± 3 °C, which is sufficient to drive the coupling without promoting decomposition of the sensitive pyrazole ring. Following the coupling, the deprotection step is conducted at controlled low temperatures (0-15 °C) to manage the exotherm associated with acid addition, ensuring the structural integrity of the nitrile group. Detailed standardized synthetic steps for replicating this high-efficiency route are provided in the guide below.
- Perform Suzuki coupling of boronic ester (I) and bromo-nitrile (II) using Pd(OAc)2/PPh3 in acetonitrile-water at 70°C.
- Separate the acetonitrile phase, cool, and add water to precipitate the protected intermediate (III).
- Treat intermediate (III) with catalytic aqueous HCl in methanol, neutralize, and precipitate the final product (V).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of the process described in CN107428695B offers tangible economic and operational benefits that extend beyond simple yield improvements. The primary driver of value is the drastic reduction in the consumption of precious metal catalysts. By lowering the palladium loading from 5 mol-% to below 1 mol-%, the direct material cost for the catalytic system is significantly reduced. This reduction also mitigates the volatility risk associated with fluctuating palladium prices, providing greater cost stability for long-term supply contracts. Additionally, the elimination of distillation-to-dryness steps translates into substantial energy savings and reduced cycle times. In a large-scale manufacturing setting, avoiding multiple solvent swaps and distillations can shorten the batch production time considerably, thereby increasing the throughput of existing manufacturing assets without the need for capital investment in new equipment.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the minimization of high-value inputs and the simplification of unit operations. The shift to a low-loading palladium catalyst system directly lowers the bill of materials for every kilogram of intermediate produced. Furthermore, the replacement of ethanolic HCl with aqueous HCl eliminates the need for specialized solvent handling and recovery systems associated with ethanol, reducing utility costs. The ability to isolate products via simple filtration after water addition, rather than complex extraction and evaporation sequences, reduces labor hours and solvent waste disposal costs. These cumulative efficiencies result in a leaner cost structure that allows for more competitive pricing in the global market for oncology intermediates.
- Enhanced Supply Chain Reliability: Operational simplicity is a key determinant of supply chain resilience. The new process reduces the number of discrete processing steps, which inherently lowers the probability of batch failures or deviations. The robustness of the acetonitrile-water system against minor variations in water content or temperature makes the process more forgiving and easier to control across different manufacturing sites. This consistency ensures a steady flow of high-quality intermediate (V), reducing the risk of stockouts that could delay the production of the final API. Moreover, the use of common, commodity solvents like acetonitrile and methanol ensures that raw material availability is not a bottleneck, unlike specialized solvent mixtures that might face supply constraints.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, the new route aligns well with green chemistry principles. The reduction in solvent usage and the elimination of energy-intensive distillation steps lower the carbon footprint of the manufacturing process. The use of aqueous acid instead of ethanolic HCl reduces the generation of hazardous organic waste streams. The process is designed to be scalable from pilot plant to multi-ton commercial production without fundamental changes to the chemistry, ensuring that technology transfer is smooth and predictable. This scalability supports the growing demand for AR antagonists as the patient population expands, ensuring that supply can keep pace with clinical and commercial needs without compromising on environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and process descriptions within CN107428695B, providing clarity on how this technology can be integrated into existing manufacturing frameworks. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this improved method for the production of androgen receptor antagonist intermediates.
Q: How does the new process reduce palladium catalyst consumption?
A: The patented method utilizes Pd(OAc)2 with triphenylphosphine in an acetonitrile-water system, allowing effective catalysis at loadings as low as 0.5 to 2 mol-%, compared to 5 mol-% in prior art.
Q: What are the advantages of the acetonitrile-water solvent system?
A: This solvent system forms two distinct liquid phases upon completion, enabling simple phase separation to remove inorganic salts and eliminating the need for energy-intensive distillation to dryness.
Q: Is ethanolic HCl still required for the deprotection step?
A: No, the improved process replaces impractical ethanolic HCl with catalytic amounts of 30% aqueous HCl in methanol, significantly simplifying the operational workflow and safety profile.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-chloro-4-(1H-pyrazol-3-yl)benzonitrile Supplier
The technological advancements detailed in patent CN107428695B underscore the importance of having a manufacturing partner who understands the intricacies of process chemistry. NINGBO INNO PHARMCHEM stands at the forefront of this capability, leveraging deep expertise in catalytic cross-coupling and heterocyclic chemistry to deliver superior intermediates. As a dedicated CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. Our commitment to quality is backed by rigorous QC labs and stringent purity specifications, guaranteeing that every batch of 2-chloro-4-(1H-pyrazol-3-yl)benzonitrile meets the exacting standards required for oncology drug development.
We invite global pharmaceutical partners to collaborate with us to unlock the full potential of this optimized synthesis. By leveraging our technical capabilities, you can achieve significant efficiencies in your supply chain while maintaining the highest standards of product quality. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our implementation of this patented process can drive value for your organization.
