Advanced Continuous Flow Nitration for High-Purity Rucaparib Intermediates and Commercial Scale-Up
Introduction to Advanced PARP Inhibitor Intermediate Synthesis
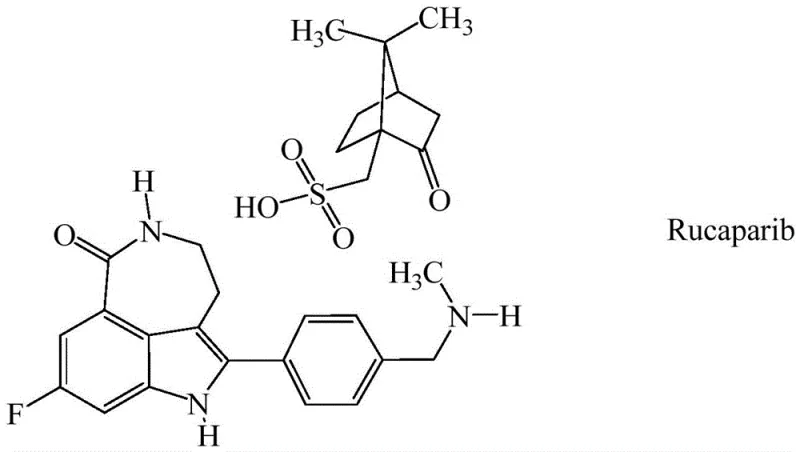
The development of potent poly ADP-ribose polymerase (PARP) inhibitors, such as Rucaparib, has revolutionized the treatment landscape for patients with BRCA-mutated cancers. As the demand for these life-saving oncology therapies grows, the pharmaceutical industry faces increasing pressure to secure reliable supply chains for critical intermediates. Patent CN110944974B introduces a groundbreaking methodology for synthesizing 5-fluoro-2-methyl-3-nitrobenzoic acid and its methyl ester, which serve as pivotal building blocks in the construction of the Rucaparib core structure. This patent addresses long-standing challenges in regioselectivity and impurity control that have plagued traditional batch synthesis routes.
By leveraging a sophisticated combination of oleum and fuming nitric acid within a continuous flow framework, this technology achieves a dramatic enhancement in product quality, transforming a historically brown, impure solid into a high-purity colorless crystalline material. For R&D directors and procurement specialists, understanding the nuances of this process is essential for evaluating potential suppliers who can deliver the stringent purity profiles required for modern API manufacturing. The shift from conventional batch processing to this novel continuous approach represents a significant leap forward in process chemistry efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
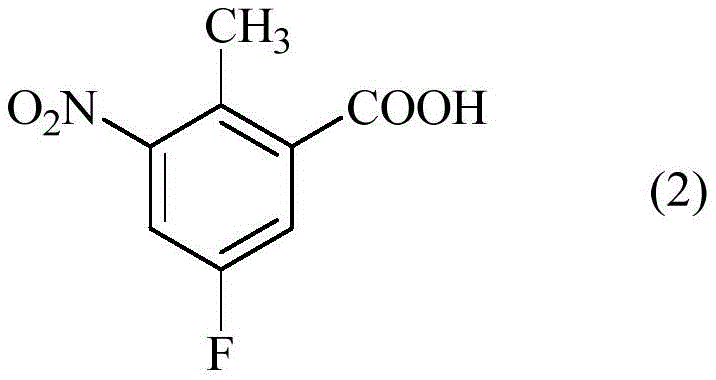
Historically, the synthesis of 5-fluoro-2-methyl-3-nitrobenzoic acid has been fraught with inefficiencies that compromise both yield and purity. Prior art, such as the methods reported by Gillmore et al., relied on the use of concentrated sulfuric acid and concentrated nitric acid for the nitration of 5-fluoro-2-methylbenzoic acid. While functional, this traditional approach suffers from severe drawbacks, primarily the uncontrolled formation of undesirable dinitro derivatives. In comparative studies, these conventional conditions resulted in dinitro impurity levels as high as 45%, necessitating complex and costly purification steps to meet pharmaceutical standards.
Furthermore, the product obtained from these older methods was typically a brown solid with a modest yield of approximately 52%. This low yield not only increases the cost of goods sold due to material loss but also generates a substantial amount of chemical waste, posing environmental disposal challenges. The inability to effectively suppress over-nitration at the 3-position while avoiding attack at other positions on the aromatic ring highlights the lack of selectivity inherent in using standard mixed acid systems without the stabilizing influence of free sulfur trioxide found in oleum.
The Novel Approach
The process disclosed in patent CN110944974B fundamentally reimagines the nitration step by introducing oleum (fuming sulfuric acid) alongside fuming nitric acid. This modification creates a super-acidic medium that alters the electrophilic character of the nitrating species, thereby enhancing regioselectivity for the desired 3-nitro position. The result is a stark contrast to previous methods: the new process yields a colorless solid with yields reaching up to 90% in optimized continuous flow examples. Most critically, the content of the problematic dinitro derivative is suppressed to less than 0.5%, effectively eliminating the need for aggressive downstream purification.
This novel approach also facilitates a telescoped synthesis where the nitration product is directly converted to the methyl ester without isolation. By integrating the nitration and esterification steps, the process minimizes handling time and exposure to potentially unstable intermediates. The use of fuming reagents ensures complete conversion of the starting material, 5-fluoro-2-methylbenzoic acid, thereby maximizing atom economy. This level of efficiency transforms the production of this key intermediate from a bottleneck into a streamlined, high-throughput operation suitable for commercial scale-up.
Mechanistic Insights into Oleum-Mediated Electrophilic Aromatic Substitution
The core chemical transformation in this patent is an electrophilic aromatic substitution where the methyl group directs the incoming nitro group to the ortho position (relative to itself), which corresponds to the 3-position on the benzoic acid ring. The presence of the fluorine atom and the carboxylic acid group exerts electron-withdrawing effects that generally deactivate the ring, making nitration challenging. However, the use of oleum introduces free sulfur trioxide (SO3) into the reaction matrix. This species acts as a powerful dehydrating agent and helps generate the nitronium ion (NO2+) more effectively than concentrated sulfuric acid alone, ensuring sufficient electrophilic strength to overcome the deactivation of the aromatic ring.
Crucially, the specific ratio of oleum to fuming nitric acid modulates the reactivity to prevent poly-nitration. In conventional mixed acid, the water produced during the reaction dilutes the acid strength over time, often leading to inconsistent reaction rates and side reactions. In contrast, oleum scavenges the water produced, maintaining a consistently high acidity and nitrating power throughout the reaction duration. This stability is key to suppressing the formation of the dinitro byproduct (Formula 4), which typically arises when the reaction conditions become too harsh or uncontrolled. The mechanistic precision offered by this reagent system ensures that the reaction stops predominantly at the mono-nitro stage.
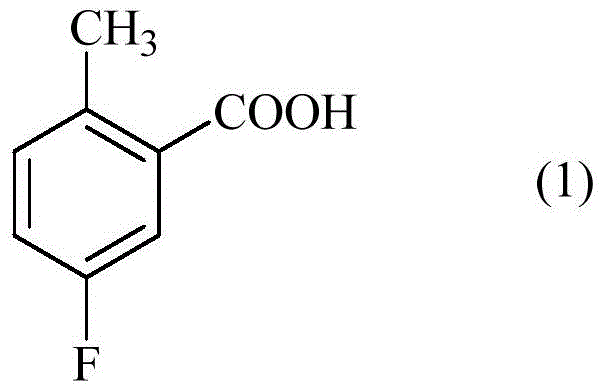
Furthermore, the subsequent esterification mechanism involves the protonation of the carboxylic acid carbonyl oxygen by the acidic medium, followed by nucleophilic attack by methanol. The patent specifies that this step can be performed continuously at elevated temperatures (80°C to 120°C) in the presence of sulfuric acid. The seamless transition from the highly acidic nitration mixture to the esterification environment without neutralization is a testament to the robustness of the chemical design. This compatibility allows for a true telescoped process, where the intermediate acid does not need to be isolated, thereby reducing the potential for degradation or contamination during work-up procedures.
How to Synthesize Methyl 5-Fluoro-2-Methyl-3-Nitrobenzoate Efficiently
To implement this high-efficiency synthesis, manufacturers must adopt a continuous flow strategy that precisely controls residence time and temperature. The patent outlines a specific protocol where two distinct feed streams are prepared: one containing the nitrating agents (fuming nitric acid, oleum, and sulfuric acid) and the other containing the substrate dissolved in a sulfuric acid/oleum mixture. These streams are pre-cooled and then merged within a microreactor or static mixer. The rapid mixing and heat exchange capabilities of flow chemistry equipment are essential to manage the exotherm of the nitration reaction safely.
- Prepare Feed 1 by mixing fuming nitric acid, concentrated sulfuric acid, and fuming sulfuric acid, and prepare Feed 2 by dissolving the starting benzoic acid in a mixture of concentrated sulfuric acid and oleum.
- Pre-cool both feeds to approximately 20°C and pump them into a microreactor or static mixer with a residence time of roughly 17 seconds to effect nitration.
- Directly feed the crude nitration mixture into a second reactor stage with methanol and acid catalyst at 100°C for esterification, followed by cooling and filtration to isolate the pure methyl ester.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the technology described in CN110944974B offers tangible strategic benefits beyond mere chemical elegance. The primary advantage lies in the drastic reduction of raw material costs per kilogram of finished product. By boosting the yield from a historical average of roughly 52% to over 90%, the process effectively nearly doubles the output from the same amount of starting material. This yield enhancement directly translates to significant cost savings in the procurement of 5-fluoro-2-methylbenzoic acid and reduces the volume of waste solvents and acids that require expensive disposal, aligning with green chemistry initiatives.
- Cost Reduction in Manufacturing: The elimination of the dinitro impurity is a major cost driver. In traditional processes, removing 45% of a structurally similar byproduct requires extensive chromatography or recrystallization, which consumes vast amounts of solvent and energy. By suppressing this impurity to below 0.5% at the source, the new process removes the need for these costly purification stages. Additionally, the telescoping of nitration and esterification eliminates an entire isolation step, saving on filtration equipment, drying time, and labor costs associated with handling intermediate solids.
- Enhanced Supply Chain Reliability: Continuous flow manufacturing inherently offers better supply continuity compared to batch processing. The ability to run the reactor for extended periods without the stop-start cycles of batch operations ensures a steady stream of product. This reliability is crucial for API manufacturers who cannot afford interruptions in their supply of critical intermediates like 5-fluoro-2-methyl-3-nitrobenzoic acid. Furthermore, the use of standard industrial reagents like oleum and fuming nitric acid ensures that raw material sourcing remains stable and unaffected by niche supply constraints.
- Scalability and Environmental Compliance: Scaling nitration reactions is notoriously dangerous due to the risk of thermal runaway. The use of microreactors, such as the FlowPlate® systems mentioned in the patent examples, mitigates this risk by providing superior surface-area-to-volume ratios for heat dissipation. This safety profile allows for safer scale-up to commercial tonnages without the need for massive, hazard-prone batch reactors. From an environmental perspective, the higher atom economy and reduced solvent usage for purification significantly lower the E-factor (mass of waste per mass of product), facilitating easier compliance with increasingly strict environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this oleum-based nitration process. These insights are derived directly from the experimental data and embodiments provided in the patent documentation, offering clarity on how this technology compares to legacy methods. Understanding these details is vital for technical teams evaluating the feasibility of adopting this route for their own supply chains.
Q: How does the use of oleum improve the purity of 5-fluoro-2-methyl-3-nitrobenzoic acid compared to conventional nitration?
A: Conventional nitration using concentrated sulfuric and nitric acids often leads to significant formation of dinitro byproducts (up to 45%). The patented process utilizes oleum (fuming sulfuric acid) and fuming nitric acid, which creates a more controlled nitrating environment. This specific reagent combination suppresses over-nitration, reducing dinitro derivatives to less than 0.5% and yielding a colorless product instead of the brown solid typically observed in older methods.
Q: What are the scalability advantages of using a microreactor for this nitration process?
A: Nitration reactions are highly exothermic and can be dangerous on a large scale. The patent demonstrates that performing the reaction in a microreactor (such as a FlowPlate®) allows for precise temperature control (-10°C to 30°C) and extremely short residence times (seconds to minutes). This enhances safety by preventing thermal runaway and improves mixing efficiency, leading to consistent quality and complete conversion that is difficult to achieve in large batch reactors.
Q: Can the nitration and esterification steps be telescoped in a continuous process?
A: Yes, the patent explicitly describes a telescoped continuous process where the crude reaction mixture from the nitration step (Feed 3) is used directly as the substrate for the subsequent esterification reaction without isolation. This eliminates intermediate work-up steps, reduces solvent usage, and significantly shortens the overall production cycle time, thereby improving the cost-efficiency of manufacturing the methyl ester derivative.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Fluoro-2-methyl-3-nitrobenzoic acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the synthesis of complex pharmaceutical intermediates requires more than just chemical knowledge; it demands engineering excellence and a commitment to quality. We have closely analyzed the advancements presented in patent CN110944974B and possess the technical capability to implement this continuous flow nitration technology at an industrial scale. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volumetric demands of global API manufacturers while maintaining stringent purity specifications through our rigorous QC labs.
We invite procurement leaders and R&D directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By leveraging our expertise in continuous processing and impurity control, we can offer a supply solution that optimizes both cost and quality for your PARP inhibitor programs. Please contact our technical procurement team to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced manufacturing capabilities can strengthen your supply chain resilience.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
