Advanced Synephrine Sulfonylated Derivatives: Scalable Synthesis for Next-Gen Antibacterial APIs
The pharmaceutical landscape is constantly evolving in the search for potent antibacterial agents capable of overcoming rising drug resistance. A significant breakthrough in this domain is documented in Chinese patent CN112110898B, which discloses a novel class of synephrine sulfonylated derivatives. These compounds, represented by the general Formula I, are not merely structural analogs but represent a strategic hybridization of the synephrine alkaloid scaffold with quinolone pharmacophores. This innovation addresses the critical need for new therapeutic options against resistant pathogens like Staphylococcus aureus and agricultural threats such as Citrus canker. The structural versatility of these molecules allows for extensive SAR (Structure-Activity Relationship) exploration, making them highly valuable candidates for drug development pipelines.
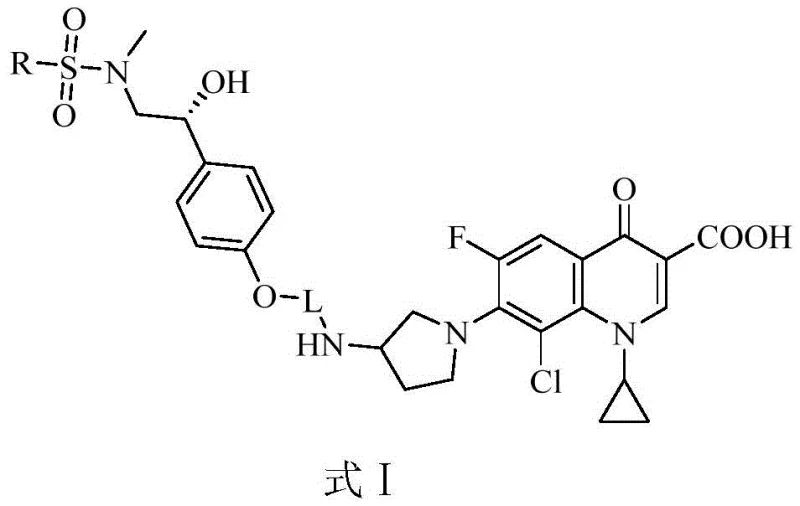
For R&D directors and procurement specialists, the significance of this technology lies in its dual utility: it serves as a potent antibacterial agent and a robust agricultural fungicide. The core structure integrates a chiral synephrine moiety linked via a pyrimidine-piperazine bridge to a fluorinated quinolone core. This complex architecture is designed to maximize binding affinity to bacterial targets while maintaining metabolic stability. As a reliable pharmaceutical intermediate supplier, understanding the nuances of this synthesis is key to securing a stable supply chain for next-generation anti-infectives.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to modifying natural alkaloids like synephrine often suffer from poor regioselectivity and limited biological efficacy. Conventional derivatization typically focuses on simple esterification or etherification, which frequently fails to significantly enhance antibacterial potency beyond the parent compound. Furthermore, many existing synthetic routes for quinolone-alkaloid hybrids involve harsh reaction conditions, such as high temperatures or strong acidic environments, which can lead to racemization of the chiral center in synephrine. This loss of stereochemical integrity is detrimental, as the (R)-enantiomer is often responsible for the desired biological activity. Additionally, older methods may lack a modular linker system, making it difficult to fine-tune the physicochemical properties required for optimal bioavailability and tissue penetration.
The Novel Approach
The methodology outlined in patent CN112110898B overcomes these hurdles through a sophisticated multi-step strategy that preserves chirality and maximizes yield. By employing a Boc-protection strategy early in the synthesis, the process safeguards the sensitive amino group of synephrine from unwanted side reactions. The introduction of a heterocyclic linker, specifically a pyrimidine ring, provides a rigid yet adaptable spacer that optimally positions the pharmacophores for receptor interaction. This novel approach allows for the late-stage diversification of the sulfonyl group, enabling the rapid generation of a library of derivatives (TM9-1 to TM9-18) without rebuilding the entire molecule. This modularity is a game-changer for cost reduction in API manufacturing, as it streamlines the production of diverse analogs from a common advanced intermediate.
Mechanistic Insights into the Multi-Step Convergent Synthesis
The synthesis of these high-purity pharmaceutical intermediates relies on a convergent strategy that couples two complex fragments: the protected synephrine-linker unit and the quinolone core. The mechanism begins with the nucleophilic substitution of the phenolic hydroxyl group of protected synephrine onto a dichloropyrimidine linker. This step is critical as it establishes the ether linkage that connects the two halves of the molecule. Subsequently, the remaining chloro-group on the pyrimidine ring undergoes a second nucleophilic aromatic substitution with the secondary amine of the piperazine ring on the clinafloxacin derivative. This coupling is facilitated by polar aprotic solvents like DMSO and mild bases like potassium carbonate, which activate the nucleophile without degrading the sensitive fluorinated quinolone structure.
Impurity control is paramount in this sequence, particularly during the final sulfonylation step. The reaction between the deprotected amine (Intermediate IMd) and various aromatic sulfonyl chlorides must be conducted at low temperatures (-5°C) to prevent over-sulfonylation or hydrolysis of the sulfonyl chloride. The use of dichloromethane as a solvent ensures good solubility of the organic substrates while allowing for easy workup. The purification process typically involves column chromatography, which effectively removes unreacted starting materials and bis-sulfonylated byproducts. This rigorous control ensures that the final TM9 series compounds meet the stringent purity specifications required for preclinical evaluation, minimizing the risk of toxicological issues arising from trace impurities.
How to Synthesize Synephrine Sulfonylated Derivatives Efficiently
The preparation of these derivatives follows a logical five-step sequence that balances yield with operational simplicity. The process starts with the protection of synephrine, followed by linker attachment, coupling with the quinolone, deprotection, and final functionalization. Each step has been optimized in the patent examples to provide reproducible results on a laboratory scale, which serves as a solid foundation for commercial scale-up of complex pharmaceutical intermediates. The following guide outlines the critical operational parameters derived directly from the patent data.
- Protect the amino group of synephrine using Boc2O and saturated sodium carbonate to form intermediate IMa.
- React intermediate IMa with a linker reagent (e.g., 4,6-dichloropyrimidine) in DCM with potassium carbonate to obtain intermediate IMb.
- Couple intermediate IMb with clinafloxacin in DMSO using K2CO3 and KI at 45°C to generate the key intermediate IMc.
- Remove the Boc protecting group from IMc using ethyl acetate-HCl to yield intermediate IMd.
- Perform the final sulfonylation by reacting IMd with aromatic sulfonyl chloride in DCM at low temperature (-5°C) to obtain the target TM9 derivatives.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the synthesis route described in CN112110898B offers distinct advantages over traditional antibiotic manufacturing processes. The reliance on readily available starting materials, such as synephrine (which can be sourced from citrus processing waste) and commercially available sulfonyl chlorides, mitigates the risk of raw material shortages. This abundance of feedstock translates directly into enhanced supply chain reliability, ensuring that production schedules are not disrupted by niche reagent scarcity. Furthermore, the reaction conditions are generally mild, avoiding the need for specialized high-pressure equipment or cryogenic cooling beyond standard low-temperature reactors, which simplifies facility requirements.
- Cost Reduction in Manufacturing: The synthetic route eliminates the need for expensive transition metal catalysts, which are often required in cross-coupling reactions for similar structures. By relying on nucleophilic substitutions mediated by inexpensive inorganic bases like potassium carbonate and sodium carbonate, the process significantly lowers the cost of goods sold (COGS). Additionally, the high yields reported in the patent examples, such as the 65.8% yield for the key coupling step, indicate a material-efficient process that minimizes waste disposal costs. The ability to purify intermediates via crystallization or simple chromatography further reduces downstream processing expenses compared to more complex purification techniques.
- Enhanced Supply Chain Reliability: The modular nature of the synthesis allows for the stocking of key intermediates like IMc and IMd. Since these intermediates themselves show biological activity, they can serve as backup products if the final sulfonylation step faces delays. The use of common organic solvents like DCM, DMSO, and methanol ensures that solvent supply chains remain robust and unaffected by geopolitical fluctuations that might impact exotic reagents. This stability is crucial for maintaining continuous production runs and meeting the demanding delivery timelines of global pharmaceutical partners.
- Scalability and Environmental Compliance: The process demonstrates excellent scalability potential, with reaction times ranging from 1.5 to 3.5 hours for the final step, suggesting a high throughput capability. The absence of heavy metals simplifies the environmental compliance profile, reducing the burden on wastewater treatment facilities. The use of aqueous workups with saturated sodium chloride and sodium carbonate solutions allows for the efficient recovery of organic solvents, aligning with green chemistry principles. This environmental friendliness is increasingly becoming a prerequisite for vendor qualification in the eyes of major multinational corporations committed to sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these synephrine derivatives. The answers are derived from the experimental data and specifications provided in the source patent, ensuring accuracy and relevance for technical decision-makers.
Q: What is the antibacterial potency of these synephrine derivatives compared to clinical drugs?
A: According to patent CN112110898B, the TM9 series exhibits strong inhibitory activity against Staphylococcus aureus, with MIC values ranging from 0.2 to 32 μg/mL. Several compounds show activity superior to or equivalent to norfloxacin, vancomycin, and ciprofloxacin.
Q: Are the intermediates IMc and IMd biologically active?
A: Yes, the patent data indicates that intermediates IMc and IMd possess significant antibacterial activity themselves, with an MIC of 0.4 μg/mL against Staphylococcus aureus, which is notably stronger than several standard clinical antibiotics.
Q: What are the critical reaction conditions for the final sulfonylation step?
A: The final coupling of intermediate IMd with aromatic sulfonyl chloride requires precise temperature control, specifically stirring at -5°C in a constant temperature low-temperature reactor, using dichloromethane as the solvent and potassium carbonate as the base.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Synephrine Derivatives Supplier
The development of synephrine sulfonylated derivatives represents a significant leap forward in antibacterial therapy, combining natural product scaffolds with modern medicinal chemistry. NINGBO INNO PHARMCHEM stands at the forefront of this innovation, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific requirements of this synthesis, including low-temperature reactors for the critical sulfonylation step and rigorous QC labs to ensure stringent purity specifications for every batch of TM9 series intermediates we produce.
We invite procurement leaders and R&D teams to collaborate with us to accelerate your drug development programs. By leveraging our expertise in process optimization, we can help you achieve substantial cost savings and reduce lead time for high-purity pharmaceutical intermediates. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis, specific COA data for the TM9 series, and comprehensive route feasibility assessments tailored to your project's unique needs.
