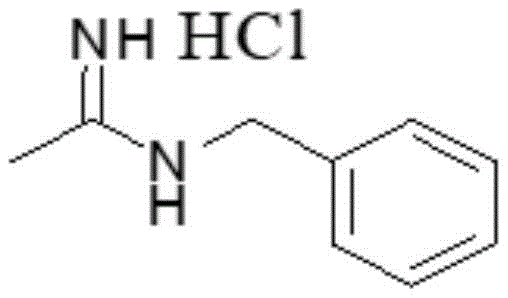
Advanced Synthesis of N-benzylacetamidine Hydrochloride for Scalable Pharmaceutical Manufacturing
Advanced Synthesis of N-benzylacetamidine Hydrochloride for Scalable Pharmaceutical Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and environmentally compliant synthetic routes for critical intermediates. Patent CN110878032B, published in March 2022, introduces a significant technological breakthrough in the synthesis of N-benzylacetamidine hydrochloride, a vital building block in the construction of complex therapeutic agents. This patent details a novel three-step process that fundamentally addresses the limitations of traditional amidine synthesis, specifically targeting the elimination of hazardous byproducts and the enhancement of overall process efficiency. For R&D directors and procurement specialists, this technology represents a shift towards greener chemistry without compromising on yield or purity. The method leverages a catalytic hydrogenation strategy that not only improves the quality of the final intermediate but also simplifies the downstream processing requirements, thereby offering a compelling value proposition for commercial-scale manufacturing operations seeking to optimize their supply chain resilience.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of amidine compounds like N-benzylacetamidine hydrochloride has been plagued by significant operational and environmental challenges. Traditional routes, such as those referenced in prior art like WO2015/5615, often rely on reaction conditions that generate substantial amounts of hydrogen chloride gas as a byproduct. This corrosive gas poses severe risks to production equipment, necessitating the use of expensive, corrosion-resistant materials for reactors and piping, which drastically increases capital expenditure. Furthermore, the management of acidic waste streams requires complex neutralization and treatment processes, adding to the operational cost and environmental footprint. From a safety perspective, the handling of corrosive gases increases the risk to personnel and requires stringent safety protocols that can slow down production throughput. These factors collectively make conventional methods less attractive for modern, high-volume manufacturing where cost efficiency and regulatory compliance are paramount.
The Novel Approach
In stark contrast, the method disclosed in patent CN110878032B offers a streamlined and environmentally benign alternative. By utilizing a sequence that involves the generation of a benzyl hydroxylamine toluene solution followed by condensation with acetonitrile and subsequent hydrogenation, the process completely avoids the generation of corrosive hydrogen chloride gas during the reaction phases. The use of Raney nickel as a catalyst under controlled hydrogen pressure allows for a clean reduction step that is both efficient and selective. This approach not only mitigates the equipment corrosion issues associated with older methods but also significantly reduces the volume of three wastes (waste water, waste gas, and waste residue) generated. The mild reaction conditions, typically ranging between 50°C and 80°C, further enhance the safety profile of the operation, making it easier to implement in standard chemical production facilities without requiring specialized high-pressure or high-temperature infrastructure.
Mechanistic Insights into Raney Nickel-Catalyzed Hydrogenation
The core of this synthetic innovation lies in the third step, where the intermediate undergoes catalytic hydrogenation. In this stage, the intermediate, dissolved in ethyl acetate, is subjected to hydrogen pressure in the presence of Raney nickel. This heterogeneous catalysis facilitates the reduction of the nitrile or imine functionality to the desired amidine structure with high selectivity. The choice of Raney nickel is critical, as it provides a high surface area for the reaction to proceed efficiently at moderate temperatures, typically optimized around 65-70°C. The mechanism ensures that side reactions are minimized, which is crucial for maintaining the high purity levels required for pharmaceutical intermediates. The subsequent addition of a hydrogen chloride methanol solution serves to convert the free base into the stable hydrochloride salt, precipitating the product upon cooling. This crystallization step is highly effective in purifying the compound, as evidenced by the patent data showing purity levels consistently above 95%.
Impurity control is another critical aspect where this mechanism excels. By avoiding the harsh acidic conditions of traditional methods, the formation of degradation products and polymeric impurities is significantly reduced. The use of toluene and ethyl acetate as solvents provides a favorable environment for the reaction, ensuring that the intermediates remain stable throughout the process. The nitrogen replacement steps prior to hydrogenation are essential to remove oxygen, preventing potential oxidation side reactions that could compromise the quality of the final product. This attention to detail in the reaction environment ensures that the impurity profile is clean, reducing the burden on downstream purification steps and increasing the overall yield of the usable product. For quality control teams, this translates to more consistent batch-to-batch performance and easier compliance with stringent pharmacopeial standards.
How to Synthesize N-benzylacetamidine Hydrochloride Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale production. The process begins with the careful preparation of the benzyl hydroxylamine free base, followed by a controlled condensation reaction with acetonitrile. The final hydrogenation step requires precise control of pressure and temperature to maximize yield. While the patent provides specific examples, scaling this process requires adherence to strict safety and operational guidelines to ensure reproducibility. The detailed standardized synthesis steps for implementing this route in a GMP environment are provided in the technical guide below.
- Generate benzyl hydroxylamine free base in toluene using sodium hydroxide at low temperature.
- Condense the free base with acetonitrile under heating to form the intermediate.
- Perform catalytic hydrogenation using Raney nickel followed by acidification to obtain the final hydrochloride salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis method offers tangible strategic benefits beyond mere technical superiority. The elimination of corrosive gas generation directly translates to reduced maintenance costs for production facilities, extending the lifespan of reactors and associated infrastructure. This durability ensures greater supply continuity, as equipment downtime due to corrosion-related failures is minimized. Furthermore, the use of common, commercially available solvents like toluene, acetonitrile, and ethyl acetate ensures that raw material sourcing is stable and not subject to the volatility associated with specialty reagents. The simplified waste treatment process also reduces the environmental compliance burden, allowing for faster regulatory approvals and smoother operations in regions with strict environmental laws.
- Cost Reduction in Manufacturing: The process significantly lowers operational costs by removing the need for expensive corrosion-resistant equipment and complex gas scrubbing systems. The high yield and purity reduce the amount of raw material required per unit of product, optimizing material costs. Additionally, the simplified post-treatment and crystallization steps reduce energy consumption and labor hours associated with purification, leading to substantial overall cost savings in the manufacturing of this pharmaceutical intermediate.
- Enhanced Supply Chain Reliability: By utilizing a robust and scalable synthetic route, suppliers can ensure consistent delivery schedules. The mild reaction conditions reduce the risk of batch failures due to thermal runaway or equipment stress, enhancing production reliability. The availability of raw materials such as benzyl hydroxylamine hydrochloride and Raney nickel from multiple global sources further mitigates supply chain risks, ensuring that production can be maintained even during market fluctuations.
- Scalability and Environmental Compliance: The green chemistry principles embedded in this method make it highly scalable from kilogram to multi-ton production without significant re-engineering. The reduction in hazardous waste generation aligns with global sustainability goals, making it easier to obtain environmental permits. This compliance advantage is critical for long-term supply contracts, as it future-proofs the production process against tightening environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and supply of N-benzylacetamidine hydrochloride. These answers are derived directly from the technical specifications and beneficial effects described in the patent literature, providing a reliable basis for decision-making.
Q: What are the primary advantages of this synthesis method over conventional routes?
A: This method eliminates the generation of corrosive hydrogen chloride gas found in prior art, significantly reducing equipment maintenance costs and environmental waste treatment burdens while achieving higher product purity.
Q: What is the expected purity and yield of the final product?
A: According to patent data, the process consistently yields products with purity exceeding 95%, with optimized examples demonstrating yields up to 90% and purity reaching 99%.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the reaction conditions are mild (50-80°C), use readily available solvents like toluene and ethyl acetate, and avoid hazardous gas emissions, making it highly scalable and compliant with green chemistry standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-benzylacetamidine Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we understand the critical role that high-quality intermediates play in the drug development lifecycle. Our team of experts possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory synthesis to industrial manufacturing is seamless. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of N-benzylacetamidine hydrochloride meets the highest industry standards. Our capability to implement the green synthesis methods described in patent CN110878032B allows us to offer a product that is not only chemically superior but also produced with a focus on sustainability and cost-efficiency.
We invite pharmaceutical companies and chemical manufacturers to collaborate with us to optimize their supply chains. By leveraging our technical expertise, you can achieve significant process improvements and cost reductions. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific production needs. We are ready to provide specific COA data and route feasibility assessments to support your next project, ensuring that you have a reliable partner for your complex chemical synthesis requirements.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
