Advanced Microwave-Assisted Synthesis of Aceclofenac for Scalable Pharmaceutical Manufacturing
Advanced Microwave-Assisted Synthesis of Aceclofenac for Scalable Pharmaceutical Manufacturing
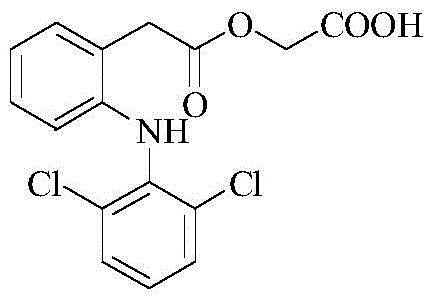
The pharmaceutical industry is constantly seeking robust, scalable, and high-purity synthetic routes for non-steroidal anti-inflammatory drugs (NSAIDs). Patent CN110143891B introduces a groundbreaking preparation method for Aceclofenac, leveraging microwave-assisted organic synthesis (MAOS) to overcome the limitations of traditional thermal heating. This technology represents a significant leap forward in the manufacturing of this critical pharmaceutical intermediate, offering a pathway that drastically reduces reaction times while enhancing product purity. By utilizing specific microwave parameters and a optimized solvent system, the process achieves superior selectivity during the acidolysis step, effectively minimizing the generation of diclofenac, a common and difficult-to-remove impurity. For R&D directors and procurement specialists alike, this patent outlines a methodology that not only improves chemical efficiency but also aligns with modern green chemistry principles by reducing energy consumption and solvent usage.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for Aceclofenac typically rely on conductive heating methods, such as oil baths or heating mantles, which suffer from inherent thermal inefficiencies. In these conventional processes, heat transfer occurs from the outside of the reaction vessel inward, often leading to significant temperature gradients and localized overheating. To drive the alkylation and subsequent acidolysis reactions to completion, manufacturers are frequently forced to employ high temperatures and extended reaction times, sometimes exceeding 24 hours for reflux conditions. This prolonged exposure to thermal stress promotes side reactions, particularly the hydrolysis of the ester linkage, resulting in elevated levels of diclofenac impurities in the final product. Furthermore, the post-processing of these crude mixtures is notoriously difficult, requiring extensive purification steps that erode overall yield and increase production costs. The reliance on harsh conditions also poses safety risks and increases the facility's carbon footprint due to excessive energy demand.
The Novel Approach
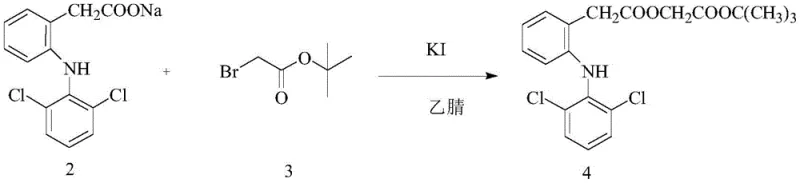
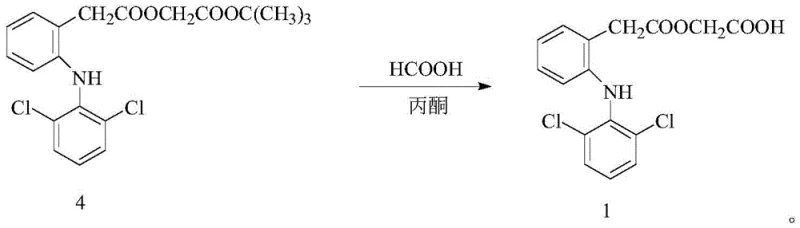
In stark contrast, the novel approach detailed in the patent utilizes microwave irradiation to drive the chemical transformations with unprecedented efficiency. The process is divided into two distinct stages, both optimized for microwave conditions. The first stage involves the reaction of diclofenac sodium with tert-butyl bromoacetate in the presence of a potassium iodide (KI) catalyst within an acetonitrile solvent system. Under microwave irradiation at a modest power of 100W and a temperature of merely 30°C, this alkylation reaches completion in just 15 minutes, yielding the aceclofenac tert-butyl ester intermediate with a remarkable yield of 91.6%. The second stage employs a mixed solvent system of formic acid and acetone to effect the acidolysis of the tert-butyl ester. Operating at 200W and 65°C for only 10 minutes, this step achieves high conversion rates while maintaining exceptional selectivity, ensuring that the ethoxy group remains intact and diclofenac formation is suppressed to negligible levels.
Mechanistic Insights into Microwave-Assisted Alkylation and Acidolysis
The efficacy of this synthesis lies in the fundamental physics of microwave dielectric heating. Unlike conventional conduction, microwaves interact directly with polar molecules and ionic species present in the reaction mixture, such as the diclofenac sodium salt and the polar solvents like acetonitrile and formic acid. This interaction causes molecular dipoles to oscillate at billions of cycles per second, generating heat volumetrically throughout the entire reaction mass simultaneously. This "internal heating" mechanism eliminates thermal gradients, ensuring that every molecule experiences the exact same activation energy environment. In the alkylation step, the KI catalyst plays a crucial role by facilitating the nucleophilic attack of the diclofenac anion on the bromoacetate electrophile. The microwave field likely enhances the mobility of these ionic species, accelerating the collision frequency and driving the equilibrium toward the product much faster than thermal agitation alone could achieve. This results in a cleaner reaction profile with fewer byproducts derived from thermal decomposition.
Furthermore, the acidolysis mechanism benefits significantly from the selective heating properties of microwaves. The transition state for the cleavage of the tert-butyl group is stabilized more effectively under rapid, uniform heating conditions. The use of formic acid as both a reactant and a microwave-absorbing medium ensures that the energy is focused exactly where the chemical bond breaking needs to occur. This precision prevents the over-heating that typically leads to the hydrolysis of the adjacent ester linkage, which is the primary source of diclofenac impurity in traditional methods. By strictly controlling the microwave power and reaction time, the process kinetically favors the desired deprotection pathway while thermodynamically disfavoring the degradation pathways. This mechanistic control is what allows the process to achieve such high purity without the need for complex chromatographic separations, relying instead on simple crystallization techniques.
How to Synthesize Aceclofenac Efficiently
The synthesis of Aceclofenac via this microwave-assisted route is designed for operational simplicity and robustness, making it an ideal candidate for technology transfer from the laboratory to pilot and commercial scales. The protocol eliminates the need for exotic reagents or ultra-low temperature cryogenics, relying instead on commodity chemicals and standard microwave reactor hardware. The critical success factors involve the precise stoichiometric balance of the reactants—specifically maintaining a slight excess of the alkylating agent to ensure complete consumption of the valuable diclofenac sodium—and the careful selection of microwave power settings to avoid runaway exotherms. Detailed standardized operating procedures for mixing, irradiation, and work-up are essential to replicate the high yields reported in the patent data. For a comprehensive breakdown of the specific molar ratios, solvent volumes, and crystallization parameters required to execute this synthesis successfully, please refer to the technical guide below.
- React diclofenac sodium with tert-butyl bromoacetate using KI catalyst in acetonitrile under microwave irradiation (100W, 30°C) to form aceclofenac tert-butyl ester.
- Purify the intermediate ester via crystallization in ethanol to remove unreacted starting materials and salts.
- Perform acidolysis of the tert-butyl ester using formic acid and acetone under microwave conditions (200W, 65°C) to yield crude aceclofenac, followed by recrystallization in toluene.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this microwave-assisted technology offers substantial strategic advantages for procurement managers and supply chain directors overseeing the sourcing of high-purity pharmaceutical intermediates. The most immediate impact is seen in the drastic reduction of cycle times. By compressing reaction times from days or many hours down to mere minutes, manufacturers can significantly increase the throughput of existing production assets without the need for capital-intensive expansion of reactor farm capacity. This acceleration translates directly into improved cash flow and the ability to respond more agilely to market demand fluctuations. Additionally, the milder reaction conditions reduce the thermal load on equipment, potentially extending the maintenance intervals for reactors and associated infrastructure, thereby lowering the total cost of ownership for the manufacturing facility.
- Cost Reduction in Manufacturing: The economic benefits of this process are multifaceted, driven primarily by the elimination of prolonged heating periods and the reduction in solvent volumes. Traditional reflux methods require vast amounts of energy to maintain boiling temperatures for extended durations, whereas microwave heating is highly energy-efficient, converting electrical energy directly into chemical potential with minimal loss. Furthermore, the high selectivity of the reaction means that the yield of the desired product is maximized while the formation of waste byproducts is minimized. This efficiency reduces the burden on waste treatment facilities and lowers the cost of raw materials per kilogram of finished product. The simplified purification process, which relies on straightforward crystallization rather than complex column chromatography, further drives down operational expenditures by reducing labor hours and consumable costs associated with downstream processing.
- Enhanced Supply Chain Reliability: Supply chain resilience is critically dependent on the consistency and predictability of the manufacturing process. The microwave-assisted method offers superior reproducibility compared to batch thermal heating, as the reaction parameters (power, temperature, time) are digitally controlled and less susceptible to external environmental variables. This consistency ensures that every batch meets stringent quality specifications, reducing the risk of batch failures or out-of-specification results that can disrupt supply schedules. Moreover, the use of readily available starting materials like diclofenac sodium and tert-butyl bromoacetate ensures that the supply chain is not vulnerable to shortages of exotic catalysts or reagents. The robustness of the process allows for reliable long-term planning and secure contracting with downstream API manufacturers.
- Scalability and Environmental Compliance: As regulatory pressures regarding environmental sustainability intensify, this process positions itself as a greener alternative to conventional synthesis. The reduction in energy consumption directly correlates to a lower carbon footprint, aligning with corporate sustainability goals and regulatory compliance standards. The use of acetonitrile and acetone, which are widely recycled in the pharmaceutical industry, facilitates solvent recovery loops that further minimize environmental impact. The scalability of microwave technology has matured significantly, with continuous flow microwave reactors now capable of handling multi-ton production campaigns. This technological maturity ensures that the process can be scaled from kilogram to metric ton quantities without losing the efficiency gains observed at the laboratory scale, providing a future-proof solution for growing market demands.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this microwave-assisted synthesis route. These insights are derived directly from the experimental data and beneficial effects described in the patent literature, providing a clear understanding of the process capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this methodology for their specific production lines.
Q: How does microwave assistance improve the purity of Aceclofenac compared to traditional heating?
A: Microwave heating provides uniform internal heating and precise temperature control, which minimizes the thermal degradation of the ethoxy group and significantly reduces the formation of diclofenac impurities during the acidolysis step.
Q: What are the key advantages of using KI as a catalyst in this synthesis route?
A: Potassium iodide acts as an efficient nucleophilic catalyst that facilitates the substitution reaction between diclofenac sodium and tert-butyl bromoacetate, allowing the reaction to proceed rapidly at lower temperatures (30°C) compared to conventional reflux methods.
Q: Is this microwave process scalable for industrial production of pharmaceutical intermediates?
A: Yes, the process utilizes standard solvents like acetonitrile and acetone and operates at mild temperatures, making it highly adaptable for continuous flow microwave reactors or large-scale batch processing with appropriate engineering controls.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aceclofenac Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient, high-quality synthesis routes in the competitive landscape of global pharmaceuticals. Our team of expert chemists has extensively analyzed the microwave-assisted preparation of Aceclofenac and possesses the technical capability to implement this advanced methodology at scale. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab bench to manufacturing plant is seamless and compliant with cGMP standards. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of Aceclofenac we deliver meets the exacting requirements of international regulatory bodies.
We invite you to collaborate with us to optimize your supply chain for this vital anti-inflammatory intermediate. By leveraging our expertise in microwave chemistry and process optimization, we can help you achieve significant operational efficiencies. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can support your long-term strategic goals.
