Scalable Synthesis of Protected 4-Aminomethylpyrrolidin-3-ones for Quinolone Antibiotic Manufacturing
Scalable Synthesis of Protected 4-Aminomethylpyrrolidin-3-ones for Quinolone Antibiotic Manufacturing
The pharmaceutical industry constantly seeks robust, scalable pathways for constructing complex heterocyclic scaffolds essential for next-generation therapeutics. Patent CN1149192C introduces a transformative methodology for the preparation of protected 4-aminomethylpyrrolidin-3-ones, specifically compounds of Formula (1), which serve as critical intermediates in the synthesis of potent quinolone antibiotics. This technology addresses long-standing inefficiencies in prior art by replacing hazardous stoichiometric reductants with a catalytic hydrogenation protocol using Raney nickel. For R&D directors and process chemists, this represents a significant leap forward in impurity profile management and operational safety, offering a streamlined route that bypasses the cumbersome re-oxidation steps previously required to restore the ketone functionality. The strategic implementation of this chemistry allows for the production of high-purity intermediates suitable for GMP manufacturing environments.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of these pivotal pyrrolidine intermediates relied heavily on reduction protocols employing sodium borohydride (NaBH4) in combination with trifluoroacetic acid (TFA). While effective on a small laboratory scale, this approach presents severe liabilities when translated to commercial production scales ranging from tens to hundreds of kilograms. A primary failure mode of the conventional method is the formation of persistent byproducts, specifically the boron-containing species depicted as Formula (3) and the over-reduced pyrrolidine derivative shown as Formula (4).  These impurities are notoriously difficult to separate via standard recrystallization techniques, leading to compromised purity profiles that fail to meet stringent regulatory standards for antibiotic intermediates. Furthermore, the reaction generates hydrogen gas discontinuously, creating unpredictable pressure spikes and significant explosion hazards that complicate reactor engineering and safety protocols. Perhaps most critically, the hydride reduction often reduces the ketone moiety to an alcohol, necessitating a subsequent, expensive oxidation step using pyridine-sulfur trioxide complexes to regenerate the required carbonyl group, thereby inflating both material costs and waste generation.
These impurities are notoriously difficult to separate via standard recrystallization techniques, leading to compromised purity profiles that fail to meet stringent regulatory standards for antibiotic intermediates. Furthermore, the reaction generates hydrogen gas discontinuously, creating unpredictable pressure spikes and significant explosion hazards that complicate reactor engineering and safety protocols. Perhaps most critically, the hydride reduction often reduces the ketone moiety to an alcohol, necessitating a subsequent, expensive oxidation step using pyridine-sulfur trioxide complexes to regenerate the required carbonyl group, thereby inflating both material costs and waste generation.
The Novel Approach
In stark contrast, the novel process disclosed in the patent utilizes a catalytic hydrogenation strategy centered on Raney nickel to selectively reduce the cyano group of the starting material, Formula (5), directly to the aminomethylene intermediate, Formula (6). This mechanistic shift fundamentally alters the impurity landscape by eliminating the formation of boron-based adducts and avoiding the uncontrolled gas evolution associated with hydride acids. The new route operates under mild conditions, typically between room temperature and 60°C and at hydrogen pressures of 4 to 10 atmospheres, which are readily achievable in standard industrial equipment. By circumventing the alcohol formation entirely, the process obviates the need for the costly and environmentally burdensome re-oxidation step, resulting in a shorter, more linear synthetic sequence. This direct conversion not only enhances the overall yield but also significantly simplifies the downstream purification workflow, making it an ideal candidate for cost-effective, large-scale manufacturing.
Mechanistic Insights into Raney Nickel-Catalyzed Hydrogenation
The core innovation of this technology lies in the selective hydrogenation of the alpha-cyano ketone derivative using a heterogeneous Raney nickel catalyst. In the first stage, the nitrile group of Formula (5) undergoes partial reduction to form the exocyclic imine or enamine species, Formula (6), rather than proceeding fully to the primary amine. This selectivity is finely tuned by the choice of solvent, with alcohols like methanol or isopropanol proving superior in accelerating reaction kinetics, and the addition of basic additives such as ammonia or acetates. These additives play a crucial role in modulating the surface activity of the catalyst, preventing over-reduction to the saturated amine at this stage and ensuring the preservation of the double bond character necessary for the subsequent protection step. The result is a high-purity intermediate, often exceeding 99.8% purity by GC analysis, which provides a clean foundation for the remainder of the synthesis.
Following the initial reduction, the amino group is protected, typically with a tert-butyloxycarbonyl (Boc) group, to yield Formula (7). The final critical transformation involves the selective reduction of the carbon-carbon double bond in Formula (7) to generate the saturated ketone Formula (1). 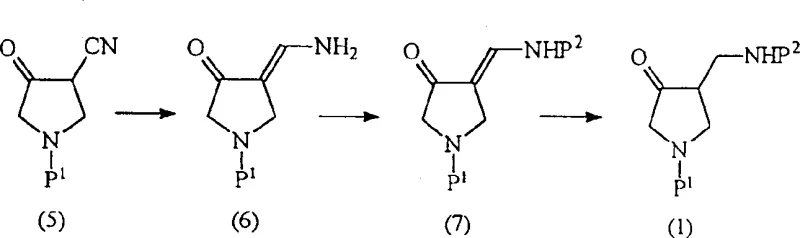 Maintaining the integrity of the 3-position ketone during this final hydrogenation is paramount. The patent discloses that this selectivity is governed by strict pH control of the reaction medium, maintained between pH 3 to 5 or pH 8 to 10 using organic amines or buffered solutions. Under these specific conditions, transition metal catalysts such as palladium on carbon or Lindlar catalysts reduce the alkene without attacking the carbonyl oxygen. This precise control prevents the formation of hydroxyl impurities, ensuring that the final product retains the oxidation state required for coupling with quinolone cores, thereby maximizing the efficiency of the final API assembly.
Maintaining the integrity of the 3-position ketone during this final hydrogenation is paramount. The patent discloses that this selectivity is governed by strict pH control of the reaction medium, maintained between pH 3 to 5 or pH 8 to 10 using organic amines or buffered solutions. Under these specific conditions, transition metal catalysts such as palladium on carbon or Lindlar catalysts reduce the alkene without attacking the carbonyl oxygen. This precise control prevents the formation of hydroxyl impurities, ensuring that the final product retains the oxidation state required for coupling with quinolone cores, thereby maximizing the efficiency of the final API assembly.
How to Synthesize Protected 4-Aminomethylpyrrolidin-3-ones Efficiently
The synthesis of these high-value intermediates follows a logical three-step progression designed for maximum operational simplicity and yield. The process begins with the catalytic hydrogenation of the cyano precursor, followed by in-situ or telescoped protection, and concludes with a selective saturation of the olefinic bond. This sequence has been validated across multiple solvent systems including methanol, ethanol, and tetrahydrofuran, demonstrating robustness against minor variations in reaction conditions. For process engineers looking to implement this technology, the detailed standardized synthesis steps are outlined below to ensure reproducibility and compliance with quality specifications.
- React the alpha-cyano ketone derivative (Formula 5) with a Raney nickel catalyst in a solvent such as methanol or isopropanol under hydrogen pressure (4-10 atm) to produce the aminomethylene intermediate (Formula 6).
- Protect the amino group of Formula 6 using a protecting group agent like di-tert-butyl dicarbonate in the presence of a base to yield the protected enamine (Formula 7).
- Selectively reduce the carbon-carbon double bond of Formula 7 using a transition metal catalyst (e.g., Pd/C) under controlled pH conditions (pH 3-5 or 8-10) to obtain the final saturated ketone (Formula 1).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this Raney nickel-based process offers tangible economic and logistical benefits over legacy hydride reduction methods. The elimination of stoichiometric reducing agents like lithium aluminum hydride or sodium borohydride removes a significant variable cost driver, as these reagents are not only expensive but also require careful handling and disposal protocols. Moreover, the removal of the re-oxidation step means that the costly pyridine-sulfur trioxide complex, a specialized and hazardous reagent, is no longer required, leading to substantial raw material savings. The simplified workflow reduces the number of unit operations, which directly correlates to lower labor costs, reduced energy consumption, and decreased solvent usage per kilogram of product. These efficiencies compound to offer a significantly reduced cost of goods sold (COGS), making the final antibiotic API more competitive in the global marketplace.
- Cost Reduction in Manufacturing: The most immediate financial impact comes from the drastic simplification of the synthetic route. By avoiding the formation of alcohol byproducts, the process completely eliminates the need for a separate oxidation step to regenerate the ketone. In traditional methods, this oxidation requires expensive reagents like pyridine-sulfur trioxide and generates sulfur-containing waste streams that are costly to treat. The new catalytic method achieves the desired oxidation state directly, removing an entire processing stage and its associated utility and material costs. Additionally, the use of Raney nickel, a reusable heterogeneous catalyst, is far more economical than the continuous consumption of stoichiometric hydrides, further driving down the variable cost per batch.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the reliance on hazardous or hard-to-source reagents. Sodium borohydride and trifluoroacetic acid mixtures pose significant storage and transport challenges due to their corrosive nature and potential for gas generation. By shifting to a hydrogenation process using gaseous hydrogen and a stable solid catalyst, the facility reduces its dependency on complex liquid reagent supply chains. The reaction conditions are mild and operate at near-ambient temperatures, reducing the risk of thermal runaways that could shut down production lines. This stability ensures consistent batch-to-batch output, minimizing the risk of stockouts caused by failed batches or extended purification times required to remove stubborn boron impurities.
- Scalability and Environmental Compliance: Scaling chemical processes often reveals hidden bottlenecks related to heat transfer and mixing, particularly with exothermic hydride reductions. The Raney nickel hydrogenation described here is highly scalable, having been demonstrated effectively from gram to multi-kilogram scales without loss of selectivity. The process generates minimal hazardous waste; specifically, it avoids the generation of boron-containing sludge and sulfur waste from oxidation steps, simplifying wastewater treatment and lowering environmental compliance costs. The ability to run the reaction at moderate pressures (4-10 atm) means it can be performed in standard glass-lined or stainless steel hydrogenation reactors available in most CDMO facilities, removing the need for specialized high-pressure equipment investments.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These answers are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on how this method resolves specific manufacturing pain points. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer.
Q: Why is the Raney Nickel method superior to the traditional NaBH4/TFA reduction for this intermediate?
A: The traditional method using sodium borohydride and trifluoroacetic acid often leads to incomplete reduction and dangerous discontinuous hydrogen generation. Furthermore, it produces difficult-to-remove boron-containing byproducts (Formula 3) and over-reduced impurities (Formula 4), necessitating a costly re-oxidation step. The Raney Nickel route avoids these byproducts entirely and eliminates the need for subsequent oxidation.
Q: How is the ketone functionality preserved during the final hydrogenation step?
A: Selectivity is achieved by strictly controlling the pH of the reaction solution between 3 to 5 or 8 to 10 using organic amines or buffered solutions. This specific pH range allows for the reduction of the exocyclic double bond while preventing the reduction of the carbonyl group to an alcohol, ensuring high purity of the final ketone product.
Q: What are the scalability advantages of this process for commercial production?
A: This process operates at moderate temperatures (room temperature to 60°C) and pressures (4-10 atm), which are easily manageable in standard industrial hydrogenation reactors. It removes the safety hazards associated with discontinuous gas evolution found in hydride reductions and avoids expensive, environmentally harmful oxidants like pyridine-sulfur trioxide complexes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Protected 4-Aminomethylpyrrolidin-3-one Supplier
The technological advancements detailed in CN1149192C underscore the complexity and precision required to manufacture high-quality quinolone intermediates. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with unwavering consistency. Our state-of-the-art facilities are equipped to handle the specific hydrogenation and protection chemistries required for this route, adhering to stringent purity specifications and rigorous QC labs to guarantee that every batch meets the exacting standards of the pharmaceutical industry. We understand that the purity of the intermediate directly impacts the quality of the final antibiotic, and our process controls are designed to minimize impurities like those seen in older synthetic routes.
We invite you to collaborate with us to optimize your supply chain for these critical building blocks. Our technical team is prepared to conduct a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how switching to this catalytic route can improve your margins. We encourage potential partners to contact our technical procurement team to request specific COA data and route feasibility assessments, allowing you to make informed decisions based on real-world performance metrics rather than theoretical projections. Let us help you secure a stable, cost-effective source for your quinolone antibiotic intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
