Advanced Manufacturing of Spiro-Quinazolinone Derivatives for Diabetic Complication Therapeutics
Advanced Manufacturing of Spiro-Quinazolinone Derivatives for Diabetic Complication Therapeutics
The pharmaceutical landscape for treating diabetic complications, particularly diabetic neurosis, relies heavily on the availability of high-purity heterocyclic intermediates. Patent CN1042914A discloses a groundbreaking preparation method for a specific class of imidazolidine derivatives, specifically spiro[1,2,3,4-tetrahydroquinazoline-4,4′-imidazolidine]-2,2′,5′-trione and its substituted analogs. These compounds have been identified as potent agents for the prevention and treatment of diabetic complications. The structural versatility of these molecules allows for significant modulation of pharmacological properties, making them a critical focus for modern drug discovery pipelines. As a leading entity in fine chemical manufacturing, understanding the nuances of this synthesis is vital for ensuring a robust supply chain for global pharmaceutical partners.
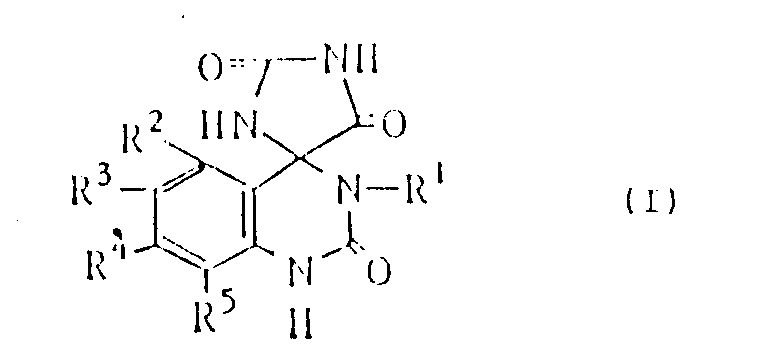
The core structure, as depicted in the patent documentation, features a unique spiro-fused system where a quinazolinone ring is connected to an imidazolidine ring. The substituents R1 through R5 offer extensive opportunities for derivatization, allowing R1 to be a lower alkyl group or hydrogen, while R2 through R5 can vary from hydrogen and halogens to lower alkyl or alkoxy groups. This structural flexibility is paramount for medicinal chemists aiming to optimize bioavailability and potency. The ability to synthesize these complex scaffolds efficiently directly impacts the speed at which new therapeutic candidates can reach clinical trials. Consequently, mastering this synthetic route is not merely a chemical exercise but a strategic imperative for reliable pharmaceutical intermediate supplier operations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex spiro-heterocycles has been plagued by multi-step sequences that necessitate the isolation and purification of unstable intermediates. Conventional routes often suffer from low atom economy and the requirement for harsh reaction conditions that can degrade sensitive functional groups. In many legacy processes, the formation of the spiro-center is the rate-limiting step, often requiring expensive catalysts or protecting group strategies that add significant cost and waste to the manufacturing process. Furthermore, the accumulation of impurities at each isolation stage drastically reduces the overall yield, sometimes rendering the process commercially unviable for large-scale production. These inefficiencies create bottlenecks in the supply chain, leading to extended lead times and increased costs for the final active pharmaceutical ingredient.
The Novel Approach
The methodology outlined in CN1042914A represents a paradigm shift by utilizing a convergent strategy starting from readily available isatin compounds. This novel approach bypasses the need for isolating key intermediates, specifically compounds (VII), (II), (III), and (IV), allowing for a telescoped process that maximizes throughput. By reacting 5-hydroxy-5-(substituted phenyl)imidazolidine compounds with lower alkanols to form 5-alkoxy intermediates, and subsequently reacting these with alkylamines, the process achieves superior yields exceeding 70%. This streamlined workflow minimizes solvent usage and operational time, addressing the critical pain points of cost reduction in pharmaceutical intermediate manufacturing. The elimination of intermediate purification steps not only boosts yield but also simplifies the equipment requirements, making it highly suitable for commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Isatin-Based Cyclization
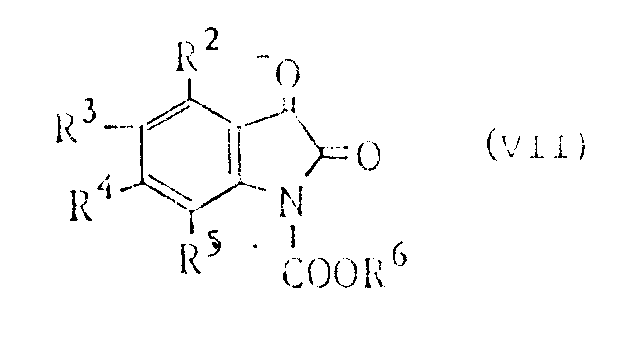
The reaction mechanism begins with the activation of the isatin core. Initially, an isatin compound (Formula V) reacts with a chloroformate (Formula VI) in the presence of a base like triethylamine to form an N-protected isatin derivative (Formula VII). This protection step is crucial for directing the subsequent nucleophilic attack by urea. The reaction with urea in a solvent such as tetrahydrofuran at temperatures between 50 to 120°C leads to the formation of the 5-hydroxy-5-(substituted phenyl)imidazolidine-2,4-dione (Formula II). This step constructs the imidazolidine ring, setting the stage for the spiro-formation. The hydroxyl group at the 5-position is then activated via etherification with a lower alkanol, such as methanol or ethanol, typically catalyzed by an acid like sulfuric acid or p-toluenesulfonic acid. This converts the poor leaving group (-OH) into a better leaving group (-OR7), facilitating the final cyclization.
The final transformation involves the reaction of the 5-alkoxyimidazolidine compound (Formula III) with a lower alkylamine or ammonia. This step triggers an intramolecular cyclization where the amine attacks the carbonyl carbon, displacing the alkoxy group and closing the quinazolinone ring to form the spiro system (Formula I). The patent highlights that this can be achieved in a one-pot manner or with minimal workup, as the intermediate (IV) can cyclize spontaneously upon further reaction with the amine. The use of aqueous amine solutions is particularly advantageous, as it eliminates the need for anhydrous conditions and expensive organic solvents in the final step. This mechanistic elegance ensures high purity and minimizes the formation of side products, which is essential for meeting the stringent purity specifications required by regulatory bodies for API intermediates.
How to Synthesize Spiro-Quinazolinone Derivatives Efficiently
To implement this synthesis effectively, operators must carefully control the stoichiometry of the amine and the temperature profiles during the cyclization phase. The process is robust enough to tolerate various substituents on the benzene ring, allowing for the generation of a diverse library of analogs without re-optimizing the entire protocol. The use of strong acid ion exchange resins, such as Amberlite IR-120, offers a greener alternative to liquid acids, simplifying the workup procedure and reducing corrosive waste. Detailed standard operating procedures regarding the addition rates and quenching methods are critical to maintaining safety and consistency. For a comprehensive breakdown of the specific operational parameters and safety guidelines, please refer to the technical guide below.
- React isatin compound or its salt with a chloroformate (e.g., ethyl chloroformate) in a solvent like THF with an amine base to form an N-protected isatin intermediate.
- Condense the N-protected isatin with urea in a solvent such as THF at elevated temperatures (50-120°C) to generate the 5-hydroxy-5-(substituted phenyl)imidazolidine compound.
- Convert the 5-hydroxy intermediate to a 5-alkoxyimidazolidine compound by reacting with a lower alkanol (e.g., methanol) in the presence of an acid catalyst.
- Perform intramolecular cyclization by reacting the 5-alkoxy compound with a lower alkylamine or ammonia to yield the final spiro-quinazolinone trione derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the adoption of this synthetic route offers substantial strategic benefits. The primary driver for cost optimization lies in the telescoping of the reaction sequence. By eliminating the isolation and purification of intermediates, manufacturers can significantly reduce the consumption of solvents, filtration media, and drying energy. This reduction in unit operations translates directly into lower manufacturing costs and a smaller environmental footprint. Furthermore, the starting materials, such as isatin and urea, are commodity chemicals with stable pricing and abundant global availability. This ensures supply chain reliability and mitigates the risk of raw material shortages that often plague more exotic synthetic routes. The ability to source these precursors reliably is a key factor in maintaining continuous production schedules.
- Cost Reduction in Manufacturing: The process described avoids the use of expensive transition metal catalysts or rare reagents, relying instead on common organic acids and amines. This fundamental shift in reagent selection drastically lowers the bill of materials. Additionally, the high yields reported (e.g., 91% in Example 1) mean that less raw material is wasted per kilogram of product produced. The elimination of intermediate purification steps also reduces labor costs and equipment occupancy time, allowing for higher throughput in existing facilities. These factors combine to deliver significant cost savings without compromising on the quality of the final intermediate.
- Enhanced Supply Chain Reliability: The robustness of the chemistry allows for flexible manufacturing. Since the reaction tolerates a range of temperatures (0 to 80°C) and solvents (methanol, ethanol, THF), production can be adapted to different plant configurations without major capital investment. The use of aqueous amine solutions further simplifies logistics, as these are safer and easier to transport than hazardous anhydrous reagents. This flexibility ensures that production can be scaled up or down rapidly in response to market demand, providing a buffer against supply chain disruptions. Partners can rely on consistent delivery timelines due to the predictable nature of the reaction kinetics.
- Scalability and Environmental Compliance: The process generates minimal hazardous waste, primarily consisting of aqueous salts and recoverable solvents. The avoidance of heavy metals simplifies the wastewater treatment process, ensuring compliance with increasingly strict environmental regulations. The high atom economy of the cyclization step means that the majority of the reactant mass ends up in the final product, reducing the volume of waste that requires disposal. This environmental efficiency is a critical selling point for pharmaceutical companies aiming to meet their sustainability goals. Scaling this process from pilot plant to multi-ton production is straightforward, leveraging standard stainless steel reactors and filtration equipment.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthesis. Understanding these details is crucial for R&D teams evaluating the feasibility of this route for their specific projects. The answers are derived directly from the experimental data and claims presented in the patent literature, ensuring accuracy and relevance. For further clarification on specific impurity profiles or custom analog synthesis, direct consultation with our technical team is recommended.
Q: What is the expected yield for the synthesis of spiro-quinazolinone derivatives using this method?
A: According to patent CN1042914A, the overall yield from the isatin starting material to the final compound is consistently higher than 70%, with specific examples demonstrating yields up to 91%.
Q: Does this process require isolation of intermediate compounds?
A: No, a key advantage of this novel method is that the desired compound can be prepared from the isatin compound without requiring the separation and purification of intermediate products such as compounds (VII), (II), (III), and (IV).
Q: What are the typical reaction conditions for the cyclization step?
A: The cyclization reaction with lower alkylamine or ammonia is typically conducted at mild temperatures ranging from 0 to 80°C, preferably between 10 to 60°C, often using aqueous solutions of the amine.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Spiro-Quinazolinone Derivative Supplier
At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and efficiency. Our facility is equipped with rigorous QC labs capable of verifying stringent purity specifications for complex heterocyclic intermediates like those described in CN1042914A. We understand that the transition from bench-scale discovery to commercial manufacturing requires a partner who can navigate regulatory hurdles and supply chain complexities seamlessly. Our commitment to quality and continuity makes us the preferred choice for global pharmaceutical innovators seeking a dependable source for critical building blocks.
We invite you to contact our technical procurement team to discuss your specific requirements. We are prepared to provide a Customized Cost-Saving Analysis tailored to your volume needs, demonstrating how our optimized processes can enhance your bottom line. Please reach out to request specific COA data and route feasibility assessments for your next project. Let us collaborate to accelerate the development of life-saving therapies for diabetic complications and beyond.
