Advanced Asymmetric Synthesis of (S)-Rivastigmine for Commercial Scale-Up
The pharmaceutical industry's relentless pursuit of effective treatments for neurodegenerative disorders has placed (S)-rivastigmine, a potent acetylcholinesterase inhibitor, at the forefront of therapeutic development. As detailed in the seminal patent CN103319374A, a breakthrough asymmetric synthetic method has been established that fundamentally alters the production landscape for this critical neurological agent. This innovative protocol utilizes m-hydroxybenzaldehyde as a readily available starting material and employs R-t-butyl sulfonamide as a chiral guiding reagent to construct the complex molecular architecture with high stereochemical fidelity. The significance of this technical advancement cannot be overstated for global supply chains, as it transitions the manufacturing paradigm from inefficient racemic resolution to a direct, enantioselective construction. By leveraging the robust chemistry of sulfinamide imines and Grignard additions, this process delivers a streamlined pathway that addresses the longstanding challenges of yield optimization and environmental safety in chiral drug synthesis.
For procurement specialists and R&D directors seeking a reliable pharmaceutical intermediates supplier, understanding the mechanistic superiority of this route is essential for long-term strategic planning. The patent explicitly outlines a sequence that bypasses the inherent 50% material loss associated with traditional resolution techniques, thereby offering a theoretically superior atom economy. Furthermore, the avoidance of hazardous demethylation agents such as boron tribromide, which are prevalent in older synthetic strategies, marks a substantial improvement in operational safety and equipment longevity. This report provides a deep-dive analysis of the reaction engineering, impurity control mechanisms, and commercial scalability inherent in this patented technology, positioning it as a cornerstone for cost reduction in pharmaceutical intermediate manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of (S)-rivastigmine has been plagued by significant inefficiencies inherent to racemic resolution strategies. Traditional methods often involve synthesizing the racemic mixture first and then attempting to separate the active (S)-enantiomer from the inactive (R)-isomer through crystallization or chromatographic techniques. This approach is fundamentally flawed from a resource utilization perspective because it inherently discards half of the synthesized material, leading to a maximum theoretical yield of only 50% even under ideal separation conditions. In practice, as noted in the background technology of the patent, the actual yields for these resolution routes often plummet to as low as 6.23% due to cumulative losses during multiple purification and recycling steps. Moreover, many existing asymmetric synthesis routes rely on harsh chemical environments; for instance, certain prior art methods necessitate the use of methyl sulfate for phenolic protection, a reagent known for its high toxicity and carcinogenic potential. Other pathways depend on boron tribromide (BBr3) for the critical demethylation step, a reagent that is extremely corrosive and requires specialized, expensive reactor linings and rigorous safety protocols to handle, thereby inflating capital expenditure and operational risks.
The Novel Approach
In stark contrast to these legacy methodologies, the novel approach disclosed in CN103319374A introduces a elegant and efficient asymmetric synthesis that circumvents the need for resolution entirely. By employing R-t-butyl sulfonamide, also known as Ellman's auxiliary, the process establishes the chiral center early in the synthesis through a highly stereoselective Grignard addition. This strategy ensures that the molecular framework is built with the correct stereochemistry from the outset, effectively doubling the potential yield compared to resolution methods. The patent data indicates an impressive overall yield of 21.7%, which represents a more than threefold increase over the comparative resolution baseline. Beyond yield improvements, this new route is characterized by its benign reaction conditions; it eliminates the need for highly toxic methylating agents and corrosive demethylation reagents. Instead, it utilizes mild mineral acids for hydrolysis and standard organic solvents like tetrahydrofuran and acetone, which are easily managed in standard stainless steel reactors. This shift not only simplifies the operational workflow but also drastically reduces the environmental burden associated with hazardous waste disposal, making it an ideal candidate for green chemistry initiatives in modern pharmaceutical manufacturing.
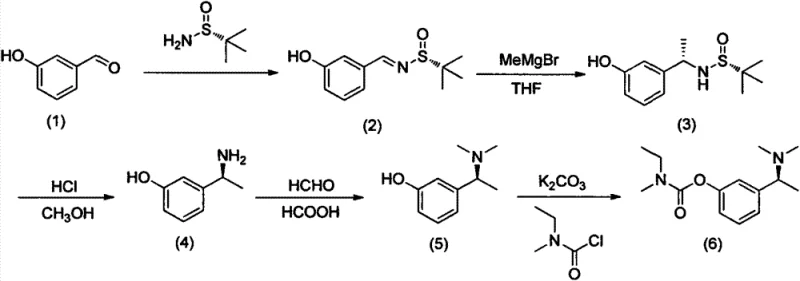
Mechanistic Insights into Ellman Auxiliary Mediated Asymmetric Induction
The core of this synthetic breakthrough lies in the sophisticated application of tert-butanesulfinamide chemistry to control stereochemistry during carbon-carbon bond formation. The process initiates with the condensation of m-hydroxybenzaldehyde and R-t-butyl sulfonamide to form a chiral N-sulfinyl imine (Compound 2). This imine formation is facilitated by dehydrating agents such as anhydrous cupric sulfate, which drive the equilibrium towards the product by sequestering water. The resulting sulfinyl imine possesses a rigid geometry where the bulky tert-butyl group and the oxygen atom of the sulfinyl moiety create a specific steric environment. When methylmagnesium bromide is introduced at cryogenic temperatures ranging from -60°C to -80°C, the magnesium cation coordinates with the sulfinyl oxygen, locking the imine into a specific conformation. This chelation control directs the nucleophilic attack of the methyl group to occur exclusively from the less hindered face of the imine double bond. This precise spatial arrangement ensures the formation of the (R)-N-((S)-1-(3-hydroxyphenyl)ethyl)-2-t-butyl sulfonamide (Compound 3) with high diastereoselectivity, effectively setting the critical chiral center required for the biological activity of the final drug substance.
Following the establishment of chirality, the removal of the auxiliary and subsequent functionalization are executed with equal precision to maintain optical purity. The hydrolysis step utilizes dilute mineral acids like hydrochloric acid in methanol to cleave the sulfinyl group, releasing the free chiral amine (Compound 4) without racemization. The subsequent Eschweiler-Clarke reaction serves as a robust method for N-methylation, converting the primary amine into the requisite dimethylamine (Compound 5). This reaction proceeds via the formation of an iminium ion intermediate using formaldehyde and formic acid, where formic acid acts as both the solvent and the hydride source for the reduction. The final esterification step couples this chiral amine with N-ethyl-N-methylcarbamoyl chloride in the presence of a base like anhydrous potassium carbonate. Throughout this multi-step sequence, the careful control of pH during workups—specifically adjusting to pH 8-11 with ammoniacal liquor—ensures that the basic amine intermediates are efficiently extracted into the organic phase while acidic impurities remain in the aqueous layer. This rigorous control over reaction parameters and workup conditions is paramount for achieving the high purity specifications demanded by regulatory bodies for neurological APIs.
How to Synthesize (S)-Rivastigmine Efficiently
The implementation of this asymmetric synthesis route requires strict adherence to the optimized reaction conditions outlined in the patent to ensure reproducibility and high yield. The process is divided into four distinct operational stages: chiral imine formation, stereoselective Grignard addition, auxiliary removal and methylation, and final carbamoylation. Each stage demands specific attention to stoichiometry, temperature control, and solvent quality. For instance, the use of freshly distilled tetrahydrofuran in the Grignard step is critical to prevent side reactions caused by moisture, while the maintenance of temperatures below -60°C is essential for maximizing diastereomeric excess. The following guide summarizes the critical operational parameters derived from the preferred embodiments of the patent, providing a roadmap for process chemists to replicate this high-efficiency pathway in a pilot or production setting.
- Condense m-hydroxybenzaldehyde with R-t-butyl sulfonamide using anhydrous cupric sulfate as a dehydrating agent to form the chiral imine intermediate.
- Perform a stereoselective Grignard addition using methylmagnesium bromide at low temperatures (-60 to -80°C) to establish the chiral center.
- Execute acidic hydrolysis to remove the sulfinyl group, followed by Eschweiler-Clarke methylation using formaldehyde and formic acid.
- Finalize the synthesis by esterifying the dimethylamine intermediate with N-ethyl-N-methylcarbamoyl chloride in the presence of potassium carbonate.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this asymmetric synthesis methodology offers profound advantages for supply chain stability and cost management. The most immediate impact is observed in raw material efficiency; by eliminating the 50% theoretical loss inherent in resolution processes, manufacturers can significantly reduce the volume of starting materials required per kilogram of final product. This improvement in atom economy translates directly into lower raw material costs and reduced logistics burdens associated with transporting and storing excess chemicals. Furthermore, the simplified reaction sequence, which avoids complex protection and deprotection cycles involving hazardous reagents, shortens the overall production cycle time. This acceleration allows for faster turnaround times on purchase orders and enhances the responsiveness of the supply chain to fluctuating market demands for Alzheimer's treatments. The use of common, non-proprietary reagents also mitigates the risk of supply disruptions caused by the scarcity of specialized catalysts or chiral ligands often required in other asymmetric methods.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous reagents such as boron tribromide and methyl sulfate removes the need for specialized corrosion-resistant equipment and extensive safety containment systems. This reduction in capital intensity allows for production in standard glass-lined or stainless steel reactors, significantly lowering depreciation costs. Additionally, the higher overall yield means that fixed costs related to labor, energy, and facility usage are amortized over a larger output of active pharmaceutical ingredient, driving down the unit cost of production substantially without compromising quality.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals like m-hydroxybenzaldehyde and methylmagnesium bromide ensures a robust and diversified supply base. Unlike processes dependent on single-source chiral catalysts or exotic reagents, this route can be scaled rapidly using materials available from multiple global vendors. This diversification minimizes the risk of production stoppages due to vendor-specific shortages. Moreover, the mild reaction conditions reduce the likelihood of batch failures caused by thermal runaways or equipment corrosion, ensuring consistent delivery schedules and reliable inventory levels for downstream formulation partners.
- Scalability and Environmental Compliance: The process is designed with industrial scalability in mind, utilizing solvents and conditions that are easily managed on a multi-ton scale. The absence of heavy metal catalysts simplifies the purification process and reduces the burden of residual metal testing, a critical checkpoint in API release. Environmentally, the reduction in hazardous waste generation aligns with increasingly stringent global environmental regulations. The ability to operate with lower toxicity profiles reduces waste disposal costs and facilitates easier permitting for manufacturing facilities, ensuring long-term operational continuity in a regulated environment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric synthesis route. These answers are derived directly from the experimental data and comparative analysis provided in patent CN103319374A, offering clarity on yield expectations, safety profiles, and scalability factors. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this process for commercial manufacturing.
Q: How does this asymmetric method compare to traditional resolution methods in terms of yield?
A: According to patent CN103319374A, the asymmetric synthesis route achieves an overall yield of 21.7%, which is significantly higher than the 6.23% yield typically associated with racemic resolution methods that inherently waste 50% of the material.
Q: Does this process utilize highly corrosive reagents like boron tribromide?
A: No, unlike prior art methods that require BBr3 for demethylation, this novel route avoids highly toxic and corrosive reagents, utilizing mild hydrolysis conditions with mineral acids like HCl instead.
Q: What is the role of R-t-butyl sulfonamide in this synthesis?
A: R-t-butyl sulfonamide acts as a chiral auxiliary (Ellman auxiliary) that directs the stereochemistry during the Grignard addition step, ensuring the formation of the desired (S)-configuration with high optical purity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S)-Rivastigmine Supplier
The technical sophistication of the asymmetric synthesis route described in CN103319374A underscores the complexity involved in producing high-purity neurological intermediates. At NINGBO INNO PHARMCHEM, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring such advanced chemistry to the marketplace. Our state-of-the-art facilities are equipped to handle the cryogenic conditions and sensitive organometallic reactions inherent in this process, ensuring that every batch meets stringent purity specifications. With our rigorous QC labs and commitment to process optimization, we guarantee a supply of (S)-rivastigmine intermediates that adhere to the highest international standards, providing our partners with the confidence needed to advance their drug development pipelines.
We invite global pharmaceutical partners to collaborate with us to leverage this efficient synthetic technology for their supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to evaluate how our advanced manufacturing capabilities can enhance your project's economic and operational performance.
