Advanced Silylation Strategy for Scalable Plazomicin Manufacturing and Supply Chain Optimization
Introduction to the Novel Plazomicin Preparation Technology
The pharmaceutical landscape for treating multidrug-resistant Gram-negative bacterial infections has been significantly advanced by the development of next-generation aminoglycosides, specifically Plazomicin. As detailed in patent CN112079882B, a groundbreaking preparation method has been established that addresses the longstanding challenges of low yield and complex processing associated with traditional synthesis routes. This innovative approach utilizes Sisomicin as the foundational starting material, implementing a sophisticated protection strategy involving HONB-PNZ for the allylic amino group, followed by a comprehensive silylation process. The technical breakthrough lies in the strategic application of hexamethyldisilazane to create a fully protected silanized intermediate, which streamlines subsequent acylation and condensation reactions. By integrating selective deprotection steps and a final purification via resin column chromatography, this method offers a robust pathway for obtaining high-purity Plazomicin suitable for clinical applications.
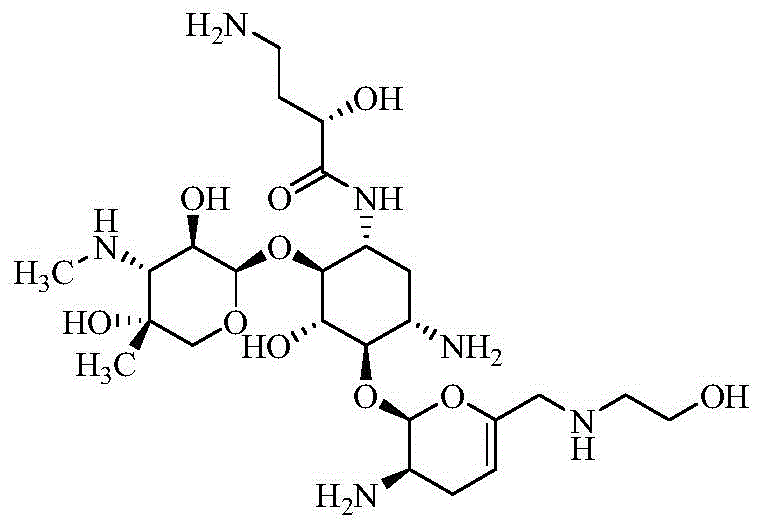
The significance of this technological advancement cannot be overstated for stakeholders in the antibiotic supply chain. Plazomicin represents a critical therapeutic option for complex urinary tract infections, and ensuring a reliable, cost-effective supply is paramount for global health security. The disclosed method not only shortens the overall reaction sequence but also operates under milder conditions compared to prior art, thereby reducing energy consumption and operational risks. For R&D directors and process chemists, this patent provides a clear blueprint for optimizing the manufacturing of this vital API intermediate, promising enhanced efficiency and reduced environmental impact through simplified post-treatment protocols.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the synthesis of Plazomicin was plagued by inefficiencies inherent in earlier patented routes, such as those described in US patent No. 08383596 and WO2010132839. The conventional methodologies often relied on a cumbersome array of protecting groups, including Fmoc, Boc, and various benzyl derivatives, which necessitated multiple introduction and removal steps. This "protecting group juggling" not only extended the synthesis timeline but also introduced significant opportunities for yield loss at each stage due to incomplete reactions or side products. Furthermore, the selectivity issues in these older routes often resulted in complex mixtures that were difficult to purify, requiring extensive chromatographic separations that are costly and hard to scale. The reliance on harsh conditions for certain deprotection steps also posed safety and environmental concerns, making these processes less attractive for modern, green chemistry-compliant manufacturing facilities.
The Novel Approach
In stark contrast, the novel approach outlined in CN112079882B introduces a paradigm shift by employing a global silylation strategy that dramatically simplifies the molecular architecture management. Instead of differentially protecting individual hydroxyl and amino groups with disparate reagents, the new method utilizes hexamethyldisilazane and trimethylchlorosilane to simultaneously protect all active sites on the Sisomicin scaffold. This unified protection scheme eliminates the need for multiple distinct protection steps, thereby collapsing the synthetic timeline. The subsequent selective acylation and deprotection sequences are designed to leverage the intrinsic reactivity differences of the functional groups, ensuring high regioselectivity without the need for excessive reagent usage. This streamlined logic results in a process that is not only shorter but also more robust, with fewer unit operations required to reach the final target molecule.
Mechanistic Insights into Silylation-Based Protection and Selective Acylation
The core mechanistic advantage of this synthesis lies in the efficiency of the silylation step and the subsequent controlled reactivity of the silanized intermediate. By converting all hydroxyl and amino groups into their corresponding trimethylsilyl (TMS) ethers and amines, the molecule becomes highly lipophilic and soluble in organic solvents, facilitating homogeneous reaction conditions that are difficult to achieve with the polar, zwitterionic nature of the parent aminoglycoside. The use of trimethylchlorosilane as a catalyst alongside hexamethyldisilazane ensures rapid and quantitative conversion, creating a stable platform for the critical acylation step. When the active ester (Compound 10) is introduced, the steric and electronic environment created by the silyl groups directs the acylation primarily to the desired amino position, minimizing the formation of regio-isomers that typically plague aminoglycoside modifications.
Furthermore, the mechanism for impurity control is elegantly integrated into the final purification stage. Although trace amounts of non-selective acylation products may form, the process accounts for this by utilizing a CD180 resin column for the final isolation. The mechanism here relies on the subtle differences in polarity and hydrogen bonding capability between the fully deprotected target Plazomicin and its structural analogues. By employing a gradient elution with low-concentration ammonia water, the resin effectively retains impurities while allowing the target compound to elute with high purity. This chromatographic resolution is far superior to traditional crystallization methods for such polar molecules, ensuring that the final API meets stringent pharmacopeial standards without the need for repetitive recrystallizations that degrade overall yield.
How to Synthesize Plazomicin Efficiently
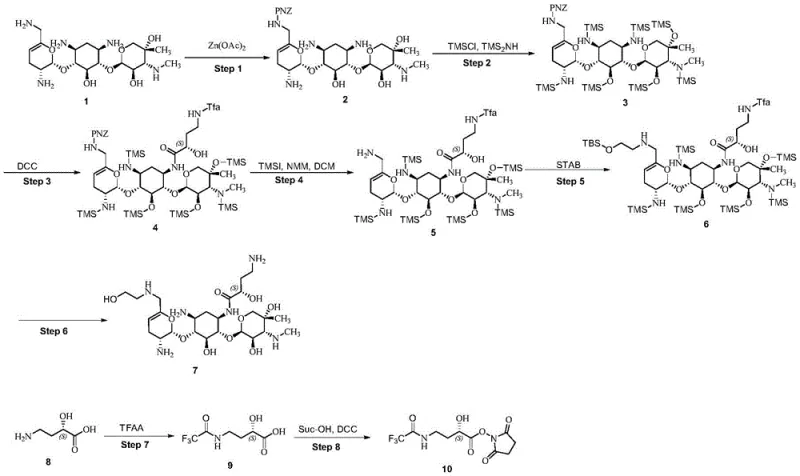
The synthesis of Plazomicin via this optimized route involves a logical sequence of protection, functionalization, and deprotection steps that can be readily adapted for pilot and commercial scale operations. The process begins with the chelation-mediated protection of the allylic amino group, followed by the global silylation that defines the efficiency of this route. Subsequent steps involve the coupling of the side chain via active ester acylation and reductive amination with a protected glycolaldehyde derivative. For a detailed breakdown of the specific reaction conditions, stoichiometry, and workup procedures required to execute this synthesis successfully, please refer to the standardized guide below.
- Protect the allylic amino group of Sisomicin using HONB-PNZ mediated by zinc acetate chelation.
- Perform global silylation using hexamethyldisilazane and trimethylchlorosilane to protect all hydroxyl and amino groups.
- Conduct selective acylation with active ester, followed by PNZ deprotection, condensation with protected glycolaldehyde, and final resin column purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route offers tangible benefits that extend beyond mere technical elegance. The reduction in the number of synthetic steps directly correlates to a decrease in raw material consumption, solvent usage, and labor hours, all of which are primary drivers of manufacturing costs. By eliminating the need for multiple distinct protecting group reagents and their corresponding removal agents, the bill of materials is simplified, reducing the complexity of inventory management and the risk of supply disruptions for niche chemicals. Additionally, the milder reaction conditions reduce the energy load on the production facility, contributing to lower utility costs and a smaller carbon footprint, which is increasingly important for meeting corporate sustainability goals.
- Cost Reduction in Manufacturing: The streamlined process significantly lowers the cost of goods sold (COGS) by minimizing the number of isolation and purification stages. Traditional routes often require intermediate purifications after every protection or deprotection step, which consumes vast amounts of silica gel, solvents, and time. In this new method, several intermediates are carried forward without purification (telescoped), which drastically reduces waste generation and processing time. The elimination of expensive and sensitive protecting group reagents like Fmoc-chloride in favor of commodity silylating agents further drives down material costs, making the final API more economically viable for broad market distribution.
- Enhanced Supply Chain Reliability: The robustness of the silylation chemistry ensures consistent batch-to-batch quality, which is critical for maintaining a reliable supply of this essential antibiotic. The use of stable intermediates and standard reagents means that the supply chain is less vulnerable to the volatility often seen with specialized fine chemicals. Moreover, the final purification via resin column is a highly scalable technique that can be easily expanded from laboratory to multi-ton production without the engineering bottlenecks associated with complex crystallization processes. This scalability ensures that manufacturers can respond rapidly to surges in demand without compromising on product quality or delivery timelines.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, this route offers a cleaner profile that simplifies waste treatment and compliance reporting. The reduction in step count inherently reduces the total volume of chemical waste generated per kilogram of product. The avoidance of heavy metal catalysts or toxic reagents in the key transformation steps aligns with modern green chemistry principles, facilitating easier regulatory approval and reducing the burden on wastewater treatment facilities. The ability to operate under milder temperatures and pressures also enhances the safety profile of the plant, lowering insurance premiums and operational risks associated with hazardous chemical processing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Plazomicin synthesis technology. These insights are derived directly from the patent specifications and are intended to clarify the operational advantages and mechanistic details for potential partners and licensees. Understanding these nuances is essential for evaluating the feasibility of technology transfer and the potential return on investment for scaling this process.
Q: How does the new silylation method improve upon previous Plazomicin synthesis routes?
A: The new method replaces complex multi-step protecting group strategies (like Fmoc/Boc juggling) with a efficient global silylation approach. This significantly reduces reaction steps, simplifies post-treatment, and improves overall yield from approximately 3.8% to over 5%.
Q: What is the role of the CD180 resin column in this process?
A: The CD180 resin column is utilized for the final purification step. It leverages the difference in binding capacity between the target product and trace impurities (formed from non-selective acylation), allowing for effective separation via gradient desorption with low-concentration ammonia water.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the route is designed for industrial scalability. It utilizes mild reaction conditions, avoids harsh reagents where possible, and employs robust purification techniques like resin chromatography that are amenable to large-scale manufacturing environments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Plazomicin Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes for life-saving antibiotics like Plazomicin. Our team of expert process chemists has extensively analyzed the technological merits of the silylation-based approach described in CN112079882B and is fully equipped to translate this laboratory innovation into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot plant to full-scale manufacturing is seamless and compliant with cGMP standards. Our state-of-the-art facilities are designed to handle complex aminoglycoside chemistry, featuring rigorous QC labs capable of verifying stringent purity specifications and comprehensive impurity profiling to guarantee product safety.
We invite pharmaceutical companies and contract manufacturing organizations to collaborate with us to optimize their Plazomicin supply chain. By leveraging our expertise in process intensification and cost-effective manufacturing, we can help you achieve significant operational efficiencies. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities can support your long-term strategic goals in the antibiotic market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
