Scalable One-Pot Synthesis of Isooctylamine: Technical Breakthroughs and Commercial Viability
The chemical industry is constantly seeking more efficient pathways to produce high-volume intermediates, and the recent disclosure in patent CN114105780A presents a compelling solution for the synthesis of isooctylamine and its derivatives. This technology leverages a sophisticated one-pot methodology that transforms readily available n-butyraldehyde into valuable amine products through a seamless combination of aldol condensation and direct reductive amination. Unlike traditional multi-step processes that require intermediate isolation and purification, this approach utilizes inexpensive skeletal nickel or copper catalysts to drive the reaction forward under relatively mild conditions. The strategic implementation of a graduated temperature profile during the condensation phase ensures high selectivity towards the desired octenal intermediate, effectively suppressing the formation of polyacetals and other disproportionation byproducts that often plague high-temperature aldehyde reactions. For R&D directors and process engineers, this represents a significant opportunity to streamline manufacturing workflows while maintaining rigorous purity standards essential for downstream applications in surfactants and pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of branched amines like isooctylamine has relied on methodologies that are either economically prohibitive or operationally complex for large-scale deployment. Conventional routes often employ expensive precious metal catalysts, such as rhodium complexes, which, while effective for asymmetric synthesis, impose a heavy financial burden on the production of commodity chemicals where margin compression is a constant threat. Furthermore, alternative pathways involving the dehydration of alcohols with ammonia typically necessitate extreme reaction conditions, including temperatures exceeding 200°C and high pressures, which escalate energy consumption and equipment maintenance costs. Another significant drawback in prior art is the reliance on multi-step sequences where the unsaturated aldehyde intermediate must be isolated before amination, leading to substantial material loss and increased solvent waste. These inefficiencies create bottlenecks in the supply chain, making it difficult to respond agilely to market fluctuations in demand for fine chemical intermediates used in agrochemicals and personal care formulations.
The Novel Approach
The innovative protocol described in the patent data fundamentally restructures the synthesis landscape by integrating condensation and amination into a single reactor vessel, thereby eliminating the need for intermediate workup procedures. By utilizing alkaline skeletal nickel or copper, the process capitalizes on the high surface area and hydrogenation activity of these base metals to catalyze both the initial carbon-carbon bond formation and the subsequent nitrogen incorporation.  This specific reaction pathway allows for the direct conversion of n-butyraldehyde into octenal through a carefully managed thermal ramp, starting at ambient temperatures and gradually increasing to optimize yield while minimizing side reactions. The ability to proceed directly to the amination step without extracting the octenal not only simplifies the operational workflow but also enhances the overall atom economy of the process. This telescoped approach significantly reduces the physical footprint required for production and lowers the barrier to entry for manufacturers seeking to produce high-purity isooctylamine derivatives competitively.
This specific reaction pathway allows for the direct conversion of n-butyraldehyde into octenal through a carefully managed thermal ramp, starting at ambient temperatures and gradually increasing to optimize yield while minimizing side reactions. The ability to proceed directly to the amination step without extracting the octenal not only simplifies the operational workflow but also enhances the overall atom economy of the process. This telescoped approach significantly reduces the physical footprint required for production and lowers the barrier to entry for manufacturers seeking to produce high-purity isooctylamine derivatives competitively.
Mechanistic Insights into Skeletal Metal-Catalyzed Reductive Amination
The core of this technological advancement lies in the dual functionality of the skeletal metal catalyst, which facilitates distinct mechanistic phases within the same reaction medium. In the initial stage, the catalyst promotes the aldol condensation of two n-butyraldehyde molecules, activating the alpha-position of one molecule and the beta-position of another to form the carbon-carbon double bond of octenal with the elimination of water. 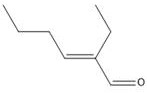 Following condensation, the system transitions to a high-pressure hydrogen environment where liquid ammonia or dimethylamine is introduced. The mechanism involves the nucleophilic attack of ammonia on the carbonyl carbon of the octenal, forming an unstable hemiaminal that rapidly dehydrates to generate an imine or enamine intermediate. This nitrogen-containing species is highly reactive and serves as the precursor for the final amine product. The presence of excess hydrogen and the active metal surface ensures that the C=N double bond is rapidly reduced to a C-N single bond, while any remaining C=C double bonds in the hydrocarbon chain are simultaneously hydrogenated to ensure saturation. This concerted reduction mechanism prevents the accumulation of reactive intermediates that could lead to polymerization or tar formation, thereby securing a cleaner reaction profile.
Following condensation, the system transitions to a high-pressure hydrogen environment where liquid ammonia or dimethylamine is introduced. The mechanism involves the nucleophilic attack of ammonia on the carbonyl carbon of the octenal, forming an unstable hemiaminal that rapidly dehydrates to generate an imine or enamine intermediate. This nitrogen-containing species is highly reactive and serves as the precursor for the final amine product. The presence of excess hydrogen and the active metal surface ensures that the C=N double bond is rapidly reduced to a C-N single bond, while any remaining C=C double bonds in the hydrocarbon chain are simultaneously hydrogenated to ensure saturation. This concerted reduction mechanism prevents the accumulation of reactive intermediates that could lead to polymerization or tar formation, thereby securing a cleaner reaction profile.
Impurity control is meticulously managed through the specific temperature programming and pressure parameters defined in the patent specifications. By initiating the condensation at temperatures below 25°C and slowly ramping to 90°C, the process kinetically favors the formation of the desired aldol product over thermodynamic byproducts like polyacetals which typically dominate at higher uncontrolled temperatures. Furthermore, the maintenance of a 3.0 MPa hydrogen pressure during the amination phase drives the equilibrium towards the fully saturated amine, effectively suppressing the formation of secondary condensation products or nitriles that might arise under hydrogen-starved conditions. 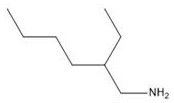 The use of skeletal nickel also provides a robust filtration profile, allowing for the efficient recovery and potential recycling of the catalyst, which is crucial for maintaining consistent batch-to-batch quality. For quality assurance teams, understanding these mechanistic nuances is vital for establishing critical process parameters (CPPs) that guarantee the stringent purity specifications required for pharmaceutical and electronic grade applications.
The use of skeletal nickel also provides a robust filtration profile, allowing for the efficient recovery and potential recycling of the catalyst, which is crucial for maintaining consistent batch-to-batch quality. For quality assurance teams, understanding these mechanistic nuances is vital for establishing critical process parameters (CPPs) that guarantee the stringent purity specifications required for pharmaceutical and electronic grade applications.
How to Synthesize Isooctylamine Efficiently
The practical execution of this synthesis route requires precise adherence to the established protocol to maximize yield and safety. The process begins with the charging of n-butyraldehyde and the skeletal metal catalyst into a high-pressure autoclave, followed by inert gas purging to eliminate oxygen and prevent premature oxidation or safety hazards. The detailed standardized synthesis steps, including specific stirring rates, pressure thresholds, and distillation parameters, are outlined in the technical guide below to ensure reproducibility across different manufacturing scales.
- Load n-butyraldehyde and skeletal nickel/copper catalyst into a high-pressure reactor under nitrogen protection.
- Execute a controlled temperature ramp (25°C to 90°C) to facilitate aldol condensation into octenal while minimizing byproducts.
- Introduce liquid ammonia and hydrogen gas (3.0 MPa) to perform direct reductive amination and hydrogenation in the same vessel.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this one-pot synthesis method offers transformative benefits that directly address the pain points of procurement managers and supply chain directors. The substitution of costly noble metal catalysts with abundant skeletal nickel or copper results in a drastic reduction in raw material expenditure, allowing for more competitive pricing structures in the final amine products. Additionally, the telescoping of two major reaction steps into a single vessel significantly reduces utility consumption, solvent requirements, and labor hours associated with intermediate handling and purification. This operational simplification translates into a more resilient supply chain capable of sustaining continuous production runs with minimal downtime for cleaning or changeover. For organizations focused on sustainability, the generation of water as the primary byproduct aligns with green chemistry principles, reducing the environmental burden associated with waste disposal and regulatory compliance.
- Cost Reduction in Manufacturing: The economic viability of this process is heavily anchored in the elimination of expensive rhodium-based catalysts and the reduction of unit operations. By avoiding the isolation of the octenal intermediate, manufacturers save substantially on solvent costs, energy for distillation, and equipment depreciation associated with additional reactors. The use of skeletal metals, which are known for their durability and ease of recovery, further lowers the long-term operational expenditure. This cost structure enables producers to offer high-purity fine chemical intermediates at a price point that is attractive for high-volume applications in the surfactant and agrochemical sectors without sacrificing margin.
- Enhanced Supply Chain Reliability: The reliance on n-butyraldehyde, a widely produced commodity chemical, ensures a stable and secure feedstock supply that is less susceptible to the geopolitical volatility often seen with precious metals. The robustness of the skeletal nickel catalyst under the described reaction conditions implies a longer catalyst lifecycle and reduced frequency of replenishment orders. This stability allows supply chain planners to forecast production capacities with greater accuracy and maintain consistent inventory levels to meet the just-in-time delivery demands of downstream pharmaceutical and industrial clients. The simplified process flow also reduces the risk of production delays caused by equipment failures in complex multi-step setups.
- Scalability and Environmental Compliance: The mild reaction conditions and the absence of hazardous reagents make this process inherently safer and easier to scale from pilot plant to commercial tonnage. The one-pot design minimizes the release of volatile organic compounds (VOCs) by reducing the number of transfer and venting operations, facilitating compliance with increasingly strict environmental regulations. The efficient catalyst recovery system further supports circular economy initiatives by minimizing solid waste generation. For facilities aiming to expand their capacity for amine production, this technology offers a scalable pathway that requires less capital investment in infrastructure compared to traditional multi-reactor configurations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the patent specifications and are intended to clarify the operational capabilities and limitations of the one-pot method for potential partners and licensees. Understanding these details is crucial for evaluating the feasibility of integrating this process into existing manufacturing portfolios.
Q: What are the primary advantages of using skeletal nickel over rhodium catalysts?
A: Skeletal nickel offers a drastically lower cost profile compared to precious metal rhodium catalysts, significantly reducing raw material expenses without compromising catalytic activity for this specific transformation.
Q: How does the one-pot method improve process safety and efficiency?
A: By eliminating the isolation of the unstable octenal intermediate, the process reduces handling risks, minimizes solvent usage, and shortens the overall production cycle time.
Q: Can this method produce derivatives other than primary amines?
A: Yes, by substituting liquid ammonia with secondary amines like dimethylamine, the process can be adapted to synthesize tertiary amine derivatives such as N,N-dimethyloctylamine.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Isooctylamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced synthesis technologies like the one described in CN114105780A for producing high-quality isooctylamine and its derivatives. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of critical intermediates. Our state-of-the-art facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch meets the exacting standards required for pharmaceutical and specialty chemical applications. We are committed to leveraging our technical expertise to optimize these novel routes for maximum efficiency and cost-effectiveness.
We invite you to collaborate with us to explore how this innovative one-pot synthesis can enhance your product portfolio and reduce your overall manufacturing costs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to support your R&D and supply chain planning efforts, ensuring a seamless transition to this superior production method.
