Advanced Synthesis of High-Purity Leflunomide Impurity for Global Pharmaceutical Quality Control
Introduction to Patent CN101817797B and Strategic Importance
The pharmaceutical industry relies heavily on the rigorous quality control of active pharmaceutical ingredients (APIs), particularly for potent immunosuppressive agents like Leflunomide used in treating rheumatoid arthritis. As detailed in the Chinese patent CN101817797B, the precise synthesis and control of specific impurities are critical for ensuring patient safety and regulatory compliance. This patent introduces a breakthrough methodology for synthesizing 3-methyl-N-[4-(trifluoromethyl)phenyl]-4-isoxazole carboxamide, a key structural isomer and potential impurity of Leflunomide. The ability to produce this compound in high purity is not merely an academic exercise but a commercial necessity for generating accurate reference standards required by pharmacopoeias worldwide.
Regulatory bodies such as the State Food and Drug Administration mandate that the content of this specific 3-methyl isomer in Leflunomide raw materials must not exceed 0.1%. Consequently, pharmaceutical manufacturers require access to ultra-high purity samples of this impurity to validate their analytical methods and ensure their final products meet these strict limits. The technology disclosed in this patent addresses the historical difficulties in producing this reference standard, offering a pathway that significantly enhances both yield and purity compared to legacy methods. For R&D directors and procurement specialists, understanding this synthetic advancement is vital for securing a reliable supply chain of critical quality control materials.

The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
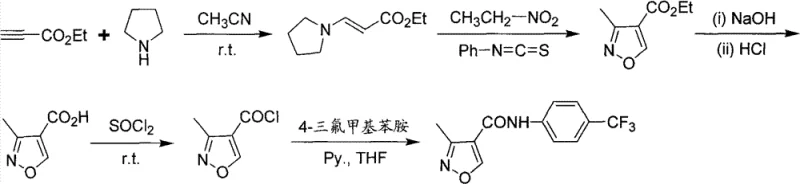
Prior to this innovation, the synthesis of this specific isoxazole carboxamide was fraught with significant chemical and operational challenges, primarily documented in patent WO9852944. Route A of the prior art utilized ethyl propiolate and tetrahydropyrrole, leading to a cycloaddition reaction that generated diphenylurea as a persistent byproduct. This impurity proved notoriously difficult to remove, often requiring extensive column chromatography which is impractical for large-scale manufacturing and drastically increases production costs. Furthermore, the multi-step nature of this route introduced additional opportunities for yield loss and contamination, making it unsuitable for the efficient production of high-purity reference standards needed for routine quality assurance testing.
Route B of the same prior art attempted to simplify the process but failed due to poor regioselectivity during the isoxazole ring formation. This lack of selectivity resulted in a molar yield of merely 8.36%, with the final product containing nearly equal amounts of the desired 3-methyl isomer and the unwanted Leflunomide isomer. Since these two compounds possess very similar polarities, separating them to the required purity levels became an arduous task, often impossible without sophisticated and expensive purification techniques. These limitations highlighted a clear gap in the market for a robust, scalable, and selective synthesis method that could reliably deliver the high-purity material demanded by regulatory standards.
The Novel Approach
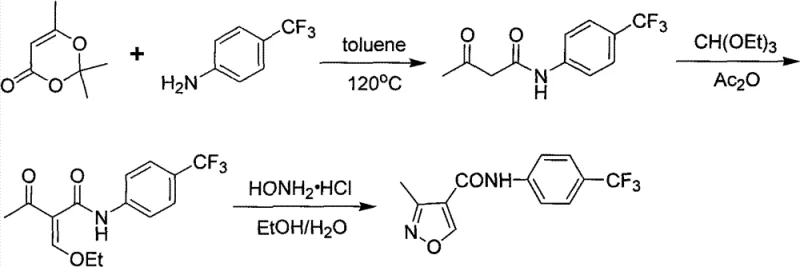
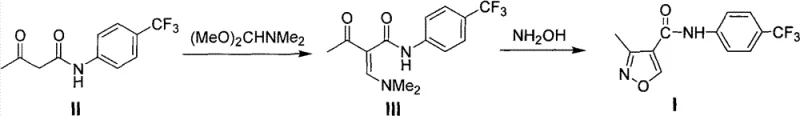
The methodology presented in CN101817797B fundamentally reengineers the synthetic pathway to overcome these regioselectivity and purification hurdles. Instead of relying on triethyl orthoformate and hydroxylamine hydrochloride, which contributed to the poor selectivity in Route B, this novel approach employs N,N-dimethylformamide dimethyl acetal (DMF-DMA) and free hydroxylamine aqueous solution. This strategic substitution allows for the formation of a specific enamine intermediate that cyclizes with high precision. By shifting the reaction conditions to a controlled pH environment, the process effectively suppresses the formation of the unwanted Leflunomide isomer, thereby solving the critical separation issue that plagued previous attempts. This represents a significant leap forward in process chemistry, transforming a low-yielding, difficult purification into a streamlined, high-efficiency operation.
Mechanistic Insights into pH-Controlled Regioselective Cyclization
The core of this technological advancement lies in the precise manipulation of reaction kinetics and thermodynamics during the cyclization step. The process begins with the condensation of N-[(4-trifluoromethyl)phenyl]acetoacetamide with DMF-DMA in a solvent like toluene. This step generates an enamine intermediate, N-[(4-trifluoromethyl)phenyl]-2-dimethylaminomethyleneacetoacetamide, which serves as a highly reactive precursor for ring closure. Unlike the rigid constraints of previous methods, this intermediate offers a flexible electronic environment that, when paired with the correct nucleophile, favors the formation of the 3-methyl isoxazole ring over the 5-methyl isomer. The use of free hydroxylamine, rather than its hydrochloride salt, eliminates the introduction of excess chloride ions and allows for finer control over the nucleophilic attack on the enamine system.
Crucially, the patent emphasizes the maintenance of the reaction pH between 5.0 and 7.0, optimally between 5.5 and 6.5, using dilute acids such as hydrochloric or sulfuric acid. This pH control is the mechanistic key to high regioselectivity; it ensures that the hydroxylamine remains sufficiently nucleophilic to attack the correct carbon center while preventing side reactions or the protonation states that lead to the wrong isomer. Conducting this reaction at low temperatures, specifically between -10°C and 20°C, further stabilizes the transition state favoring the desired product. This combination of reagent selection and environmental control results in a clean reaction profile where the target molecule precipitates or crystallizes out with minimal isomeric contamination, enabling purification via simple recrystallization rather than complex chromatography.
How to Synthesize 3-methyl-N-[4-(trifluoromethyl)phenyl]-4-isoxazole carboxamide Efficiently
The implementation of this synthesis protocol requires careful attention to solvent selection and temperature gradients to maximize the benefits of the new chemistry. The initial condensation step is best performed in toluene under reflux conditions to drive the equilibrium towards the enamine intermediate, followed by a controlled cooling phase to induce crystallization of the intermediate if necessary. The subsequent cyclization step demands rigorous monitoring of pH and temperature, utilizing methanol or ethanol as the reaction medium to ensure solubility of the reagents while facilitating the precipitation of the final product. Detailed standardized operating procedures for scaling this reaction from gram to kilogram quantities are essential for maintaining the high purity profiles observed in the laboratory examples.
- Condense N-[(4-trifluoromethyl)phenyl]acetoacetamide with N,N-dimethylformamide dimethyl acetal in toluene under reflux to form the enamine intermediate.
- React the intermediate with aqueous free hydroxylamine in methanol, strictly maintaining pH between 5.5 and 6.5 using dilute acid at low temperatures.
- Purify the final crude product via recrystallization from ethyl acetate to achieve high-purity reference standards suitable for HPLC analysis.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible benefits that extend beyond mere chemical elegance. The primary advantage is the drastic simplification of the purification process. By eliminating the need for column chromatography—a technique that is solvent-intensive, time-consuming, and difficult to scale—manufacturers can significantly reduce the operational expenditure associated with producing this reference standard. The ability to purify the final product through recrystallization from ethyl acetate implies a much lower consumption of silica gel and organic solvents, directly translating to reduced waste disposal costs and a smaller environmental footprint. This efficiency makes the commercial production of high-purity batches far more economically viable.
Furthermore, the improved regioselectivity directly impacts supply chain reliability by increasing the effective yield of the usable product. In previous methods, a significant portion of the reaction mass was lost to inseparable isomers or difficult byproducts, creating a bottleneck in supply. With the new method achieving high purity (demonstrated at 99.99% in examples) and improved yields, suppliers can fulfill orders for reference standards more consistently and with shorter lead times. This reliability is crucial for pharmaceutical companies that depend on these standards for batch release testing; any delay in obtaining the standard can halt the distribution of the final drug product. Therefore, this technology secures the supply chain against the volatility of low-yield chemical processes.
- Cost Reduction in Manufacturing: The elimination of column chromatography and the use of common, inexpensive solvents like toluene and methanol drastically lower the cost of goods sold. The process avoids expensive reagents and reduces the labor hours required for purification, allowing for substantial cost savings in the production of high-value reference materials without compromising on quality specifications.
- Enhanced Supply Chain Reliability: High regioselectivity means less raw material is wasted on unwanted isomers, ensuring a more predictable output volume. This consistency allows suppliers to maintain healthier inventory levels of critical impurity standards, reducing the risk of stockouts that could disrupt the quality control operations of downstream pharmaceutical clients.
- Scalability and Environmental Compliance: The reliance on recrystallization rather than chromatography makes this process inherently more scalable to industrial volumes. Additionally, the reduction in solvent usage and hazardous waste generation aligns with modern green chemistry principles, helping manufacturers meet increasingly stringent environmental regulations and sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of this specific isoxazole derivative. These answers are derived directly from the technical disclosures within the patent documentation, providing clarity on the process capabilities and limitations. Understanding these details helps stakeholders make informed decisions regarding the sourcing and utilization of this critical pharmaceutical intermediate.
Q: Why is the regioselectivity of this new method superior to previous routes?
A: The new method utilizes N,N-dimethylformamide dimethyl acetal and free hydroxylamine under controlled pH conditions, which directs the cyclization specifically to the 3-methyl isomer, avoiding the formation of the leflunomide isomer that plagues Route B.
Q: How does this process eliminate the need for column chromatography?
A: By avoiding the formation of difficult-to-remove byproducts like diphenylurea (found in Route A) and minimizing isomeric impurities, the final product can be purified simply through recrystallization, significantly reducing processing time and cost.
Q: What is the achieved purity level for this impurity standard?
A: The patent examples demonstrate that this synthetic route can consistently achieve product purity levels of 99.99% as analyzed by HPLC, meeting the stringent requirements for pharmaceutical reference standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-methyl-N-[4-(trifluoromethyl)phenyl]-4-isoxazole carboxamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity impurity standards play in the global pharmaceutical landscape. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated chemistry described in patent CN101817797B can be translated into reliable industrial supply. We adhere to stringent purity specifications and utilize rigorous QC labs to verify that every batch of 3-methyl-N-[4-(trifluoromethyl)phenyl]-4-isoxazole carboxamide meets the exacting standards required for HPLC calibration and method validation. Our commitment to quality ensures that your quality control processes remain uninterrupted and compliant with international regulations.
We invite pharmaceutical partners to leverage our expertise for their specific sourcing needs. By contacting our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, ensuring that your supply of this critical reference standard is secure, cost-effective, and of the highest possible quality.
