Revolutionizing Chiral Synthesis: Dual-Handed Control with Dehydroabietic Amine-Thiourea Catalysts
The landscape of asymmetric organocatalysis has been significantly advanced by the innovations detailed in Chinese Patent CN100584459C, which introduces a groundbreaking class of dehydroabietic amine-chiral thiourea catalysts. This technology addresses a critical bottleneck in the synthesis of chiral pharmaceutical intermediates, specifically gamma-nitro-aromatic ketones and gamma-nitro-aromatic heteroketones, by enabling high-efficiency dual-handed stereocontrol. Traditionally, accessing both enantiomers of a chiral drug intermediate requires entirely different catalyst scaffolds or complex resolution processes, but this invention utilizes a robust natural chiral source, dehydroabietic amine, to create a versatile catalytic system. The patent demonstrates that by simply switching the configuration of the cyclohexanediamine moiety attached to the thiourea core, manufacturers can selectively produce either the (S) or (R) enantiomer with exceptional optical purity exceeding 99% ee. This capability represents a paradigm shift for process chemists seeking reliable routes to complex chiral building blocks without the burden of developing multiple distinct catalytic systems.
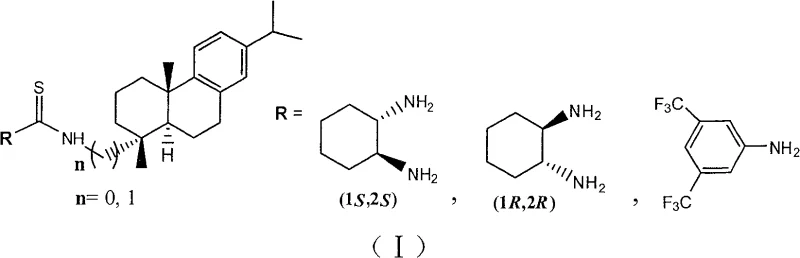
The limitations of conventional methods in asymmetric Michael additions have long plagued the fine chemical industry, particularly regarding the inflexibility of catalyst design. Prior art, such as the seminal work by Takemoto and subsequent modifications by Ma and others, often suffered from a significant drawback where the catalyst's efficiency was heavily dependent on the specific match between the catalyst backbone and the diamine configuration. For instance, previous sugar-ring derived thiourea catalysts exhibited excellent activity for producing (S)-gamma-nitro ketones when paired with (1R,2R)-cyclohexanediamine but showed drastically reduced yields and enantioselectivity when the (1S,2S)-diamine was employed to target the (R)-enantiomer. This asymmetry in catalytic performance forces manufacturers to source or synthesize two completely different catalyst families to access both enantiomers, thereby doubling the R&D overhead and complicating supply chain logistics. Furthermore, many existing protocols demand stringent anhydrous and oxygen-free conditions, necessitating expensive infrastructure and increasing the operational expenditure for large-scale production facilities.
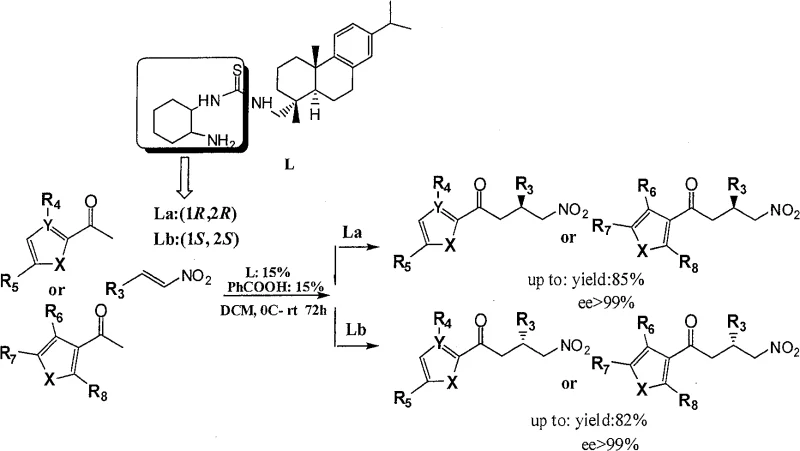
In stark contrast, the novel approach utilizing dehydroabietic amine-thiourea catalysts overcomes these historical inefficiencies through a uniquely adaptable molecular architecture. The invention successfully leverages the rigid, bulky structure of the dehydroabietic backbone to create a chiral environment that is highly tolerant to variations in the diamine component. Experimental data from the patent confirms that whether the catalyst incorporates (1R,2R)-cyclohexanediamine or (1S,2S)-cyclohexanediamine, the system consistently delivers high yields up to 99% and enantioselectivity greater than 99% for the respective target enantiomers. This "dual-handed" capability means that a single catalyst platform can service the production of both enantiomeric forms of a drug intermediate, streamlining inventory management and reducing the complexity of process validation. Additionally, the reaction proceeds smoothly in dichloromethane at temperatures ranging from 0°C to room temperature and is tolerant to air, removing the need for specialized glovebox techniques and allowing for simpler, more cost-effective reactor setups.
Mechanistic Insights into Bifunctional Thiourea Catalysis
The exceptional performance of the dehydroabietic amine-thiourea catalyst stems from its sophisticated bifunctional activation mechanism, which simultaneously engages both the nucleophile and the electrophile through a precise network of non-covalent interactions. In this catalytic cycle, the thiourea moiety acts as a double hydrogen bond donor, effectively activating the nitro group of the nitroalkene substrate by stabilizing the developing negative charge in the transition state. Concurrently, the tertiary amine or the proximal basic sites within the dehydroabietic framework facilitate the enolization of the ketone substrate, generating the reactive nucleophilic species. The bulky dehydroabietic skeleton plays a crucial steric role, shielding one face of the reacting species and thereby enforcing high facial selectivity during the carbon-carbon bond formation. This rigid chiral pocket ensures that the approach of the nucleophile is strictly controlled, leading to the observed high enantiomeric excess values. Unlike flexible linear catalysts that may adopt multiple conformations leading to background racemic reactions, the fused ring system of the dehydroabietic core locks the catalyst into a defined geometry that maximizes stereochemical induction.
Impurity control is another critical aspect where this mechanistic design offers substantial advantages for pharmaceutical manufacturing. The high specificity of the hydrogen-bonding activation minimizes side reactions such as polymerization of the nitroalkene or self-condensation of the ketone, which are common pitfalls in base-catalyzed Michael additions. By operating under mild conditions without strong bases or Lewis acids, the process avoids the formation of degradation byproducts that are difficult to remove downstream. The patent data indicates that the reaction cleanly produces the desired gamma-nitro carbonyl compounds with minimal formation of diastereomers or regioisomers. This high level of chemoselectivity simplifies the purification process, often requiring only standard silica gel chromatography to achieve analytical purity. For regulatory compliance in API synthesis, minimizing the impurity profile is paramount, and this organocatalytic route provides a clean reaction trajectory that aligns well with strict quality control standards required by global health authorities.
How to Synthesize Gamma-Nitro-Aromatic Ketones Efficiently
The practical implementation of this technology involves a straightforward protocol that balances reaction kinetics with stereochemical fidelity. The process begins by dissolving the nitroalkene substrate and the specific enantiomer of the dehydroabietic amine-thiourea catalyst (15mol%) along with a benzoic acid additive (15mol%) in dichloromethane. The inclusion of benzoic acid is critical for proton transfer steps that accelerate the catalytic turnover without compromising enantioselectivity. Following the dissolution, the aromatic ketone or heteroketone is added in a three-fold molar excess at 0°C to ensure controlled initiation of the reaction. The mixture is then allowed to warm to room temperature and stirred for an extended period, typically between 60 to 72 hours, to ensure complete conversion of the starting materials.
- Prepare the reaction mixture by dissolving the nitroalkene substrate and 15mol% of the dehydroabietic amine-chiral thiourea catalyst (either (1R,2R) or (1S,2S) configuration) along with 15mol% benzoic acid in dichloromethane.
- Cool the solution to 0°C and add the aromatic ketone or heteroketone compound in a 3-fold molar excess relative to the nitroalkene substrate to drive the equilibrium forward.
- Allow the reaction to proceed at 0°C for 2 hours, then warm to room temperature and continue stirring for 60 to 72 hours until completion, followed by purification via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this dehydroabietic amine-thiourea catalyst technology presents a compelling value proposition centered on cost efficiency and operational resilience. The elimination of transition metals from the synthetic route is a primary driver for cost reduction, as it removes the necessity for expensive metal scavengers and the associated validation testing for residual heavy metals in the final product. This metal-free characteristic not only lowers the direct material costs but also significantly reduces the environmental burden associated with metal waste disposal, aligning with increasingly stringent green chemistry regulations. Furthermore, the use of dehydroabietic amine, a derivative of rosin acid obtained from renewable pine tree sources, ensures a sustainable and potentially lower-cost feedstock compared to fully synthetic chiral ligands that rely on petrochemical precursors. The stability of the catalyst and its ability to function in air further reduces the capital expenditure required for specialized reactor equipment, making it accessible for both pilot-scale and commercial-scale manufacturing facilities.
- Cost Reduction in Manufacturing: The economic benefits of this process are multifaceted, starting with the avoidance of precious metal catalysts which are subject to volatile market pricing and supply constraints. By utilizing an organocatalytic system, manufacturers can bypass the complex and costly downstream processing steps required to reduce metal residues to ppm levels, which is a mandatory requirement for pharmaceutical ingredients. The high yields reported, often reaching up to 99%, mean that raw material utilization is maximized, reducing the cost per kilogram of the active intermediate. Additionally, the recyclability of the catalyst mentioned in the patent suggests that the effective catalyst loading could be even lower in a continuous flow or recycled batch process, further driving down the variable costs associated with reagent consumption. The mild reaction conditions also translate to lower energy costs, as there is no need for cryogenic cooling or high-temperature heating, allowing the reaction to proceed efficiently at ambient temperatures.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the use of readily available natural chiral sources and simple reagents that are not subject to the geopolitical risks often associated with rare earth metals or specialized synthetic ligands. The robustness of the catalyst, which tolerates air and moisture to a significant degree, reduces the risk of batch failures due to minor deviations in operational conditions, thereby improving overall production reliability. The ability to produce both (S) and (R) enantiomers using the same core catalyst scaffold simplifies the supplier qualification process, as vendors do not need to maintain separate inventory lines for opposing enantiomeric catalysts. This flexibility allows for rapid response to changes in market demand for specific drug isomers without the lead time penalties associated with sourcing new catalytic materials. The simplified logistics of handling stable, solid organocatalysts compared to sensitive metal complexes also reduces storage and transportation costs.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by the absence of hazardous reagents and the use of common organic solvents like dichloromethane, which are well-understood in industrial settings. The environmental profile of the process is superior to traditional methods, as it generates less hazardous waste and avoids the toxicity issues linked to heavy metal contamination. This aligns with the principles of green chemistry, potentially qualifying the manufacturing process for environmental incentives or preferred supplier status with eco-conscious pharmaceutical partners. The high atom economy of the Michael addition reaction, combined with the high selectivity of the catalyst, ensures that waste generation is minimized, reducing the costs associated with waste treatment and disposal. The potential for catalyst recovery and reuse further enhances the sustainability metrics of the process, making it an attractive option for companies aiming to reduce their carbon footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this catalytic technology in industrial settings. These answers are derived directly from the experimental data and claims presented in the patent documentation to ensure accuracy and relevance for technical decision-makers. Understanding these details is crucial for evaluating the feasibility of integrating this method into existing production workflows.
Q: What is the primary advantage of the dehydroabietic amine-thiourea catalyst over traditional thiourea catalysts?
A: Unlike traditional catalysts that often lose efficiency when switching diamine configurations, this catalyst maintains high activity and enantioselectivity (>99% ee) for both (S) and (R) product configurations using the same dehydroabietic backbone.
Q: Does this synthesis method require expensive transition metals or harsh conditions?
A: No, the process is completely metal-free, operates under mild conditions (0°C to room temperature), and tolerates air, eliminating the need for costly inert atmosphere setups and heavy metal removal steps.
Q: Can this catalyst be recycled for industrial scale-up?
A: Yes, the patent highlights that the catalyst is easy to separate from the product and can be recycled, which significantly enhances its economic viability for large-scale pharmaceutical manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Gamma-Nitro-Aromatic Ketones Supplier
The technological breakthroughs described in patent CN100584459C underscore the immense potential of dehydroabietic amine-thiourea catalysts in modernizing the synthesis of chiral pharmaceutical intermediates. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our commitment to quality is evidenced by our stringent purity specifications and rigorous QC labs, which guarantee that every batch of gamma-nitro-aromatic ketones meets the highest international standards for enantiomeric excess and chemical purity. We understand the critical nature of supply chain consistency for our partners and have optimized our manufacturing protocols to deliver these complex intermediates with reliable lead times and competitive pricing structures.
We invite potential partners to engage with our technical procurement team to discuss how this advanced organocatalytic technology can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how switching to this metal-free methodology can optimize your overall production budget. We encourage you to contact us to obtain specific COA data for our catalog compounds or to initiate route feasibility assessments for custom synthesis projects. Our team is ready to provide the technical support and commercial flexibility required to accelerate your drug development timeline while maintaining the highest standards of quality and compliance.
