Advancing Oncology Lead Discovery with Efficient Chiral 2,5-Dihydropyrrole Manufacturing
Advancing Oncology Lead Discovery with Efficient Chiral 2,5-Dihydropyrrole Manufacturing
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for more efficient, stereoselective, and cost-effective synthetic routes. A significant breakthrough in this domain is documented in Chinese Patent CN102351769A, which discloses a novel class of chiral 2,2-diethoxycarbonyl-4-acyl-5-aryl or alkyl substituted 2,5-dihydropyrroles. These compounds represent a potent bioactive molecular skeleton with demonstrated cytotoxic activity against MCF7 breast cancer cells, making them highly valuable for oncology drug discovery programs. The innovation lies not just in the molecule itself, but in the methodology: an asymmetric multicomponent reaction catalyzed by small organic molecules that constructs the chiral ring system in a single step from achiral precursors. This approach addresses critical pain points in modern medicinal chemistry, specifically the demand for high-purity pharmaceutical intermediates with defined stereochemistry without the prohibitive costs associated with traditional chiral resolution or chiral pool synthesis.
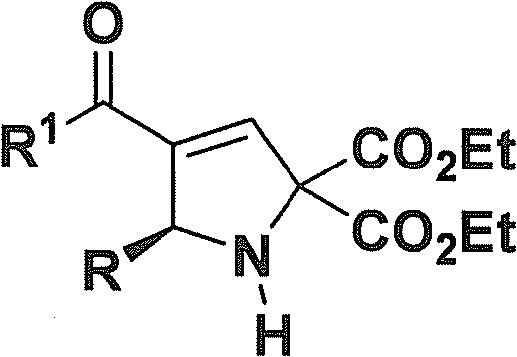
For R&D Directors and process chemists, the structural versatility offered by this patent is compelling. The general formula allows for extensive diversification at the R and R1 positions, accommodating a wide range of aryl, heteroaryl, and alkyl substituents. This flexibility is crucial for structure-activity relationship (SAR) studies, enabling rapid iteration of lead compounds. Furthermore, the reported optical purity is exceptional, with enantiomeric excess (ee) values frequently exceeding 99%. Achieving such high stereocontrol directly from the reaction mixture significantly reduces the burden on downstream purification processes, a factor that directly impacts the overall cost of goods (COGS) and timeline for clinical candidate selection.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral 2,5-dihydropyrrole derivatives has been fraught with inefficiencies that hinder scalable production. Traditional literature methods often rely on optically pure starting materials, such as vinyl epoxides or hydroxyamides, which are inherently expensive and limited in commercial availability. For instance, prior art describes routes involving aminolysis and ring-closing olefin metathesis or sequential Wittig-Horner reactions followed by intramolecular Michael additions. These multi-step sequences are not only operationally cumbersome but also suffer from cumulative yield losses at each stage. Moreover, the reliance on chiral starting materials restricts the structural diversity accessible to chemists, as the stereochemistry is locked in by the initial feedstock. This lack of flexibility makes it difficult to explore broad chemical space efficiently, slowing down the drug discovery pipeline and increasing the financial risk associated with developing new therapeutic agents.
The Novel Approach
In stark contrast, the methodology outlined in CN102351769A represents a paradigm shift towards atom-economical and step-efficient synthesis. By employing an asymmetric multicomponent reaction, the process combines a ynone, an aldehyde, and diethyl aminomalonate in a single vessel. This one-pot strategy eliminates the need for isolating unstable intermediates and drastically reduces solvent consumption and waste generation. The use of a chiral phosphoric acid catalyst allows for the dynamic kinetic resolution or asymmetric induction necessary to generate the chiral center de novo from achiral inputs. This capability is transformative for cost reduction in pharmaceutical intermediate manufacturing, as it replaces costly chiral building blocks with commodity chemicals. The reaction conditions are notably mild, typically proceeding in toluene at temperatures ranging from -10°C to 30°C, which simplifies equipment requirements and enhances safety profiles compared to cryogenic or high-pressure alternatives.
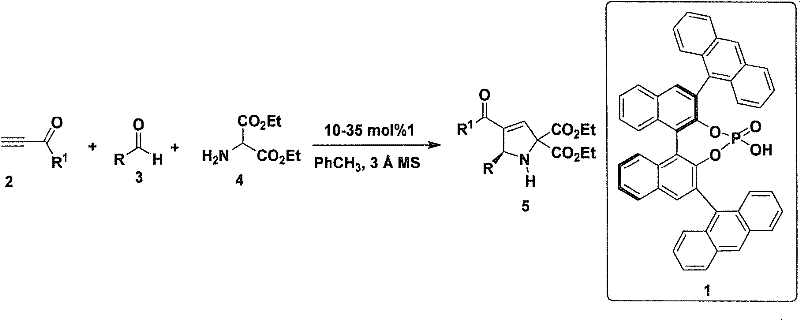
Mechanistic Insights into Chiral Phosphoric Acid Catalyzed Cycloaddition
Understanding the mechanistic underpinnings of this transformation is vital for process optimization and scale-up. The reaction proceeds via a chiral phosphoric acid-catalyzed 1,3-dipolar cycloaddition. Initially, the aldehyde and diethyl aminomalonate condense in situ to form an azomethine ylide or a similar reactive imine species. The chiral phosphoric acid, acting as a bifunctional organocatalyst, simultaneously activates the electrophilic ynone and the nucleophilic imine intermediate through hydrogen bonding interactions. This dual activation organizes the transition state within a well-defined chiral environment, ensuring high facial selectivity during the ring-closing event. The specific architecture of the catalyst, often derived from BINOL scaffolds as shown in the patent examples, creates a steric pocket that discriminates between enantiotopic faces of the reacting species.
From an impurity control perspective, this mechanism offers distinct advantages. Because the stereochemistry is established during the bond-forming step rather than through post-reaction resolution, the formation of the undesired enantiomer is suppressed at the source. This intrinsic selectivity minimizes the generation of diastereomeric impurities that are notoriously difficult to separate. Furthermore, the mild reaction conditions prevent the degradation of sensitive functional groups often present in complex drug-like molecules. For quality assurance teams, this translates to a cleaner crude reaction profile, simplifying the analytical workload and ensuring that the final high-purity pharmaceutical intermediates meet stringent regulatory specifications with greater consistency. The robustness of the catalytic cycle across a wide substrate scope further attests to the reliability of this mechanistic pathway for diverse chemical libraries.
How to Synthesize Chiral 2,5-Dihydropyrrole Efficiently
Implementing this synthesis route requires careful attention to reaction parameters to maximize yield and enantioselectivity. The protocol involves mixing the ynone, aldehyde, and diethyl aminomalonate in anhydrous toluene in the presence of activated molecular sieves to drive the equilibrium by removing water. The chiral phosphoric acid catalyst is then introduced, and the mixture is stirred at controlled temperatures. Following the reaction, standard workup procedures involving extraction and silica gel chromatography are employed to isolate the target compounds. The detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and safety in your laboratory operations.
- Combine ynone, aldehyde, and diethyl aminomalonate with molecular sieves in toluene solvent.
- Add chiral phosphoric acid catalyst (10-35 mol%) and stir at temperatures between -10°C to 30°C.
- Quench the reaction with saturated sodium bicarbonate, extract with ethyl acetate, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology offers substantial strategic benefits beyond mere technical elegance. The shift from chiral pool synthesis to asymmetric organocatalysis fundamentally alters the cost structure of producing these valuable intermediates. By utilizing achiral, commodity-grade starting materials such as simple aldehydes and ynones, the dependency on volatile markets for specialized chiral reagents is eliminated. This stability in raw material sourcing is critical for maintaining supply chain continuity and mitigating the risk of production delays caused by feedstock shortages. Additionally, the simplified workflow reduces the number of unit operations required, leading to lower labor costs and reduced facility occupancy time.
- Cost Reduction in Manufacturing: The elimination of expensive chiral starting materials and the reduction of synthetic steps result in significant operational expenditure savings. Traditional routes often require protecting group strategies and multiple purification stages, each adding cost and reducing overall mass balance. In contrast, this one-pot approach streamlines the process, minimizing solvent usage and waste disposal fees. The high catalytic efficiency means that less catalyst is required per kilogram of product, further driving down the variable costs associated with large-scale production. These efficiencies collectively contribute to a more competitive pricing model for high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Sourcing achiral building blocks is inherently more reliable than procuring specialized chiral compounds, which often have limited suppliers and long lead times. The robustness of the reaction conditions allows for flexible manufacturing scheduling, as the process is less sensitive to minor fluctuations in temperature or reagent quality compared to sensitive organometallic reactions. This resilience ensures a steady flow of materials to downstream customers, reducing the likelihood of stockouts that can disrupt clinical trial timelines. Furthermore, the scalability of the process from gram to kilogram scales has been demonstrated, providing confidence in the ability to meet increasing demand as a drug candidate progresses through development phases.
- Scalability and Environmental Compliance: The use of toluene as a solvent and the absence of heavy metal catalysts align well with modern green chemistry principles and environmental regulations. Heavy metal residues are a major concern in pharmaceutical manufacturing, often requiring costly removal steps to meet strict limits. By employing an organic catalyst, this risk is effectively removed, simplifying the validation process and reducing the environmental footprint of the manufacturing site. The high atom economy of the multicomponent reaction also means less waste is generated per unit of product, supporting corporate sustainability goals and reducing the burden on waste treatment facilities.
Frequently Asked Questions (FAQ)
To address common inquiries regarding the technical and commercial viability of this synthesis method, we have compiled answers based on the specific data and embodiments found within the patent documentation. These insights are intended to clarify the practical implications for industrial application and help stakeholders make informed decisions regarding the integration of this technology into their existing pipelines. Understanding these nuances is key to leveraging the full potential of this innovative chemical platform.
Q: What is the primary advantage of this synthesis method over traditional routes?
A: This method utilizes achiral starting materials and a one-pot asymmetric multicomponent reaction, eliminating the need for expensive chiral pools and complex multi-step sequences found in conventional olefin metathesis or Wittig-Horner approaches.
Q: What level of enantiomeric excess (ee) can be achieved?
A: The patent data demonstrates that this organocatalytic protocol consistently achieves high optical purity, with enantiomeric excess (ee) values reaching up to >99% for various substrates.
Q: Are the resulting compounds biologically active?
A: Yes, preliminary biological testing indicated that specific derivatives, such as compound 5aa, exhibit cytotoxic activity against MCF7 breast cancer cells, validating their potential as drug leads.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral 2,5-Dihydropyrrole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing advanced synthetic technologies to accelerate drug development. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries can be seamlessly translated into industrial reality. We are committed to delivering chiral 2,5-dihydropyrrole derivatives that adhere to stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our expertise in asymmetric synthesis allows us to optimize these organocatalytic processes for maximum efficiency and yield, providing our partners with a secure and high-quality supply of essential pharmaceutical intermediates.
We invite you to collaborate with us to explore how this novel synthesis route can enhance your project's economics and timeline. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities can support your journey from lead optimization to commercial launch. Let us be your partner in turning complex chemical challenges into commercial successes.
