Scalable Synthesis of 3-Aminopyrrole-2-Formamide Compounds for Pharmaceutical Manufacturing
Scalable Synthesis of 3-Aminopyrrole-2-Formamide Compounds for Pharmaceutical Manufacturing
The development of efficient synthetic routes for heterocyclic compounds remains a cornerstone of modern pharmaceutical process chemistry, particularly for scaffolds utilized in potent therapeutic agents. Patent CN111825593A discloses a groundbreaking synthetic method for 3-aminopyrrole-2-formamide compounds, which serve as critical intermediates in the manufacture of phosphodiesterase type V (PDE5) inhibitors. This specific class of pyrrolopyrimidinone derivatives has garnered significant attention in the treatment of various physiological conditions, necessitating robust and scalable production methods. The disclosed technology addresses long-standing inefficiencies in prior art by introducing a chloroacetamide-based pathway that fundamentally alters the reaction landscape. By shifting away from traditional ester-based starting materials, this innovation offers a streamlined approach that enhances both chemical yield and operational safety. For global procurement teams and R&D directors, understanding the nuances of this patented methodology is essential for securing a reliable pharmaceutical intermediate supplier capable of meeting stringent quality and volume demands.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
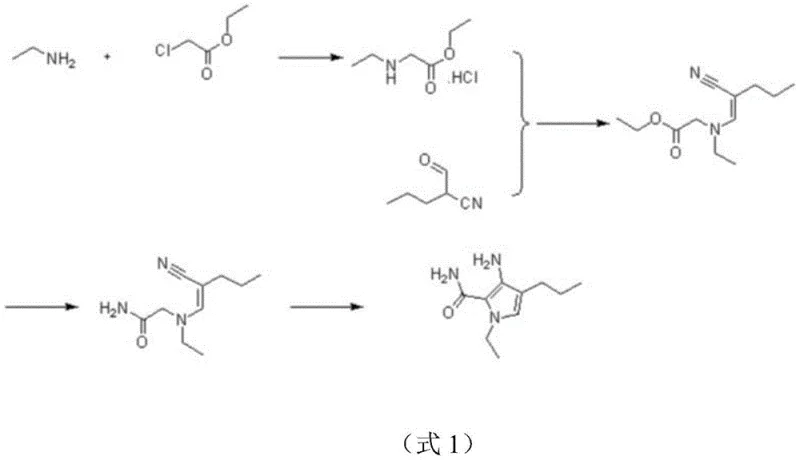
Historically, the synthesis of these vital pyrrole intermediates relied heavily on routes exemplified by U.S. Pat. No. 2003/0171361, which utilized ethyl chloroacetate as the primary alkylating agent. While chemically feasible, this conventional approach suffers from inherent thermodynamic and kinetic instabilities that complicate large-scale manufacturing. The ester functional group in ethyl chloroacetate is highly susceptible to nucleophilic attack, leading to unavoidable aminolysis side reactions when exposed to amine reagents like ethylamine. Furthermore, the potential for ester hydrolysis under varying pH conditions introduces a complex impurity profile that is difficult to manage. These side reactions not only consume valuable raw materials but also generate byproducts that require rigorous and costly purification steps, often involving column chromatography which is impractical for ton-scale production. Consequently, the overall yield of the target compound is compromised, and the cost of goods sold (COGS) increases significantly due to material loss and extended processing times.
The Novel Approach
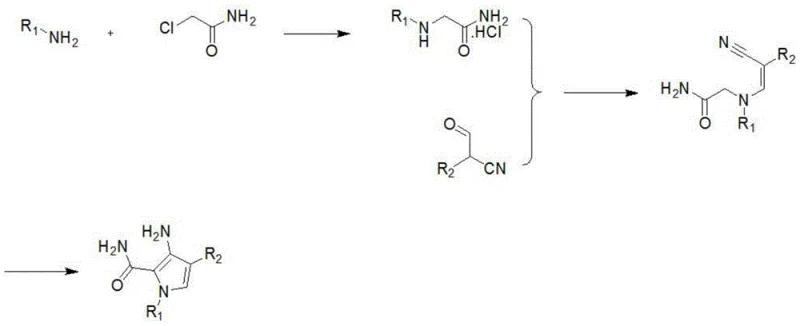
In stark contrast, the novel methodology presented in the patent data replaces the reactive ester moiety with a stable amide functionality by employing chloroacetamide as the key starting material. This strategic substitution effectively eliminates the risk of ester exchange and hydrolysis, thereby stabilizing the reaction pathway and minimizing the formation of unwanted byproducts. The new route proceeds through a concise three-step sequence that utilizes water and simple alcohols as solvents, aligning with green chemistry principles and reducing environmental impact. The stability of the chloroacetamide allows for milder reaction conditions, typically maintained below 100°C, which lowers energy consumption and enhances operational safety. Moreover, the post-treatment processes are significantly simplified, relying on standard unit operations such as distillation, extraction, and recrystallization rather than complex chromatographic separations. This transition represents a paradigm shift in cost reduction in API manufacturing, offering a more direct and economically viable path to high-purity intermediates.
Mechanistic Insights into Chloroacetamide-Based Cyclization
The core of this innovative synthesis lies in a carefully orchestrated sequence of nucleophilic substitution, condensation, and intramolecular cyclization. The first step involves the reaction of chloroacetamide with an alkylamine or ammonia gas in an aqueous medium under ice-bath conditions. This exothermic nucleophilic substitution forms a glycine derivative hydrochloride salt with high efficiency, leveraging the superior leaving group ability of the chloride ion while the amide group remains inert to the reaction conditions. Following isolation, this intermediate undergoes a condensation reaction with a specific cyano-aldehyde derivative in the presence of sodium acetate. This step constructs the carbon-nitrogen backbone required for the pyrrole ring, proceeding smoothly in alcohol solvents like methanol or ethanol. The final and most critical stage is the base-catalyzed ring-closing reaction, where an alkoxide base such as sodium ethoxide facilitates the intramolecular cyclization. This transformation closes the five-membered pyrrole ring, establishing the aromatic system essential for the biological activity of the final PDE5 inhibitor.
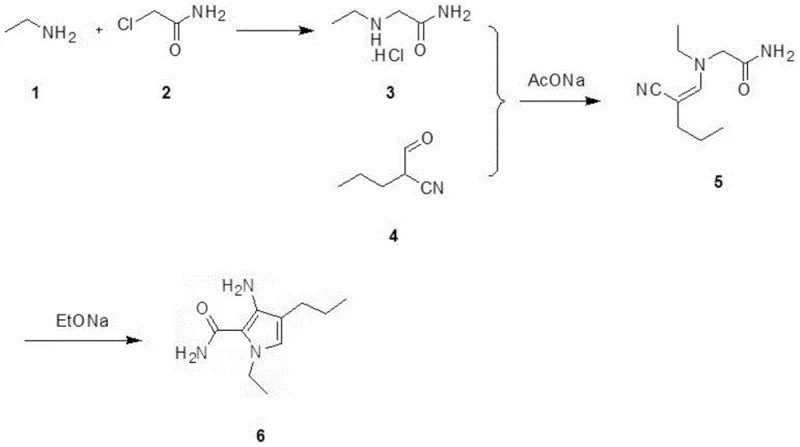
Impurity control is inherently built into this mechanistic design due to the stability of the amide bond throughout the synthetic sequence. Unlike ester-based routes where transesterification can create a myriad of analogues, the amide linkage ensures that the molecular skeleton remains intact until the intended cyclization event. The use of sodium acetate in the condensation step provides a buffered environment that prevents premature degradation of sensitive functional groups, while the final recrystallization from dichloromethane and hexanes ensures the removal of any residual salts or unreacted starting materials. This rigorous control over the reaction trajectory results in a product with a clean impurity profile, which is paramount for regulatory compliance in pharmaceutical applications. The ability to achieve high purity without resorting to preparative HPLC or flash chromatography underscores the robustness of this chemical design for industrial applications.
How to Synthesize 3-Aminopyrrole-2-Formamide Efficiently
The practical implementation of this synthesis requires precise control over stoichiometry and temperature to maximize yield and safety. The process begins with the careful addition of chloroacetamide to a cooled aqueous solution of the amine, ensuring the exotherm is managed effectively to prevent thermal runaway. Subsequent steps involve standard organic workups including pH adjustments and solvent exchanges, which are well-suited for existing reactor infrastructure. The detailed standardized synthetic steps see the guide below for specific operational parameters and molar ratios derived from the patent examples.
- React alkylamine or ammonia with chloroacetamide in water under ice-bath conditions to form the glycine derivative intermediate.
- Condense the glycine intermediate with a cyano-aldehyde derivative using sodium acetate in an alcohol solvent.
- Perform ring-closing cyclization using an alkoxide base (e.g., sodium ethoxide) in alcohol solvent at 50-70°C to yield the final pyrrole compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this chloroacetamide-based route offers substantial strategic benefits beyond mere chemical elegance. The elimination of unstable ester intermediates translates directly into improved supply chain reliability, as the raw materials are commodity chemicals with stable market availability and pricing. The simplified workup procedures reduce the dependency on specialized purification media like silica gel, which can be a bottleneck in large-scale campaigns. Furthermore, the use of green solvents such as water and ethanol minimizes waste disposal costs and aligns with increasingly stringent environmental regulations, facilitating smoother regulatory approvals for manufacturing sites.
- Cost Reduction in Manufacturing: The replacement of ethyl chloroacetate with chloroacetamide fundamentally lowers the cost structure by eliminating expensive purification steps associated with ester side reactions. By avoiding the need for column chromatography and reducing the formation of hard-to-remove impurities, the process significantly cuts down on solvent consumption and labor hours. The higher overall yield means less raw material is required per kilogram of finished product, driving down the variable cost of production. Additionally, the mild reaction temperatures reduce energy loads on heating and cooling systems, contributing to further operational savings.
- Enhanced Supply Chain Reliability: Sourcing chloroacetamide and simple alkylamines is far more predictable than managing specialty esters that may have limited suppliers. The robustness of the reaction against moisture and minor temperature fluctuations ensures consistent batch-to-batch quality, reducing the risk of production delays due to out-of-specification results. This stability allows for longer campaign runs and better inventory planning, ensuring that downstream API synthesis is not interrupted by intermediate shortages. The simplified logistics of handling non-hazardous solvents like water and ethanol also streamline transportation and storage requirements.
- Scalability and Environmental Compliance: The process is inherently designed for commercial scale-up of complex pharmaceutical intermediates, utilizing unit operations that are standard in multi-purpose chemical plants. The absence of heavy metal catalysts or toxic reagents simplifies waste stream treatment, lowering the environmental footprint and associated disposal fees. The ability to recycle solvents like ethanol and dichloromethane further enhances the sustainability profile of the manufacturing process. This alignment with green chemistry principles not only reduces costs but also strengthens the corporate social responsibility profile of the supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic route. These insights are derived directly from the patent specifications and are intended to clarify the operational advantages for potential partners.
Q: Why is chloroacetamide preferred over ethyl chloroacetate in this synthesis?
A: Chloroacetamide avoids the ester aminolysis and hydrolysis side reactions common with ethyl chloroacetate, leading to higher yields and simpler purification without column chromatography.
Q: What are the typical reaction conditions for the cyclization step?
A: The cyclization is performed in an alcohol solvent (methanol or ethanol) using a matching alkoxide base at mild temperatures between 50°C and 70°C for 1 to 6 hours.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process uses green solvents like water and alcohols, operates at low temperatures (<100°C), and avoids complex purification steps, making it highly scalable and cost-effective.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Aminopyrrole-2-Formamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your drug development programs. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements without compromising on quality. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 3-aminopyrrole-2-formamide meets the highest industry standards. Our commitment to process excellence allows us to deliver materials that facilitate smooth downstream synthesis for our global clients.
We invite you to contact our technical procurement team to discuss how this advanced synthetic route can benefit your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic advantages of switching to this chloroacetamide-based method. We are ready to provide specific COA data and route feasibility assessments to support your decision-making process and help you secure a competitive edge in the marketplace.
