Advanced Catalytic Synthesis of Chiral Alpha-Methylene Beta-Lactams for Commercial Drug Development
The pharmaceutical industry continuously seeks robust methodologies for constructing chiral heterocycles, particularly beta-lactams, which serve as privileged scaffolds in medicinal chemistry. Patent CN103570600A discloses a groundbreaking preparation method for chiral alpha-methylene beta-lactam compounds, utilizing a sophisticated palladium-catalyzed asymmetric allylic amination strategy. This technology addresses the longstanding challenge of achieving high regio- and enantioselectivity in the functionalization of Morita-Baylis-Hillman (MBH) adducts. By employing a novel chiral phosphine ligand complexed with palladium, the process enables the efficient synthesis of key chiral beta-amido alpha-methylene carboxylic acid derivatives. These intermediates are subsequently converted into bioactive beta-lactam structures through a streamlined cyclization step. For R&D teams focused on oncology, this pathway offers access to compounds with demonstrated antitumor activity, providing a reliable foundation for developing next-generation therapeutic agents.
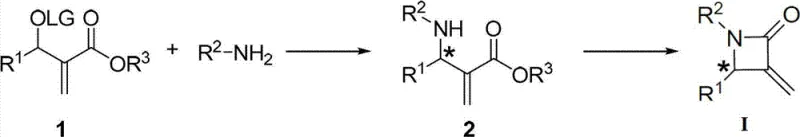
Traditional methods for synthesizing beta-lactam derivatives often suffer from limitations regarding substrate scope, harsh reaction conditions, or poor stereocontrol, which can severely impact the viability of a drug candidate. Conventional approaches may require multiple protection and deprotection steps, leading to increased waste generation and reduced overall atom economy. Furthermore, achieving high enantiomeric excess without expensive chiral auxiliaries has historically been a bottleneck in the cost reduction in pharmaceutical intermediates manufacturing. In contrast, the novel approach detailed in the patent leverages transition-metal catalysis to activate weak nucleophiles, such as aromatic amines, which were previously difficult to employ effectively. This methodology not only expands the chemical space accessible to medicinal chemists but also simplifies the synthetic route by reducing the number of operational steps required to reach the target scaffold.
The core of this innovation lies in the specific design of the chiral spiroketal-based bisphosphine ligands which coordinate with palladium to form highly active catalytic species. These ligands create a rigid chiral environment that dictates the facial selectivity of the allylic amination reaction, ensuring that the resulting carbon-nitrogen bond is formed with precise stereochemistry. The mechanism involves the oxidative addition of the palladium catalyst to the allylic substrate, followed by nucleophilic attack by the amine, and subsequent reductive elimination to release the product. This catalytic cycle is remarkably tolerant of various functional groups, allowing for the diversification of the R1 and R2 substituents on the beta-lactam ring. Such mechanistic precision is critical for minimizing the formation of diastereomeric impurities, thereby reducing the burden on downstream purification processes and enhancing the overall quality of the high-purity pharmaceutical intermediates produced.
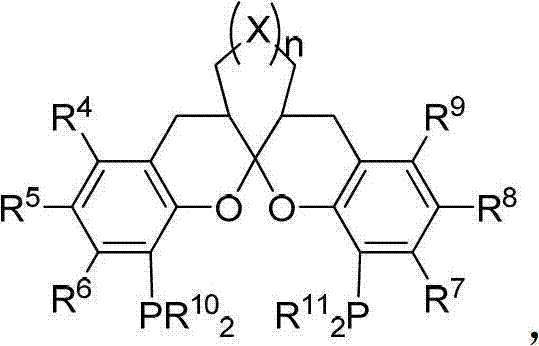
Controlling the impurity profile is paramount when scaling up synthetic routes for clinical candidates, and this patent provides specific insights into managing stereoisomeric purity. The use of defined chiral ligands ensures that the major enantiomer is produced preferentially, often with ee values exceeding 90 percent, which significantly lowers the risk of toxicological issues associated with minor enantiomers. Additionally, the reaction conditions allow for the use of aqueous bases in certain embodiments, which can simplify the quenching and extraction phases of the workup. This reduces the reliance on large volumes of organic solvents for washing, contributing to a greener process profile. For supply chain managers, understanding these impurity control mechanisms is essential for assessing the robustness of the manufacturing process and ensuring consistent batch-to-batch quality, which is a prerequisite for regulatory approval in the competitive landscape of commercial scale-up of complex polymer additives and fine chemicals.
How to Synthesize Chiral Alpha-Methylene Beta-Lactams Efficiently
Implementing this synthesis route requires careful attention to catalyst preparation and reaction parameters to maximize yield and selectivity. The process begins with the in situ formation of the palladium-ligand complex under an inert atmosphere, typically using solvents like dichloromethane or tetrahydrofuran. The subsequent amination step is conducted at mild temperatures, ranging from sub-zero to ambient conditions, depending on the specific substrate reactivity. Following the isolation of the chiral amino-ester intermediate, the final cyclization is achieved using strong non-nucleophilic bases to induce ring closure. Detailed standardized synthesis steps are provided in the guide below to assist technical teams in replicating these results accurately.
- Prepare the catalyst complex by mixing a chiral spiroketal-based bisphosphine ligand with a palladium precursor in an organic solvent under inert atmosphere.
- React the Morita-Baylis-Hillman (MBH) adduct with an amine nucleophile in the presence of the catalyst and a base to form the chiral beta-amido intermediate.
- Subject the chiral intermediate to cyclization conditions using a strong base to close the beta-lactam ring and obtain the final chiral alpha-methylene beta-lactam compound.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the adoption of this catalytic technology offers substantial strategic benefits regarding cost efficiency and supply security. The ability to use readily available starting materials, such as MBH adducts and common amines, mitigates the risk of raw material shortages that often plague specialized synthetic routes. Furthermore, the high catalytic efficiency means that lower loadings of expensive palladium metals may be utilized while maintaining high conversion rates, directly contributing to cost reduction in API manufacturing. The streamlined nature of the synthesis, which avoids lengthy protection group manipulations, reduces the total processing time and labor costs associated with production. These factors combine to create a more resilient supply chain capable of meeting the demanding timelines of modern drug development programs without compromising on quality or compliance standards.
- Cost Reduction in Manufacturing: The elimination of multiple synthetic steps and the use of efficient catalytic systems significantly lower the operational expenses associated with producing these complex intermediates. By avoiding the need for stoichiometric chiral reagents and reducing solvent consumption through simplified workups, the overall cost of goods sold is optimized. This economic efficiency allows for more competitive pricing strategies when sourcing high-value pharmaceutical building blocks, ensuring that budget allocations for R&D projects are utilized effectively without unnecessary expenditure on inefficient chemical transformations.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions ensures consistent output even when scaling from laboratory to pilot plant volumes. The tolerance of the catalyst system to various substrates means that a single manufacturing line can potentially produce a diverse range of analogues, increasing flexibility in response to changing project requirements. This adaptability reduces lead time for high-purity pharmaceutical intermediates by minimizing the need for process re-validation when structural modifications are required. Reliable suppliers can leverage this flexibility to maintain continuous supply streams, preventing delays in clinical trial material production.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing reaction conditions that are safe and manageable in large-scale reactors. The potential use of aqueous bases and the reduction of hazardous waste streams align with increasingly stringent environmental regulations globally. This compliance reduces the regulatory burden and associated disposal costs for manufacturing partners. Scalable processes that minimize environmental impact are increasingly preferred by top-tier pharmaceutical companies, making this technology a sustainable choice for long-term commercial partnerships and reducing the ecological footprint of chemical production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. Understanding these details helps stakeholders evaluate the feasibility of integrating this method into their existing development pipelines. The answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, ensuring accuracy and relevance for decision-makers.
Q: What is the enantioselectivity of this beta-lactam synthesis method?
A: The patented process utilizes chiral spiroketal bisphosphine ligands which achieve high enantioselectivity, often exceeding 90% ee, ensuring high purity of the chiral centers critical for biological activity.
Q: Can this synthesis method be scaled for industrial production?
A: Yes, the reaction conditions utilize common organic solvents and commercially available palladium precursors. The use of aqueous bases in certain steps simplifies workup, facilitating commercial scale-up of complex pharmaceutical intermediates.
Q: What are the primary applications of these chiral beta-lactam compounds?
A: These compounds exhibit significant antitumor activity against various cell lines including leukemia and lung cancer cells, making them valuable high-purity pharmaceutical intermediates for oncology drug development.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Beta-Lactam Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis, offering extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is well-versed in the nuances of palladium-catalyzed reactions and chiral resolution techniques, ensuring that the transition from bench-scale discovery to full-scale manufacturing is seamless. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of chiral alpha-methylene beta-lactams meets the exacting standards required for oncology drug development. Our commitment to quality assurance ensures that your supply chain remains uninterrupted and compliant with international regulatory frameworks.
We invite you to collaborate with us to leverage this advanced technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can accelerate your timeline to market. Let us be your partner in transforming innovative chemical concepts into commercial realities.
