Advancing Antiviral Intermediate Production: Scalable Immobilized Enzyme Catalysis for 2'-Fluoro-Deoxyribonucleosides
The pharmaceutical industry's relentless pursuit of potent antiviral and anticancer agents has placed 2'-fluoro-deoxyribonucleosides at the forefront of medicinal chemistry research. As critical intermediates for blockbuster drugs like Sofosbuvir and various HIV therapies, the demand for efficient, scalable, and cost-effective synthesis methods is paramount. Patent CN115851694A introduces a groundbreaking preparation method and application of immobilized enzymes specifically designed for synthesizing these high-value compounds. Unlike traditional liquid enzyme systems which suffer from instability and single-use limitations, this innovation leverages high-porosity polymer carriers to anchor uridine phosphorylase and base phosphorylase. This technological leap not only enhances the catalytic activity and loading capacity of the enzymes but also fundamentally transforms the economic model of nucleoside manufacturing by enabling catalyst reusability and robust operational stability under varying pH and temperature conditions.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2'-fluoro-2'-deoxyribonucleosides has relied heavily on multi-step chemical pathways that are fraught with inefficiencies and environmental hazards. Conventional chemical routes typically involve the use of 9-β-D-arabinofuranosyl adenine as a starting material, necessitating a cumbersome five-step reaction sequence to achieve the final product. These chemical processes often require harsh reaction conditions, including extreme temperatures and the use of toxic organic solvents, which pose significant safety risks and generate substantial hazardous waste. Furthermore, chemical glycosylation frequently struggles with stereoselectivity, leading to complex mixtures of anomers that require difficult and costly purification steps. The reliance on protecting group strategies in chemical synthesis further elongates the production timeline and increases the overall cost of goods, making it challenging for manufacturers to meet the growing global demand for affordable antiviral medications without compromising on purity or yield.
The Novel Approach
In stark contrast, the novel approach detailed in CN115851694A utilizes a biocatalytic transglycosylation strategy that bypasses the need for complex protection and deprotection sequences. By employing recombinant E. coli strains expressing both uridine phosphorylase and purine phosphorylase, the method facilitates a direct sugar exchange between a donor nucleoside and an acceptor base. The key innovation lies in the immobilization of these enzymes onto a polymer carrier, which creates a heterogeneous catalytic system. This setup allows the reaction to proceed in a packed column format, ensuring continuous contact between the substrate and the biocatalyst while preventing enzyme leaching. The result is a streamlined process that operates under mild, environmentally friendly conditions, significantly reducing the number of unit operations required and minimizing the generation of toxic by-products, thereby offering a sustainable alternative to legacy chemical manufacturing.
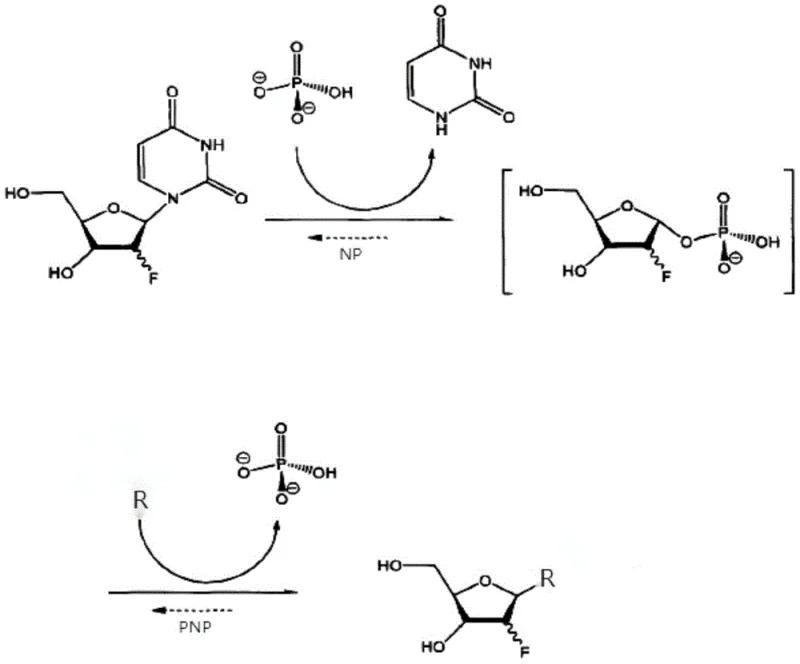
Mechanistic Insights into Dual-Enzyme Transglycosylation
The core of this technology rests on a sophisticated dual-enzyme cascade mechanism involving Uridine Phosphorylase (UPase) and Purine Nucleoside Phosphorylase (PNPase). In the first step, UPase catalyzes the phosphorolysis of the donor substrate, typically 2'-fluoro-2'-deoxyuridine, cleaving the N-glycosidic bond to release the fluorinated ribose-1-phosphate intermediate and uracil. Subsequently, PNPase mediates the condensation of this activated sugar phosphate with the acceptor base, such as adenine, cytosine, or thymine, to form the desired 2'-fluoro-deoxyribonucleoside product. This reversible equilibrium is driven forward by the thermodynamic stability of the product or by optimizing the molar ratios of the substrates. The immobilization matrix, often composed of materials like MI-150IDA or MI-300IDA, provides a high specific surface area that maximizes enzyme loading while maintaining the conformational flexibility necessary for catalytic turnover. This structural arrangement ensures that mass transfer limitations are minimized, allowing substrates to diffuse efficiently into the porous carrier to access the active sites of the entrapped enzymes.
Controlling impurity profiles in nucleoside synthesis is critical for regulatory compliance, and this biocatalytic method offers inherent advantages in selectivity. The enzymatic active sites are highly specific for the β-anomeric configuration, effectively suppressing the formation of unwanted α-anomers that are common side products in chemical glycosylation. Additionally, the mild aqueous environment prevents the degradation of the sensitive 2'-fluoro moiety, which can be prone to elimination reactions under acidic or basic chemical conditions. The immobilization process itself acts as a purification step; since the enzymes are covalently or physically bound to the solid support, they do not contaminate the final liquid product stream. This eliminates the need for aggressive protein precipitation or ultrafiltration steps post-reaction, resulting in a cleaner crude product that requires less intensive chromatographic purification. Consequently, the overall impurity spectrum is narrowed, facilitating easier validation and higher final purity for the API intermediate.
How to Synthesize 2'-Fluoro-Deoxyribonucleosides Efficiently
The practical implementation of this synthesis route involves a well-defined sequence of bioprocessing steps, beginning with the fermentation of recombinant E. coli strains. The patent outlines the cloning of specific genes into vectors like pET28a or pBAD, followed by induction with agents such as IPTG or L-arabinose to maximize enzyme expression. Once the biomass is harvested and lysed via high-pressure homogenization, the crude enzyme extract is incubated with the polymer carrier to form the immobilized biocatalyst. This solid catalyst is then packed into a column reactor, where the substrate solution containing the fluorinated donor and the nitrogenous base is circulated. Detailed standardized synthetic steps for optimizing flow rates, residence times, and substrate concentrations are provided in the guide below to ensure maximum conversion efficiency.
- Clone genes encoding uridine phosphorylase and purine phosphorylase into E. coli vectors (e.g., pET28a or pBAD) and ferment in LB medium with inducers like IPTG or L-arabinose.
- Harvest cells via centrifugation, disrupt them using high-pressure homogenization, and bind the crude enzyme extract to a polymer carrier (e.g., MI-150IDA) to create the immobilized biocatalyst.
- Pack the immobilized enzyme into a column, feed a solution of 2'-fluoro-2'-deoxyuridine and a base acceptor (e.g., adenine), and maintain pH 6.0-8.0 at 40-60°C for transglycosylation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the shift from chemical synthesis to this immobilized enzyme technology represents a strategic opportunity to optimize cost structures and mitigate supply risks. Traditional chemical routes for nucleoside analogs are often bottlenecked by the availability of specialized reagents and the capacity of facilities equipped to handle hazardous solvents. By transitioning to an aqueous biocatalytic process, manufacturers can leverage existing fermentation infrastructure and reduce dependency on volatile petrochemical-derived solvents. The ability to reuse the immobilized enzyme over multiple batches significantly amortizes the cost of the biocatalyst, leading to substantial reductions in variable production costs. Furthermore, the simplified downstream processing reduces the consumption of chromatography resins and solvents, contributing to a leaner and more cost-efficient manufacturing operation that is less susceptible to raw material price fluctuations.
- Cost Reduction in Manufacturing: The implementation of immobilized enzymes eliminates the need for expensive transition metal catalysts and complex protecting group chemistry, which are major cost drivers in traditional nucleoside synthesis. By enabling the repeated reuse of the biocatalyst, the effective cost per kilogram of enzyme usage is drastically lowered compared to single-use liquid enzyme preparations. Additionally, the high selectivity of the enzymatic reaction reduces the burden on purification systems, lowering the consumption of silica gel and HPLC columns, which translates directly into improved gross margins for the final pharmaceutical intermediate.
- Enhanced Supply Chain Reliability: Biocatalytic processes are inherently more robust against supply chain disruptions associated with specialty chemical reagents. The primary inputs for this method—glucose for fermentation and simple nitrogenous bases—are commodity chemicals with stable global supply lines. The stability of the immobilized enzyme also allows for the creation of strategic stockpiles of the biocatalyst, ensuring continuity of production even if upstream fermentation faces temporary delays. This resilience is crucial for maintaining consistent delivery schedules to downstream API manufacturers who operate on tight just-in-time inventory models.
- Scalability and Environmental Compliance: Scaling biocatalytic reactions is generally more straightforward than scaling exothermic chemical reactions, as the heat generation is minimal and easily managed in aqueous systems. The process generates significantly less hazardous waste, simplifying compliance with increasingly stringent environmental regulations regarding solvent discharge and heavy metal residues. This 'green' profile not only reduces waste disposal costs but also enhances the corporate sustainability credentials of the supply chain, a factor that is becoming increasingly important for multinational pharmaceutical clients auditing their vendor networks.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this immobilized enzyme technology. These insights are derived directly from the experimental data and claims presented in patent CN115851694A, providing a factual basis for evaluating the feasibility of adopting this route for commercial production. Understanding these nuances is essential for R&D teams planning pilot studies and procurement officers negotiating supply contracts.
Q: What are the primary advantages of using immobilized enzymes over free liquid enzymes for nucleoside synthesis?
A: According to patent CN115851694A, immobilized enzymes offer significantly higher stability against temperature and pH fluctuations, allow for repeated reuse which drastically lowers catalyst costs, and simplify downstream processing by enabling easy separation from the reaction mixture, thereby reducing protein impurities in the final API intermediate.
Q: Which specific enzymes are utilized in this biocatalytic transglycosylation process?
A: The process utilizes a dual-enzyme system comprising Uridine Phosphorylase (UPase) and Purine Nucleoside Phosphorylase (PNPase), expressed in recombinant E. coli strains. These enzymes work synergistically to transfer the 2'-fluoro-ribose moiety from the donor nucleoside to the acceptor base.
Q: How does this method impact the environmental footprint of antiviral drug manufacturing?
A: This biocatalytic route operates under mild aqueous conditions (pH 6.0-8.0, 40-80°C), eliminating the need for hazardous organic solvents and heavy metal catalysts often required in traditional chemical synthesis. This results in reduced three-waste discharge and aligns with green chemistry principles for sustainable pharmaceutical production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2'-Fluoro-Deoxyribonucleoside Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the immobilized enzyme technology described in CN115851694A for the production of high-value antiviral intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes are seamlessly translated into robust industrial operations. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced fermentation suites capable of handling recombinant organisms, allowing us to meet stringent purity specifications required for GMP-grade pharmaceutical ingredients. We are committed to delivering consistent quality and reliability, leveraging our technical expertise to optimize yield and minimize impurities in every batch.
We invite global pharmaceutical partners to collaborate with us to harness the efficiencies of this biocatalytic route. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out for specific COA data and route feasibility assessments to determine how our optimized manufacturing capabilities can support your supply chain goals and accelerate your drug development timelines.
