Scalable Enzymatic Synthesis of Rare Sugar Nucleotides for Commercial Pharmaceutical Applications
The landscape of glycobiology and pharmaceutical intermediate manufacturing is undergoing a significant transformation driven by the innovations detailed in patent CN116287064A. This intellectual property introduces a groundbreaking universal method for synthesizing rare sugar nucleotides directly from UDP-GlcNAc, addressing long-standing bottlenecks in availability and cost. Rare sugar nucleotides are critical donors for glycosyltransferases, essential for constructing complex glycans and glycoconjugates used in drug development. However, traditional access to these molecules has been severely limited by expensive chemical synthesis routes and inefficient biological pathways. The disclosed technology leverages a One-Pot Multi-Enzyme (OPME) strategy coupled with advanced Cofactor Regeneration Systems (CRS) to overcome these hurdles. By enabling large-scale, high-efficiency production without complicated purification operations, this method offers a robust solution for reliable rare sugar nucleotides supplier networks seeking to enhance their portfolio. The integration of enzymatic specificity with economic manufacturing principles marks a pivotal shift in how high-purity pharmaceutical intermediates are sourced globally.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the acquisition of rare sugar nucleotides has been plagued by substantial technical and economic challenges that hinder widespread research and commercial application. Conventional chemical synthesis often requires multi-step protection and deprotection strategies, leading to low overall yields and the generation of significant chemical waste. Furthermore, the polar and charged nature of sugar nucleotides makes them poorly soluble in organic solvents, complicating reaction conditions and downstream processing. On the biological front, natural biosynthetic pathways are often too complex and impractical for industrial replication due to the requirement for expensive stoichiometric cofactors like NADH, NADPH, and AcCoA. Without regeneration, the cost of these cofactors renders large-scale production economically unviable. Additionally, traditional step-wise enzymatic methods often suffer from the accumulation of unstable intermediates that hydrolyze rapidly, reducing final product purity and necessitating rigorous and costly purification techniques such as HPLC. These factors collectively create a high barrier to entry for cost reduction in pharmaceutical intermediates manufacturing.
The Novel Approach
The methodology presented in CN116287064A fundamentally reengineers the synthesis workflow by combining OPME technology with efficient cofactor regeneration. Instead of relying on expensive stoichiometric amounts of cofactors, this approach utilizes catalytic amounts regenerated in situ from inexpensive substrates like D-glucose, pyruvate, or sodium acetate. This shift drastically lowers the raw material costs while maintaining high reaction driving forces through irreversible regeneration steps. The one-pot design minimizes unit operations by allowing multiple enzymatic transformations to occur sequentially in a single vessel, thereby reducing labor, time, and equipment requirements. Crucially, the system is designed to avoid the accumulation of unstable intermediates by coupling their formation immediately with consumption in subsequent steps. This results in a streamlined process where the final product can often be isolated through simple desalting on a P2 column, bypassing the need for complex chromatography. This novel approach not only enhances yield but also significantly simplifies the supply chain for complex pharmaceutical intermediates.
Mechanistic Insights into OPME and Cofactor Regeneration Systems
The core technical advantage of this patent lies in the sophisticated coupling of specific enzyme cascades with tailored cofactor regeneration loops. For instance, in reduction-directed pathways, enzymes such as Mg534 and Mg535 catalyze dehydration and epimerization steps that require NADH or NADPH. Rather than adding these costly reduced cofactors directly, the system employs a regeneration cycle where D-glucose dehydrogenase (BsGH) oxidizes D-glucose to gluconolactone, simultaneously reducing NAD+ to NADH. This cycle ensures a constant supply of reducing power throughout the reaction, driving the equilibrium towards the desired rare sugar nucleotide product. The specificity of the enzymes ensures high stereo- and regioselectivity, which is critical for the biological activity of the resulting glycans. By carefully selecting enzyme combinations from sources like Megavirus chiliensis or Campylobacter jejuni, the process can be adapted to synthesize a diverse array of structures including UDP-L-RhaNAc and UDP-D-QuiNAc with high fidelity.
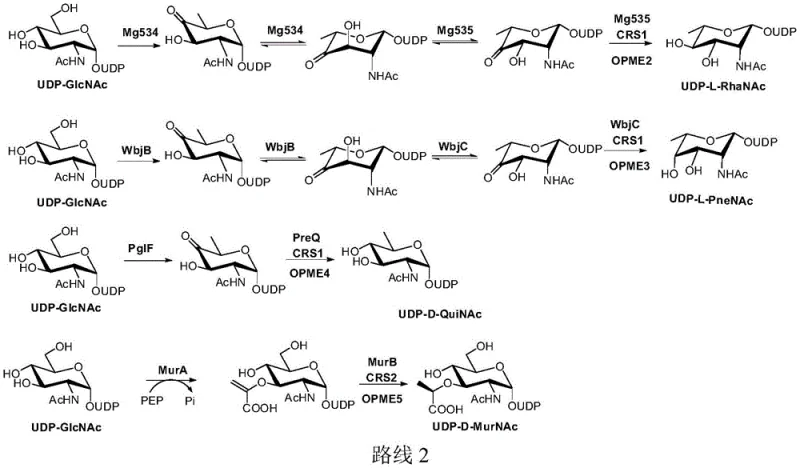
Furthermore, the impurity control mechanism is inherently built into the reaction design. Since the intermediates are consumed as soon as they are formed, their concentration remains low, minimizing side reactions such as hydrolysis which typically degrade sugar nucleotides. The use of specific regeneration systems like CRS1 for reduction or CRS3 for oxidation ensures that the reaction environment remains optimized for the primary enzymatic activity. For example, in oxidation pathways, the regeneration of NAD+ from NADH prevents product inhibition that often plagues dehydrogenase reactions. This mechanistic precision allows for the commercial scale-up of complex pharmaceutical intermediates that were previously only available in milligram quantities. The ability to tune the regeneration system based on the specific redox or group transfer requirements of the target molecule provides a versatile platform for synthesizing various high-purity oligosaccharide precursors needed in modern drug discovery.
How to Synthesize Rare Sugar Nucleotides Efficiently
Implementing this synthesis route requires a precise understanding of the enzymatic components and reaction conditions outlined in the patent. The process begins with the preparation of a buffered reaction system containing the starting substrate UDP-GlcNAc, which can be sourced commercially or synthesized via Route 1 as described in the patent data. Specific enzyme cocktails are then selected based on the target nucleotide structure, such as using PglF and PreQ for UDP-D-QuiNAc or WbpO for UDP-D-GlcNAcA. The critical step involves the addition of the appropriate Cofactor Regeneration System (CRS), which must be matched to the enzymatic requirements; for instance, CRS1 is utilized for NADH-dependent reductions while CRS5 is employed for AcCoA-dependent acetylations. The detailed standardized synthesis steps see the guide below, which outlines the exact molar ratios, pH controls, and temperature settings required to achieve optimal conversion rates and yields.
- Prepare the reaction buffer with UDP-GlcNAc substrate and specific enzymes tailored for the target nucleotide structure.
- Introduce the Cofactor Regeneration System (CRS) such as CRS1 or CRS3 to sustain enzymatic activity without stoichiometric cofactor costs.
- Execute the one-pot reaction at controlled pH and temperature, followed by simple desalting purification to isolate the final product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this technology offers transformative benefits that directly impact the bottom line and operational reliability. The primary advantage is the drastic reduction in raw material costs achieved by replacing expensive stoichiometric cofactors with catalytic systems driven by cheap commodities like glucose and acetate. This economic efficiency makes the production of rare sugar nucleotides viable for large-scale applications, opening up new markets for glycan-based therapeutics. Additionally, the simplification of the purification process reduces the dependency on specialized chromatography resins and solvents, further lowering operational expenditures. The robustness of the one-pot system also enhances supply chain reliability by minimizing the number of process steps where failures or delays could occur. This streamlined workflow ensures consistent quality and availability, which is crucial for maintaining continuous manufacturing schedules in the pharmaceutical industry.
- Cost Reduction in Manufacturing: The implementation of cofactor regeneration systems fundamentally alters the cost structure of nucleotide synthesis by eliminating the need for purchasing expensive reduced cofactors in large quantities. Instead of consuming NADH or AcCoA stoichiometrically, the process recycles these molecules continuously, meaning only a small catalytic amount is required to drive the reaction to completion. This shift results in substantial cost savings on raw materials, which is particularly significant given the high market price of purified cofactors. Furthermore, the use of inexpensive substrates like D-glucose and sodium acetate as sacrificial donors ensures that the recurring material costs remain low and predictable. The reduction in purification complexity also lowers the cost of goods sold by decreasing solvent usage and waste disposal fees associated with traditional multi-step synthesis.
- Enhanced Supply Chain Reliability: The reliance on readily available and stable starting materials such as UDP-GlcNAc and common buffer components significantly mitigates supply chain risks. Unlike specialized chemical reagents that may have long lead times or limited suppliers, the substrates for this enzymatic process are commoditized and easily sourced from multiple vendors. The robustness of the enzymatic reactions under mild conditions (e.g., 37°C, pH 7.5) reduces the need for specialized high-pressure or high-temperature equipment, simplifying facility requirements. This accessibility ensures that production can be scaled up rapidly to meet demand fluctuations without being bottlenecked by rare reagent availability. Consequently, partners can expect reduced lead time for high-purity pharmaceutical intermediates and a more resilient supply network capable of withstanding market volatility.
- Scalability and Environmental Compliance: The one-pot nature of this synthesis route is inherently scalable, allowing for seamless transition from laboratory benchtop to industrial bioreactors without significant process reengineering. The elimination of organic solvents and harsh chemical reagents aligns with green chemistry principles, reducing the environmental footprint of the manufacturing process. Waste generation is minimized because the byproducts of the regeneration systems, such as gluconolactone or lactate, are generally benign and easier to treat than toxic chemical waste. This environmental compliance simplifies regulatory approvals and reduces the costs associated with waste management. The high selectivity of the enzymes also means fewer byproducts are formed, leading to higher atom economy and a more sustainable production model that meets the increasing demands for eco-friendly chemical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this OPME technology for rare sugar nucleotide production. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on yield, purity, and scalability. Understanding these aspects is vital for R&D and procurement teams evaluating this method for integration into their existing workflows. The responses highlight the practical advantages of the cofactor regeneration strategy and the versatility of the enzyme systems involved.
Q: How does the cofactor regeneration system reduce production costs?
A: The system regenerates expensive cofactors like NADH and AcCoA in situ using cheap substrates like glucose, shifting from stoichiometric to catalytic usage.
Q: What is the purity level achievable with this OPME method?
A: The method avoids unstable intermediates and allows for simple P2 column desalting, yielding high-purity products suitable for pharmaceutical research.
Q: Is this synthesis route scalable for industrial manufacturing?
A: Yes, the one-pot design eliminates complex isolation steps, making it highly adaptable for commercial scale-up from kilograms to metric tons.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Rare Sugar Nucleotides Supplier
NINGBO INNO PHARMCHEM stands at the forefront of adopting such advanced synthetic methodologies to deliver high-quality chemical solutions to the global market. As a specialized CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes like the OPME system are successfully translated into industrial reality. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that verify every batch against the highest industry standards. We understand the critical nature of sugar nucleotides in drug development and are equipped to handle the sensitivities of these molecules with precision. By leveraging our technical expertise, we can help you secure a stable supply of these essential intermediates, enabling your research and production teams to focus on innovation rather than sourcing challenges.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs through the adoption of these efficient enzymatic routes. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and target molecules. We encourage you to reach out to request specific COA data and route feasibility assessments to determine how this technology can benefit your projects. By partnering with us, you gain access to a reliable network capable of delivering complex intermediates with speed and precision. Let us help you navigate the complexities of glycan synthesis and accelerate your time to market with our superior manufacturing capabilities.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
