Advanced Copper-Catalyzed Asymmetric Synthesis of Chiral Octopamine Derivatives for Commercial Pharmaceutical Applications
Advanced Copper-Catalyzed Asymmetric Synthesis of Chiral Octopamine Derivatives for Commercial Pharmaceutical Applications
The pharmaceutical industry continuously seeks robust methodologies for accessing high-value chiral intermediates, particularly those exhibiting potent biological activities such as anticancer properties. Patent CN102059145A introduces a groundbreaking catalytic system designed to streamline the production of (S)-octopamine and its downstream derivative, (S)-N-trans-feruloyl norsynephrine. This specific phenolic cyclic amide has garnered significant attention due to its superior efficacy in killing cancerous white blood cells compared to its enantiomer. The disclosed technology leverages a novel chiral ligand coordinated with divalent copper to facilitate an asymmetric Henry reaction, bypassing the inefficiencies of traditional racemic resolution. By enabling the direct construction of the chiral center from achiral precursors like p-hydroxybenzaldehyde and nitromethane, this invention offers a scalable pathway for reliable pharmaceutical intermediate supplier networks aiming to secure high-purity chiral amines.
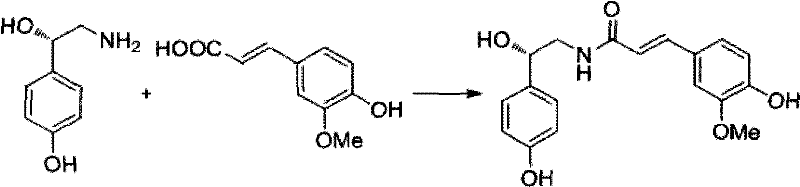
The strategic value of this patent lies in its comprehensive approach to synthesizing complex bioactive molecules. The process begins with the generation of a chiral nitro-alcohol precursor, which is subsequently reduced to the amine and coupled with ferulic acid. This multi-step sequence is optimized for operational simplicity, utilizing mild reaction temperatures ranging from 0°C to 25°C and common solvents like water and ethanol. For procurement managers focused on cost reduction in API manufacturing, the ability to operate without extreme cryogenic conditions or exotic reagents translates directly into lower utility costs and simplified infrastructure requirements. Furthermore, the high stereoselectivity reported in the patent examples ensures that the resulting intermediates meet the stringent purity specifications demanded by regulatory bodies for clinical applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the acquisition of optically pure (S)-octopamine has been fraught with significant technical and economic challenges that hinder large-scale commercialization. Traditional approaches often relied on the resolution of racemic mixtures using chiral acids, such as camphorsulfonic acid, a method inherently limited by a maximum theoretical yield of 50% for the desired enantiomer. This inefficiency necessitates the disposal or recycling of the unwanted (R)-enantiomer, creating substantial waste streams and driving up the cost per kilogram of the final active ingredient. Additionally, alternative synthetic routes involving functional group transformations frequently employ hazardous reagents like sodium azide, which poses severe safety risks regarding toxicity and explosiveness during scale-up. These legacy methods also tend to involve cumbersome protection and deprotection steps, elongating the production timeline and increasing the cumulative loss of material at each stage, thereby compromising the overall supply chain reliability for high-purity chiral amines.
The Novel Approach
In stark contrast to these inefficient legacy protocols, the methodology described in patent CN102059145A utilizes a direct asymmetric catalytic route that constructs the chiral center in a single bond-forming event. By employing a specialized copper-ligand complex, the process achieves high enantioselectivity directly from simple, commercially available starting materials, effectively doubling the potential yield compared to resolution strategies. The reaction conditions are notably mild, proceeding efficiently in aqueous or alcoholic media at near-ambient temperatures, which drastically reduces energy consumption and eliminates the need for specialized low-temperature reactors. This streamlined approach not only simplifies the operational workflow but also enhances the safety profile of the manufacturing process by avoiding toxic azides and explosive intermediates. Consequently, this novel approach represents a paradigm shift towards greener, more economical, and safer production of complex phenolic amides.
Mechanistic Insights into Cu-Catalyzed Asymmetric Henry Reaction
The core innovation of this technology resides in the sophisticated design of the chiral ligand, which features a pyrrolidine backbone substituted with bulky diphenylhydroxymethyl groups and specific ortho-substituents like trifluoromethyl or methyl groups. When coordinated with a divalent copper salt, such as copper bromide, this ligand forms a rigid chiral pocket that dictates the stereochemical outcome of the nucleophilic attack. The mechanism involves the activation of the nitroalkane by the basic components of the catalyst system, generating a nitronate species that attacks the aldehyde coordinated to the copper center. The steric bulk of the ligand's phenyl rings and the specific electronic influence of the ortho-substituent create a highly differentiated environment, favoring the formation of the (S)-configured beta-nitro alcohol with exceptional selectivity.
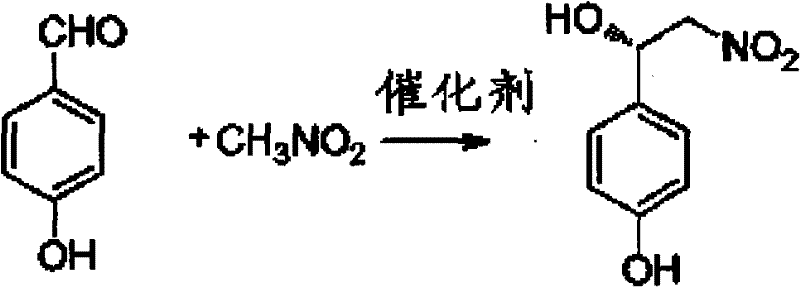
Impurity control is intrinsically managed through the high fidelity of this catalytic cycle. Because the chiral information is transferred during the initial carbon-carbon bond formation, the generation of the undesired (R)-enantiomer is suppressed at the source rather than removed downstream. The patent data indicates that the catalyst system maintains high activity even with low catalyst loading, minimizing the risk of metal contamination in the final product. Furthermore, the use of phase transfer catalysts like tetrabutylammonium bromide facilitates the interaction between organic substrates and inorganic bases in biphasic systems, ensuring homogeneous reaction kinetics and preventing localized hotspots that could lead to side reactions. This precise control over the reaction trajectory ensures that the crude product profile is clean, reducing the burden on downstream purification units and facilitating the commercial scale-up of complex polymer additives or pharmaceutical intermediates.
How to Synthesize (S)-N-trans-Feruloyl Norsynephrine Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for translating laboratory success into industrial reality. The process is divided into three distinct phases: the asymmetric construction of the nitro-alcohol scaffold, the chemoselective reduction of the nitro group to an amine, and the final amide coupling with ferulic acid. Each step is optimized for high conversion and ease of workup. For instance, the initial Henry reaction can be performed in water, allowing for simple extraction protocols where the product moves to the organic phase while the catalyst components remain in the aqueous layer for recycling. This modularity allows process engineers to adapt the workflow to existing facility capabilities without requiring massive capital expenditure on new equipment.
- Perform an asymmetric Henry reaction between p-hydroxybenzaldehyde and nitromethane using a chiral copper-ligand complex catalyst to generate the (S)-octopamine precursor.
- Conduct a catalytic hydrogenation reduction of the nitro-alcohol precursor using Pd/C or Pb/C under mild pressure to yield (S)-octopamine.
- Execute an amidation coupling reaction between the resulting (S)-octopamine and ferulic acid using standard condensing agents like EDCI/HOBt to form the final target molecule.
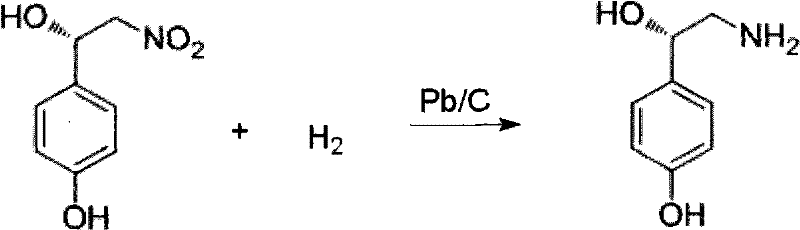
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders, the transition to this catalytic methodology offers profound advantages in terms of raw material security and logistical stability. The starting materials, including p-hydroxybenzaldehyde and nitromethane, are commodity chemicals available from multiple global sources, mitigating the risk of supply bottlenecks associated with exotic chiral pool starting materials. The robustness of the catalyst system, which tolerates mild conditions and common solvents, ensures consistent batch-to-batch quality, a critical factor for maintaining long-term contracts with pharmaceutical clients. Moreover, the demonstrated ability to recover and reuse the expensive chiral ligand significantly lowers the variable cost of production, providing a buffer against raw material price volatility and enhancing the overall margin structure of the manufacturing operation.
- Cost Reduction in Manufacturing: The elimination of stoichiometric chiral resolving agents and the avoidance of hazardous reagents like sodium azide result in substantial cost savings. By shifting from a resolution-based model (max 50% yield) to a direct asymmetric synthesis, the effective throughput of the reactor is doubled without increasing capital investment. Additionally, the recovery of the chiral ligand from the aqueous phase, as demonstrated in the patent examples with recovery rates exceeding 80%, means that the most expensive component of the reaction mixture is not consumed but rather circulated, drastically reducing the cost of goods sold over the lifecycle of the product.
- Enhanced Supply Chain Reliability: The reliance on stable, shelf-stable reagents and the absence of sensitive organometallic species that require inert atmosphere handling simplify logistics and storage requirements. The process operates effectively at ambient temperatures, removing the dependency on complex cryogenic cooling systems that are prone to failure and energy-intensive. This operational resilience ensures that production schedules can be maintained consistently, reducing lead time for high-purity chiral amines and allowing suppliers to respond more agilely to fluctuating market demands without compromising on delivery commitments.
- Scalability and Environmental Compliance: The use of water and ethanol as primary solvents aligns perfectly with modern green chemistry principles and stringent environmental regulations. Unlike processes relying on chlorinated solvents or heavy metal catalysts that require complex remediation, this aqueous-based system generates waste streams that are easier to treat and dispose of. The simplicity of the workup procedure, involving standard liquid-liquid extraction and filtration, facilitates seamless scale-up from pilot plants to multi-ton commercial production, ensuring that environmental compliance costs remain manageable while maximizing production capacity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this catalytic technology. They are derived from the specific experimental data and beneficial effects detailed in the patent documentation, providing clarity on the practical aspects of adopting this synthesis route. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this process into their existing manufacturing portfolios.
Q: What represents the primary advantage of this copper-catalyzed method over traditional resolution techniques?
A: Unlike traditional resolution methods which theoretically limit yield to 50% and require expensive chiral acids, this direct asymmetric synthesis builds chirality from achiral starting materials, offering potentially higher atom economy and eliminating the need for wasteful separation of the unwanted enantiomer.
Q: Is the chiral ligand used in this process recoverable for industrial reuse?
A: Yes, the patent explicitly demonstrates that the chiral ligand remains in the aqueous phase after extraction and can be recovered with high efficiency (up to 84% recovery rate reported in examples), significantly reducing the cost of goods for large-scale manufacturing.
Q: What are the typical reaction conditions required for the key asymmetric step?
A: The critical Henry reaction operates under remarkably mild conditions, typically between 0°C and 25°C, utilizing environmentally benign solvents such as water or ethanol, which simplifies thermal management and safety protocols compared to cryogenic organometallic processes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S)-N-trans-Feruloyl Norsynephrine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the catalytic technologies described in patent CN102059145A for the production of high-value anticancer intermediates. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale discovery to industrial manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of chiral intermediate meets the exacting standards required for pharmaceutical development and commercial drug substance manufacturing.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage these advanced synthetic routes for their pipeline projects. By partnering with our technical procurement team, you can obtain a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us directly to request specific COA data and route feasibility assessments, allowing us to demonstrate how our expertise in asymmetric catalysis can accelerate your development timelines and optimize your supply chain economics.
