Advanced Catalytic Synthesis of Optically Active Oxoheptenoic Acid Esters for Statin Production
The pharmaceutical industry's relentless pursuit of high-purity intermediates for cardiovascular therapeutics has found a significant breakthrough in the methodology detailed in patent CN1288136C. This document outlines a novel, highly efficient process for producing optically active (E)-7-[2-cyclopropyl-4-(4-fluorophenyl)quinolin-3-yl]-5-hydroxy-3-oxohept-6-enoic acid esters, which serve as pivotal precursors for next-generation statin drugs. Unlike traditional synthetic routes that struggle with stereochemical control and harsh reaction conditions, this innovation leverages a sophisticated binaphthol-titanium catalytic system enhanced by specific additives. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, understanding the mechanistic advantages of this route is essential for securing a robust supply chain for lipid-lowering medications. The ability to consistently achieve optical purities exceeding 99% ee represents a paradigm shift in the manufacturing of these complex chiral molecules.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the synthesis of this critical statin intermediate relied heavily on methods involving optically active Schiff base-titanium coordination compounds. These legacy processes were fraught with significant operational inefficiencies and quality control challenges. The preparation of the Schiff base ligand itself required multiple synthetic steps, introducing complexity and potential points of failure in the supply chain. Furthermore, the resulting optical purity of the target compound was often limited to approximately 78% ee, necessitating costly and yield-reducing optical resolution steps to meet the stringent standards required for API production. Alternative literature methods, while offering slightly better purity, demanded rigorous exclusion of moisture through the use of high-temperature dried molecular sieves and strict solvent distillation protocols. These苛刻 conditions not only increased energy consumption but also posed significant hurdles for commercial scale-up, making the reliable production of high-purity pharmaceutical intermediates a persistent bottleneck in the industry.
The Novel Approach
The patented process introduces a streamlined and highly effective alternative by utilizing an optically active dinaphthol-titanium coordination compound derived directly from 1,1'-bi-2-naphthol and titanium tetraisopropoxide. The true genius of this method lies in the strategic addition of a metal salt, specifically lithium chloride, and an amine, such as N,N,N',N'-Tetramethyl Ethylene Diamine (TMEDA), directly into the reaction system. This modification eliminates the need for complex ligand synthesis and harsh drying agents. The reaction proceeds between a 1,3-bis(trimethylsilyloxy)-1-alkoxybuta-1,3-diene, represented as Formula (I), and an aldehyde precursor, Formula (II). 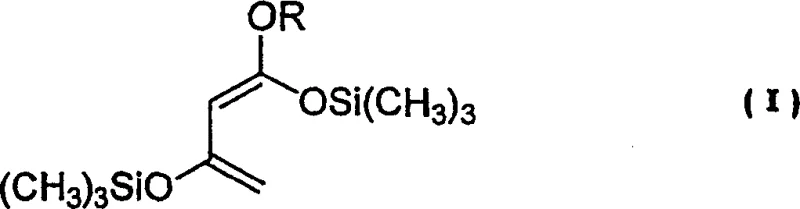
![Chemical structure of (E)-3-[2-cyclopropyl-4-(4-fluorophenyl)-quinolin-3-yl]-prop-2-en-1-al (Formula II)](/insights/img/oxoheptenoic-acid-ester-synthesis-pharma-supplier-20260315163850-02.webp) By simply mixing these components in a solvent like tetrahydrofuran at mild temperatures ranging from 0 to 45°C, the process achieves yields greater than 85% with optical purity surpassing 99% ee. This dramatic improvement in both efficiency and selectivity underscores the value of this technology for cost reduction in API manufacturing.
By simply mixing these components in a solvent like tetrahydrofuran at mild temperatures ranging from 0 to 45°C, the process achieves yields greater than 85% with optical purity surpassing 99% ee. This dramatic improvement in both efficiency and selectivity underscores the value of this technology for cost reduction in API manufacturing.
Mechanistic Insights into Binaphthol-Titanium Catalyzed Asymmetric Addition
The core of this technological advancement is the formation of a chiral Lewis acid complex that dictates the stereochemical outcome of the carbon-carbon bond-forming reaction. The binaphthol-titanium complex acts as a chiral template, coordinating with the carbonyl oxygen of the aldehyde substrate to activate it towards nucleophilic attack by the silyl diene. The introduction of lithium chloride plays a dual role: it likely acts as a Lewis acid co-catalyst to further activate the electrophile and may also assist in the aggregation state of the titanium species, creating a more rigid and selective transition state. Simultaneously, the amine additive, particularly TMEDA, modulates the coordination sphere of the titanium center. This modulation prevents the formation of inactive catalyst aggregates and accelerates the turnover frequency of the catalytic cycle. The synergy between the chiral ligand, the metal salt, and the amine creates a highly organized chiral environment that effectively discriminates between the enantiotopic faces of the aldehyde, ensuring the exclusive formation of the desired (S)-configured alcohol center.
From an impurity control perspective, this mechanism offers substantial benefits. The high stereoselectivity (>99% ee) means that the formation of the undesired enantiomer is virtually suppressed, eliminating the need for downstream chiral chromatography or recrystallization steps that typically result in significant material loss. Furthermore, the mild reaction conditions prevent the degradation of sensitive functional groups present in the quinoline moiety, such as the cyclopropyl ring and the fluorophenyl group. The subsequent desilylation step is performed directly in the reaction mixture or after a simple extractive workup, using aqueous acid to cleave the silyl ethers. This telescoped approach minimizes the handling of unstable intermediates and reduces the generation of solvent waste, aligning with green chemistry principles while ensuring the final product meets the rigorous purity specifications demanded by global regulatory bodies for cardiovascular drug substances.
How to Synthesize Optically Active Oxoheptenoic Acid Ester Efficiently
The implementation of this synthesis route requires precise control over stoichiometry and addition sequences to maximize the benefits of the catalytic system. The process begins with the in situ or pre-formation of the chiral titanium catalyst, followed by the sequential addition of the lithium salt and amine promoters before introducing the substrates. Maintaining an inert atmosphere is crucial to prevent catalyst deactivation by moisture. The reaction is typically conducted in tetrahydrofuran at temperatures between 0°C and 45°C for a duration of 2 to 6 hours. Following the coupling reaction, the mixture is treated with aqueous acid to effect desilylation, yielding the final hydroxy-keto ester. For a detailed, step-by-step standard operating procedure including exact molar ratios and workup parameters, please refer to the technical guide below.
- Prepare the optically active dinaphthol-titanium coordination compound by reacting 1,1'-bi-2-naphthol with titanium tetraisopropoxide in an organic solvent.
- React the silyl diene (Formula I) with the quinoline aldehyde (Formula II) in the presence of the catalyst, adding lithium chloride and an amine like TMEDA to enhance stereoselectivity.
- Perform desilylation on the reaction mixture using acid treatment followed by neutralization and crystallization to isolate the high-purity product (>99% ee).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented process translates into tangible strategic advantages beyond mere technical superiority. The elimination of multi-step ligand synthesis and the removal of stringent drying requirements significantly simplify the manufacturing workflow. This simplification directly correlates to reduced operational expenditures and a lower risk of batch failures due to environmental factors. The ability to run the reaction at near-ambient temperatures (0-45°C) rather than cryogenic conditions drastically lowers energy costs associated with cooling, making the process economically viable for large-scale production. Furthermore, the use of commodity chemicals like lithium chloride and TMEDA ensures that the supply chain for reagents remains stable and cost-effective, mitigating the risks associated with sourcing exotic or expensive catalysts.
- Cost Reduction in Manufacturing: The most significant economic driver of this technology is the drastic increase in reaction yield and optical purity. By achieving yields over 85% and optical purity above 99% ee in a single pass, the process eliminates the need for expensive and yield-eroding purification steps such as chiral resolution. In traditional methods, losing nearly half the material to reach high purity was a common hidden cost; this new route preserves material throughput. Additionally, the simplified catalyst preparation avoids the labor and solvent costs associated with synthesizing complex Schiff bases. The overall result is a substantially lower cost of goods sold (COGS) for the intermediate, providing a competitive edge in pricing negotiations for the final API.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by complex synthetic routes that rely on fragile intermediates or hard-to-source reagents. This process utilizes robust, commercially available starting materials including 1,1'-bi-2-naphthol and titanium tetraisopropoxide. The reaction tolerance is improved, reducing the sensitivity to minor fluctuations in raw material quality or environmental conditions. The shortened reaction time, dropping from potentially days in older methods to just a few hours, increases the throughput capacity of existing manufacturing assets. This agility allows suppliers to respond more rapidly to demand spikes, ensuring that downstream API manufacturers face fewer delays and can maintain consistent inventory levels for their own production schedules.
- Scalability and Environmental Compliance: Scaling chemical processes often exposes safety and environmental weaknesses, but this methodology is inherently designed for expansion. The exotherm is manageable at the recommended temperatures, and the solvent system (THF) is well-understood in industrial settings. The reduction in solvent usage for purification, driven by the high crude purity of the product, leads to a smaller environmental footprint. Less waste solvent means lower disposal costs and easier compliance with increasingly strict environmental regulations. The process avoids the use of heavy metals or toxic reagents that would require specialized waste treatment, streamlining the path to regulatory approval for the manufacturing site and ensuring long-term operational sustainability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthetic route. These answers are derived directly from the experimental data and claims within the patent documentation, providing clarity on the feasibility and benefits of the technology for potential partners and licensees.
Q: What optical purity can be achieved with this new titanium-catalyzed process?
A: The patented process achieves an exceptional optical purity of over 99% ee, significantly surpassing conventional methods which typically yield around 78% ee.
Q: Why are lithium chloride and amine additives critical in this reaction?
A: The addition of metal salts like lithium chloride and amines such as TMEDA drastically accelerates the reaction rate and improves stereocontrol, allowing the reaction to complete in hours rather than days while maintaining high enantioselectivity.
Q: What is the primary pharmaceutical application of this intermediate?
A: This optically active oxoheptenoic acid ester serves as a crucial synthetic intermediate for producing statins used in the prevention and treatment of hyperlipidemia and arteriosclerosis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Oxoheptenoic Acid Ester Supplier
The transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. NINGBO INNO PHARMCHEM stands ready to leverage this advanced titanium-catalyzed technology to deliver high-quality statin intermediates to the global market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in practice. We operate with stringent purity specifications and utilize rigorous QC labs to guarantee that every batch meets the >99% ee benchmark required for modern pharmaceutical applications. Our commitment to quality assurance ensures that your supply chain remains uninterrupted and compliant with international standards.
We invite you to collaborate with us to optimize your sourcing strategy for cardiovascular intermediates. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage potential partners to contact us to obtain specific COA data and route feasibility assessments that demonstrate how this innovative process can enhance your product portfolio. Let us help you secure a competitive advantage through superior chemistry and reliable supply.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
