Advanced Synthesis of Gypsy Moth Sex Pheromone: Scalable Routes for Agrochemical Leaders
Advanced Synthesis of Gypsy Moth Sex Pheromone: Scalable Routes for Agrochemical Leaders
The global demand for environmentally friendly pest control solutions has intensified the search for efficient synthetic routes to semiochemicals, specifically the gypsy moth sex pheromone known as Disparlure. Patent CN102321050A, published in early 2012, introduces a groundbreaking methodology for synthesizing the active enantiomers (+)-(7R,8S)-7,8-epoxy-2-methyloctadecane (I) and its counterpart (-)-(7S,8R)-7,8-epoxy-2-methyloctadecane (II). This technology represents a significant leap forward in agrochemical intermediate manufacturing, utilizing cheap and readily available 2-propargyl alcohol as the starting material to achieve high efficiency and exceptional enantioselectivity without the need for complex protecting group strategies.
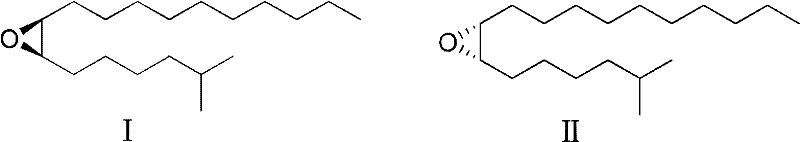
The biological efficacy of these epoxy compounds is strictly dependent on their stereochemistry, with the natural (7R,8S)-isomer exhibiting potent attraction capabilities while the enantiomer can act as an inhibitor. The patented process addresses the critical industry need for high-purity intermediates by delivering an enantiomeric excess (e.e.) exceeding 98% for isomer I and 99% for isomer II. For R&D directors and procurement specialists in the agrochemical sector, this level of optical purity translates directly to reduced field application rates and minimized environmental impact, positioning this synthesis route as a superior choice for commercial scale-up of complex epoxy compounds.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of disparlure has been plagued by inefficient multi-step sequences that rely on expensive chiral pool starting materials or cumbersome resolution techniques. Early methodologies, such as the route reported by K. Mori in 1979 starting from natural L-(+)-tartrate, required up to 12 reaction steps to construct the carbon skeleton, resulting in a dismal overall yield of merely 5%. These legacy processes often involve the repeated use of toxic organocopper reagents and necessitate the installation and removal of multiple protecting groups, which drastically increases the E-factor and production costs.
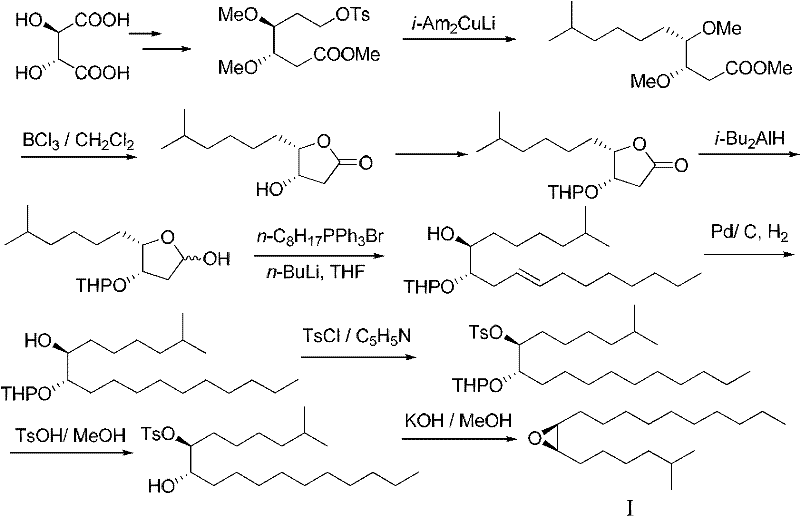
Furthermore, alternative approaches utilizing enzyme-catalyzed kinetic resolutions or olefin metathesis have struggled to balance step economy with stereocontrol. For instance, routes employing Grubbs catalysts introduce significant cost burdens due to the expense of the transition metal catalysts, while often failing to achieve the stringent optical purity required for effective pest management. The reliance on such complex sequences creates substantial supply chain vulnerabilities, as each additional step introduces potential yield losses and purification bottlenecks that hinder the reliable supply of high-purity agrochemical intermediates.
The Novel Approach
In stark contrast, the methodology disclosed in CN102321050A streamlines the entire production workflow into a concise 6-step sequence that bypasses the need for protecting groups entirely. By leveraging 2-propargyl alcohol as a versatile building block, the process constructs the requisite carbon chain through efficient alkylation and coupling reactions. The core innovation lies in the strategic application of Sharpless asymmetric epoxidation to install the critical chiral epoxide moiety with high fidelity, followed by a robust coupling with 4-methyl-1-pentyne derivatives.
This novel approach not only boosts the overall yield to an impressive 29% for the (7R,8S)-isomer and 27% for the (7S,8R)-isomer but also utilizes common, inexpensive reagents such as n-Butyl Lithium and titanium tetraisopropoxide. The elimination of protecting group manipulations significantly reduces solvent consumption and waste generation, aligning perfectly with modern green chemistry principles. For supply chain heads, this simplified protocol意味着 a drastic reduction in lead time for high-purity agrochemical intermediates, ensuring consistent availability for large-scale pheromone trap manufacturing.
Mechanistic Insights into Sharpless Asymmetric Epoxidation and Alkyne Coupling
The stereochemical integrity of the final pheromone is established primarily through the Sharpless asymmetric epoxidation, a reaction renowned for its predictability and broad scope. In this specific pathway, the Z-alkene intermediate (compound 3) is subjected to oxidation using tert-butyl hydroperoxide (TBHP) in the presence of titanium tetraisopropoxide and a chiral diethyl tartrate ligand. The choice between D-(-)-diethyl tartrate and L-(+)-diethyl tartrate allows for the precise divergence towards either the (7R,8S) or (7S,8R) configuration, providing a flexible platform for producing both the attractant and the inhibitor enantiomers from a common precursor.
Following the establishment of the chiral epoxide, the synthesis employs a nucleophilic substitution strategy to extend the carbon chain. The epoxide alcohol is activated as a triflate ester (compound 5 or 8) using trifluoromethanesulfonic anhydride, creating a highly reactive electrophile. This species then undergoes coupling with a lithiated alkyne derivative (generated from 4-methyl-1-pentyne) under cryogenic conditions to prevent racemization. The subsequent catalytic hydrogenation using palladium on carbon saturates the triple bond without affecting the sensitive epoxide ring, a testament to the chemoselectivity of the chosen conditions. This mechanistic precision ensures that impurity profiles remain minimal, a critical factor for regulatory compliance in agrochemical registrations.
How to Synthesize Disparlure Efficiently
The implementation of this synthesis route requires careful control of reaction temperatures and stoichiometry, particularly during the lithiation and epoxidation steps. The process begins with the alkylation of 2-propargyl alcohol at -78°C to form the C13 backbone, followed by semi-hydrogenation to the Z-alkene. The detailed standardized synthesis steps, including specific solvent choices like dichloromethane for epoxidation and ether for coupling, are outlined below to guide process chemists in replicating this high-yielding protocol.
- Coupling of 2-propargyl alcohol with 1-bromo-decane using n-Butyl Lithium to form the C13 alkyne backbone.
- Stereoselective hydrogenation using Lindlar catalyst to generate the Z-alkene intermediate.
- Sharpless asymmetric epoxidation using titanium tetraisopropoxide and chiral tartrate esters to establish the 7R,8S or 7S,8R configuration.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting this patented synthesis route offers transformative economic benefits for manufacturers of agrochemical intermediates. By condensing the production timeline from over 10 steps to just 6, facilities can significantly increase throughput capacity without expanding physical infrastructure. The use of commodity chemicals like 2-propargyl alcohol and 1-bromo-decane ensures raw material security and shields production costs from the volatility associated with specialized chiral pool starting materials. This stability is crucial for long-term procurement planning and budget forecasting in the competitive pest control market.
- Cost Reduction in Manufacturing: The elimination of protecting group chemistry removes entire classes of reagents and solvents from the bill of materials, directly lowering the variable cost per kilogram. Furthermore, the avoidance of expensive transition metal catalysts like Grubbs or Rhodium complexes, which are common in alternative metathesis routes, results in substantial cost savings. The higher overall yield means less starting material is wasted, optimizing atom economy and reducing the cost burden of raw material procurement.
- Enhanced Supply Chain Reliability: Shorter synthetic routes inherently reduce the risk of batch failures and supply interruptions. With fewer unit operations, there are fewer opportunities for equipment downtime or quality deviations, leading to more predictable delivery schedules. The reliance on stable, shelf-stable reagents rather than sensitive biological enzymes or air-sensitive organometallics simplifies inventory management and storage requirements, enhancing the resilience of the supply chain against external disruptions.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing standard unit operations such as extraction, distillation, and column chromatography that are easily transferred from pilot plant to commercial production. The reduction in waste generation, driven by higher yields and fewer steps, simplifies effluent treatment and lowers the environmental compliance burden. This aligns with increasingly stringent global regulations on industrial emissions, facilitating smoother regulatory approvals for new manufacturing sites.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of gypsy moth sex pheromones using this advanced methodology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing clarity for stakeholders evaluating this technology for integration into their supply chains.
Q: What is the enantiomeric excess achieved in this new synthesis method?
A: The patented process achieves an enantiomeric excess (e.e.) of greater than 98% for the (7R,8S)-isomer and greater than 99% for the (7S,8R)-isomer, ensuring high biological activity for pest control applications.
Q: How does this route compare to traditional methods in terms of step count?
A: Traditional methods reported in literature often require 10 to 12 steps with overall yields as low as 5%. This novel approach reduces the synthesis to only 6 steps with a significantly improved overall yield of 27-29%.
Q: Are protective groups required for this synthesis?
A: No, a key advantage of this methodology is that it proceeds without the need for protecting groups, which simplifies the workflow, reduces reagent costs, and minimizes waste generation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Disparlure Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity semiochemicals play in sustainable agriculture. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated stereochemical control required for Disparlure synthesis is maintained at every scale. We operate stringent purity specifications and utilize rigorous QC labs to guarantee that every batch meets the exacting standards required for effective pest management programs worldwide.
We invite you to collaborate with us to optimize your supply chain for agrochemical intermediates. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your growth in the bio-rational pesticide sector.
