Advanced Synthesis of Cardiocyclene-BODIPY Dyads for High-Efficiency Solid-State Fluorescence
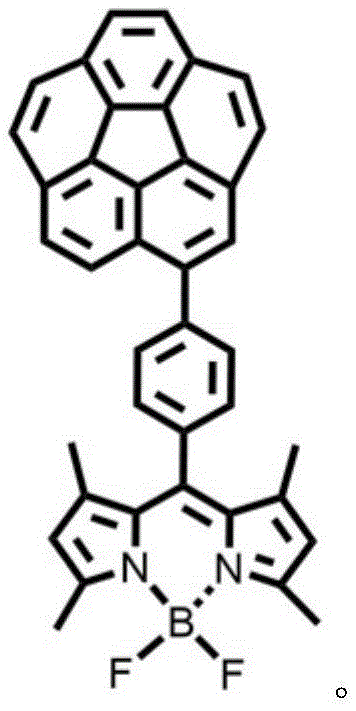
The rapid advancement of optoelectronic technologies has created an urgent demand for organic fluorescent materials that maintain high quantum efficiency in the solid state, a challenge that traditional planar fluorophores often fail to meet due to aggregation-caused quenching. Patent CN111499659B addresses this critical bottleneck by disclosing a novel cardiocyclene-fluoroboron dipyrrole binary fluorescent material, synthesized through a strategic covalent linkage of a curved fused-ring aromatic hydrocarbon to a BODIPY unit. This innovation represents a significant leap forward for manufacturers seeking a reliable OLED material supplier, as it effectively decouples molecular packing from fluorescence efficiency. By utilizing a classic Suzuki coupling reaction, the patent demonstrates a reproducible pathway to create dyads that exhibit robust luminescence in both solution and aggregated solid powder states, solving a persistent issue in the commercialization of high-performance display and sensing materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional BODIPY dyes are renowned for their exceptional photophysical properties in dilute solutions, including high fluorescence quantum yields, large molar absorption coefficients, and excellent chemical stability. However, their application in solid-state devices such as Organic Light-Emitting Diodes (OLEDs) and Organic Field-Effect Transistors (OFETs) has been severely hampered by the phenomenon of concentration quenching. In the solid state, the planar nature of standard BODIPY cores facilitates tight intermolecular π-π stacking, which creates non-radiative decay pathways that drastically reduce or completely extinguish fluorescence emission. Previous attempts to mitigate this effect often involved introducing bulky substituents to increase intermolecular distance, but these modifications frequently compromised the electronic properties or synthetic accessibility of the dye, limiting their utility in cost reduction in electronic chemical manufacturing.
The Novel Approach
The patented technology introduces a paradigm shift by integrating a rigid, curved cardiocyclene scaffold directly onto the BODIPY framework, creating a binary molecular architecture that inherently resists planar stacking. Unlike simple steric bulk, the bowl-shaped geometry of the cardiocyclene unit imposes a three-dimensional constraint that physically prevents the close approach of adjacent fluorophores in the aggregated state. This structural design preserves the intrinsic advantages of the BODIPY chromophore, such as its narrow emission band and pH insensitivity, while simultaneously unlocking strong solid-state fluorescence. For supply chain heads focused on the commercial scale-up of complex polymer additives or electronic materials, this approach offers a distinct advantage by utilizing well-established cross-coupling chemistry that avoids the need for exotic or unstable reagents, ensuring a consistent and high-quality supply of advanced fluorescent intermediates.
Mechanistic Insights into Suzuki-Catalyzed Dyad Formation
The core of this synthesis lies in the palladium-catalyzed cross-coupling between a monobrominated cardiocyclene derivative and a boronate-functionalized BODIPY precursor. The reaction mechanism proceeds through the canonical oxidative addition of the aryl bromide to the Pd(0) center, followed by transmetallation with the organoboron species in the presence of a base such as potassium carbonate. The unique curvature of the cardiocyclene moiety does not inhibit the catalytic cycle; rather, it ensures that the resulting dyad maintains a specific spatial orientation that minimizes intermolecular interactions post-synthesis. This precise control over molecular geometry is critical for R&D directors evaluating the purity and杂质 profile of the final product, as it reduces the formation of excimers or aggregates that could otherwise act as impurities affecting device performance.
Furthermore, the choice of reaction conditions plays a pivotal role in maximizing yield and minimizing side reactions. The patent specifies a biphasic solvent system comprising toluene, ethanol, and water in a 2:2:1 volume ratio, which optimizes the solubility of both the organic substrates and the inorganic base required for the activation of the boron species. Heating the mixture to reflux at temperatures between 85°C and 90°C for 20 to 24 hours ensures complete conversion of the starting materials, driving the equilibrium towards the desired binary product. This robust mechanistic pathway allows for the production of high-purity OLED material with minimal byproduct formation, simplifying downstream purification and enhancing the overall economic viability of the process for industrial applications.
How to Synthesize Cardiocyclene-BODIPY Dyad Efficiently
The preparation of this advanced fluorescent material follows a streamlined three-step protocol that balances reaction efficiency with ease of purification, making it highly suitable for scale-up operations. The process begins with the careful assembly of reactants under inert conditions to prevent catalyst deactivation, followed by a prolonged heating phase to ensure high conversion rates. The final isolation involves standard extraction and chromatographic techniques that are familiar to process chemists, allowing for the rapid transition from laboratory bench to pilot plant. Detailed standardized synthesis steps are provided in the guide below to assist technical teams in replicating these results with precision.
- Prepare the reaction mixture by combining boronate BODIPY, monobrominated cardiocyclene, potassium carbonate, and tetrakis(triphenylphosphine)palladium(0) in a toluene/ethanol/water solvent system under inert atmosphere.
- Heat the mixture to reflux at 85°C to 90°C for 20 to 24 hours to ensure complete cross-coupling, followed by organic extraction and thorough washing to remove inorganic salts.
- Purify the crude orange-red product via silica gel column chromatography using a petroleum ether and dichloromethane eluent system to isolate the high-purity fluorescent dyad.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this synthetic route offers tangible benefits rooted in process simplicity and raw material availability. The reliance on the Suzuki coupling reaction, a cornerstone of modern organic synthesis, means that the necessary infrastructure and expertise are widely available across the global fine chemical sector, reducing the risk associated with technology transfer. Moreover, the starting materials, specifically the monobrominated cardiocyclene and boronate BODIPY, are either commercially accessible or can be synthesized via established literature methods, ensuring a stable supply chain for reducing lead time for high-purity electronic chemicals.
- Cost Reduction in Manufacturing: The synthetic route eliminates the need for complex multi-step functionalization or the use of precious metal catalysts beyond standard palladium loads, which significantly lowers the raw material cost per kilogram. By achieving yields exceeding 70% through a single coupling step, the process minimizes waste generation and solvent consumption, leading to substantial operational savings compared to traditional methods that require extensive protection-deprotection sequences. This efficiency translates directly into a more competitive pricing structure for the final fluorescent material without compromising on quality.
- Enhanced Supply Chain Reliability: The use of robust reagents like potassium carbonate and tetrakis(triphenylphosphine)palladium(0) ensures that the reaction is tolerant to minor variations in operating conditions, reducing the likelihood of batch failures. Since the reaction can be performed in common solvents like toluene and ethanol, there is no dependency on specialized or hazardous reagents that might face regulatory restrictions or supply shortages. This resilience makes the manufacturing process highly reliable for long-term contracts and large-volume orders required by major display manufacturers.
- Scalability and Environmental Compliance: The workup procedure involves straightforward liquid-liquid extraction and silica gel chromatography, techniques that are easily adaptable to continuous flow reactors or large-scale batch processing. The absence of toxic heavy metal residues, beyond trace palladium which is easily removed, simplifies waste treatment and ensures compliance with stringent environmental regulations. This scalability supports the commercial expansion of production capacity from grams to tons, meeting the growing demand for high-performance solid-state emitters in the optoelectronics industry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of this novel fluorescent dyad, based on the detailed experimental data provided in the patent documentation. These insights are designed to clarify the operational parameters and performance benefits for potential partners evaluating this technology for integration into their product lines.
Q: How does the cardiocyclene unit improve the solid-state fluorescence of BODIPY?
A: The curved, bowl-shaped structure of the cardiocyclene unit introduces significant steric hindrance when covalently bonded to the planar BODIPY core. This geometric distortion effectively prevents tight π-π stacking between molecules in the aggregated solid state, thereby mitigating Aggregation-Caused Quenching (ACQ) and maintaining strong fluorescence emission in solid powder form.
Q: What are the critical reaction conditions for this Suzuki coupling synthesis?
A: The synthesis requires a biphasic solvent system of toluene, ethanol, and water (2:2:1 v/v) to facilitate the interaction between organic substrates and the inorganic base. The reaction must be conducted under an inert nitrogen atmosphere at a controlled reflux temperature between 85°C and 90°C for 20 to 24 hours to achieve optimal conversion yields exceeding 70%.
Q: Is this material suitable for large-scale optoelectronic applications?
A: Yes, the material retains the high absorption coefficient and photochemical stability characteristic of BODIPY dyes while overcoming solid-state quenching. The use of standard Suzuki coupling reagents and commercially available starting materials makes the process highly scalable for industrial production of OLED materials and biological imaging agents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cardiocyclene-BODIPY Dyad Supplier
As a premier CDMO partner, NINGBO INNO PHARMCHEM possesses the technical capability to translate this patented laboratory methodology into a robust industrial process, ensuring consistent quality and supply continuity. Our facilities are equipped with extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, allowing us to handle the specific thermal and mixing requirements of this Suzuki coupling reaction with precision. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Cardiocyclene-BODIPY Dyad meets the exacting standards required for high-end optoelectronic applications, providing our clients with peace of mind regarding material performance.
We invite you to engage with our technical procurement team to discuss how this innovative material can enhance your product portfolio. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits of switching to this superior fluorescent architecture. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your specific volume requirements and application needs.
