Advanced Catalytic Oxidation Strategy for Scalable 4-Acetoxyazetidinone Production
Advanced Catalytic Oxidation Strategy for Scalable 4-Acetoxyazetidinone Production
The pharmaceutical landscape for beta-lactam antibiotics continues to evolve, driven by the urgent need for more efficient and environmentally sustainable manufacturing processes. Patent CN111675734B introduces a groundbreaking preparation method for 4-acetoxyazetidinone (4-AA), a critical chiral intermediate in the synthesis of carbapenem antibiotics such as meropenem and imipenem. This technology represents a significant leap forward in fine chemical synthesis, replacing traditional, hazardous heavy metal catalysts with abundant, low-cost transition metals like cobalt and iron. For R&D directors and procurement specialists alike, this innovation offers a pathway to reduce raw material costs while simultaneously addressing stringent environmental compliance standards. The core of this invention lies in a catalytic oxidation system that operates under mild conditions, utilizing either pre-prepared peracetic acid or an in-situ generated oxidant system involving acetaldehyde and oxygen. By leveraging this novel approach, manufacturers can achieve high yields and excellent stereoselectivity, ensuring the production of high-purity pharmaceutical intermediates essential for the global supply of life-saving antibiotics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4-acetoxyazetidinone has been plagued by significant technical and economic challenges associated with the oxidative acetylation step. Conventional industrial routes frequently depend on stoichiometric amounts or catalytic quantities of precious and toxic transition metals, including mercury (Hg), lead (Pb), ruthenium (Ru), and osmium (Os). These heavy metals not only command exorbitant market prices, driving up the cost of goods sold, but they also introduce severe environmental liabilities. The post-treatment processes required to remove trace heavy metal residues from the final active pharmaceutical ingredient (API) are complex, energy-intensive, and generate substantial volumes of hazardous waste water and solid residues. Furthermore, the handling of these toxic materials necessitates specialized containment infrastructure and rigorous safety protocols, which can bottleneck production capacity and increase operational overhead. For supply chain managers, reliance on such volatile and regulated materials poses a continuous risk of disruption, making the search for a greener alternative not just an ecological preference but a strategic imperative for business continuity.
The Novel Approach
In stark contrast to the legacy methodologies, the technology disclosed in CN111675734B utilizes a catalytic oxidation system driven by earth-abundant metals such as cobalt, iron, copper, or manganese. This shift fundamentally alters the economic and environmental profile of the synthesis. By employing catalysts like cobalt chloride or cobalt acetate, the process achieves remarkable efficiency without the burden of heavy metal toxicity. The reaction is versatile, accommodating different oxidant systems, including direct peracetic acid addition or a safer, in-situ generation method using acetaldehyde and oxygen gas. This flexibility allows manufacturers to optimize their specific production lines for either maximum throughput or maximum safety. The result is a streamlined workflow that eliminates the need for expensive metal scavenging steps and simplifies waste management. For a reliable pharmaceutical intermediate supplier, adopting this methodology translates directly into a more robust and cost-effective supply chain, capable of delivering high-quality intermediates with reduced lead times and lower regulatory friction.
Mechanistic Insights into Cobalt-Catalyzed Oxidative Acetylation
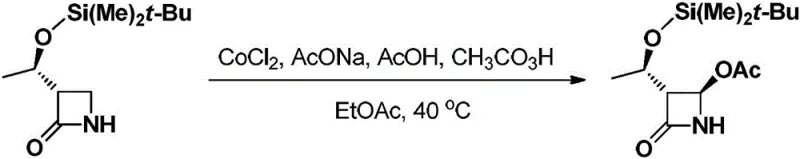
The mechanistic elegance of this process lies in the activation of the oxidant by the low-valent metal center, facilitating a highly selective oxidation of the azetidinone substrate. In the preferred embodiment utilizing cobalt catalysts, the metal coordinates with the oxidant species, likely generating a reactive metal-oxo or radical intermediate that selectively targets the specific carbon-hydrogen bond adjacent to the nitrogen atom. This selectivity is crucial for preserving the integrity of the sensitive beta-lactam ring, which is prone to hydrolysis under harsh acidic or basic conditions. The patent specifies the inclusion of acetic acid and sodium acetate in the reaction mixture, which serves a dual purpose: providing the necessary acidic environment for the oxidation while buffering the pH to approximately 4. This buffering action is a critical control point that prevents the unwanted ring-opening of the lactam, a common side reaction that leads to impurities and yield loss. The ability to maintain high enantioselectivity and diastereoselectivity under these mild conditions (0°C to 40°C) underscores the sophistication of the catalytic cycle, ensuring that the resulting 4-AA possesses the precise stereochemistry required for downstream coupling reactions in carbapenem synthesis.
Furthermore, the option to use an acetaldehyde and oxygen combination for in-situ oxidant generation adds another layer of process safety and control. In this variation, the metal catalyst facilitates the autoxidation of acetaldehyde to peracetic acid directly within the reaction vessel. This eliminates the need to store and transport large quantities of concentrated peracetic acid, which can be unstable and pose explosion risks over time. By generating the oxidant exactly where and when it is needed, the process minimizes thermal runaway risks and allows for tighter control over the reaction kinetics. This mechanistic feature is particularly valuable for scale-up operations, where heat dissipation becomes a limiting factor. The result is a cleaner reaction profile with fewer by-products, simplifying the downstream purification process and enhancing the overall purity of the final crystalline product, which is vital for meeting the stringent specifications of global regulatory bodies.
How to Synthesize 4-Acetoxyazetidinone Efficiently
Implementing this catalytic oxidation strategy requires careful attention to reagent quality and reaction parameters to ensure reproducibility and high yield. The process begins with the preparation of the oxidant, where hydrogen peroxide is reacted with acetic anhydride at low temperatures to form peracetic acid, or alternatively, by setting up an oxygen sparging system with acetaldehyde. The substrate, a protected azetidinone, is then dissolved in a suitable organic solvent such as ethyl acetate, dichloromethane, or toluene. The choice of solvent can influence the solubility of the catalyst and the stability of the intermediates, with ethyl acetate often preferred for its balance of performance and environmental profile. Once the reaction mixture is prepared and buffered with acetic acid and sodium acetate, the catalyst—typically cobalt chloride or acetate—is introduced. The oxidant is then added dropwise under strict temperature control, usually starting at 0°C and gradually warming to 40°C to drive the reaction to completion. Detailed standardized synthesis steps for this procedure are provided below.
- Prepare the oxidant by reacting acetic anhydride with hydrogen peroxide at low temperature to generate peracetic acid, or utilize an acetaldehyde/oxygen system for in-situ generation.
- Combine the azetidinone substrate with a cheap metal catalyst such as cobalt chloride or cobalt acetate in an organic solvent like ethyl acetate.
- Add the oxidant slowly at controlled temperatures between 0°C and 40°C, maintaining a buffered acidic environment with acetic acid and sodium acetate to prevent ring opening.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this catalytic oxidation technology offers transformative advantages that extend far beyond simple yield improvements. The primary driver of value is the drastic reduction in raw material costs associated with replacing precious metals like ruthenium and osmium with commodity chemicals like cobalt and iron salts. This substitution does not merely lower the invoice price of the catalyst; it fundamentally reshapes the cost structure of the entire manufacturing campaign by eliminating the need for specialized heavy metal removal resins and the associated validation testing. Moreover, the simplified waste stream, devoid of toxic heavy metals, significantly reduces the cost of environmental compliance and waste disposal, which are often hidden but substantial expenses in fine chemical manufacturing. The ability to source catalyst materials from a broad, stable global market further insulates the supply chain from geopolitical volatility and price spikes often seen with rare earth elements.
- Cost Reduction in Manufacturing: The elimination of expensive transition metals such as ruthenium and osmium leads to substantial direct savings on bill of materials. Additionally, the simplified workup procedure, which avoids complex metal scavenging steps, reduces labor hours and consumable usage, resulting in a leaner and more cost-efficient production process that enhances overall margin potential.
- Enhanced Supply Chain Reliability: By relying on widely available base metals and common solvents like ethyl acetate, the risk of supply disruption is minimized. The robustness of the reaction conditions, which tolerate a range of temperatures and oxidant concentrations, ensures consistent batch-to-batch quality, allowing for more predictable production scheduling and reliable delivery timelines to downstream API manufacturers.
- Scalability and Environmental Compliance: The mild reaction temperatures and the option for in-situ oxidant generation make this process inherently safer and easier to scale from pilot plant to multi-ton commercial production. The absence of toxic heavy metals simplifies regulatory filings and environmental permitting, accelerating time-to-market for new generic entries and ensuring long-term sustainability of the manufacturing site.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this catalytic oxidation technology for 4-AA production. These insights are derived directly from the experimental data and beneficial effects described in the patent literature, providing a clear understanding of the process capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of technology transfer and for commercial teams assessing the strategic value of this synthetic route.
Q: Why is the new catalytic oxidation method superior to traditional heavy metal processes?
A: Traditional methods often rely on expensive and toxic transition metals like mercury, lead, ruthenium, or osmium, which create significant environmental hazards and disposal costs. The new method utilizes abundant, low-cost metals like cobalt and iron, drastically reducing toxicity and waste treatment burdens while maintaining high stereoselectivity.
Q: What are the optimal reaction conditions for maximizing yield in 4-AA synthesis?
A: The patent data indicates that using cobalt acetate as a catalyst with peracetic acid concentrations of at least 30% yields the best results, often exceeding 90%. The reaction should be conducted in solvents like ethyl acetate at mild temperatures ranging from 0°C to 40°C, with pH buffering to protect the lactam ring.
Q: Can this process be scaled for industrial carbapenem production?
A: Yes, the process is designed for scalability. It avoids hazardous reagents that are difficult to handle on large scales and uses common industrial solvents. The mild temperature requirements and the ability to generate oxidants in situ further enhance its feasibility for commercial-scale manufacturing of penem antibiotics.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Acetoxyazetidinone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to greener, more efficient synthetic routes is critical for the future of the pharmaceutical industry. Our team of expert chemists has extensively evaluated the catalytic oxidation pathways described in CN111675734B and possesses the technical capability to implement this technology at scale. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are realized in practical, high-volume manufacturing. Our facilities are equipped with rigorous QC labs and advanced analytical instrumentation to guarantee that every batch of 4-acetoxyazetidinone meets stringent purity specifications, free from the heavy metal contaminants that plague older synthesis methods.
We invite you to collaborate with us to optimize your supply chain for carbapenem intermediates. By leveraging our expertise in this novel catalytic chemistry, we can help you achieve significant cost efficiencies and improve the environmental footprint of your production. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our advanced manufacturing capabilities can support your long-term strategic goals.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
