Advanced Synthesis of Trifluoromethyl-Triphenylene Discotic Liquid Crystals for Organic Electronics
Advanced Synthesis of Trifluoromethyl-Triphenylene Discotic Liquid Crystals for Organic Electronics
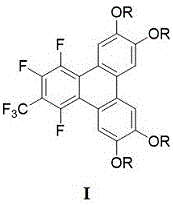
The rapid evolution of organic electronics demands materials with precise molecular architectures capable of efficient charge transport. Patent CN115477572A introduces a groundbreaking preparation method for 1,2,4-trifluoro-6,7,10,11-tetra(alkoxy)-3-trifluoromethyl-triphenylene discotic liquid crystal compounds. These materials represent a significant leap forward in the design of organic semiconductors, specifically targeting applications in organic light-emitting diodes (OLEDs), organic field-effect transistors (OFETs), and photovoltaic cells. The core innovation lies in a streamlined one-pot synthesis that constructs the complex fused-ring aromatic system directly from readily available biphenyl precursors. By leveraging specific organolithium chemistry, this technology enables the formation of stable columnar phases essential for one-dimensional electron and hole transport. For R&D directors and procurement specialists in the electronic chemical sector, this patent outlines a pathway to high-performance materials that balance structural complexity with synthetic feasibility.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for functionalized triphenylene derivatives often suffer from excessive step counts and harsh reaction conditions that hinder commercial scalability. Conventional methods typically rely on oxidative cyclotrimerization of benzene derivatives or multi-step cross-coupling reactions that require expensive transition metal catalysts such as palladium or iron salts. These processes frequently result in complex impurity profiles that are difficult to separate, leading to reduced overall yields and increased production costs. Furthermore, the introduction of specific fluorine substituents to tune liquid crystal properties usually requires additional protection and deprotection steps, further elongating the supply chain timeline. The reliance on heavy metal catalysts also introduces stringent environmental compliance burdens regarding waste disposal and residual metal limits in the final electronic grade product. Such inefficiencies create bottlenecks for manufacturers seeking reliable OLED material suppliers who can deliver consistent quality at scale.
The Novel Approach
The methodology disclosed in the patent data revolutionizes this landscape by employing a direct lithiation-cyclization strategy that drastically simplifies the synthetic route. This novel approach utilizes 2,2'-dibromo-4,4',5,5'-tetraalkoxy-1,1'-biphenyl as the starting scaffold, which undergoes lithium-halogen exchange followed by immediate reaction with perfluorotoluene (C6F5CF3). This one-pot procedure eliminates the need for intermediate isolation and avoids the use of transition metal catalysts entirely. The process operates under relatively mild thermal conditions, ranging from cryogenic cooling at -78 °C to moderate heating at 40 °C, depending on the specific variant employed. By integrating the ring-closing step directly with the functionalization, the method achieves a high degree of atom economy. This streamlined workflow not only reduces the consumption of solvents and reagents but also minimizes the generation of hazardous waste, aligning perfectly with modern green chemistry principles required for cost reduction in electronic chemical manufacturing.
Mechanistic Insights into Organolithium-Mediated Cyclization
The core chemical transformation relies on the precise generation of aryllithium intermediates which act as potent nucleophiles. In the first stage, the dibromo-biphenyl precursor is treated with an alkyllithium base, either n-butyllithium or tert-butyllithium, at low temperatures to ensure selective lithium-halogen exchange without side reactions. This generates a dilithio-species that is highly reactive towards electrophiles. Upon the addition of perfluorotoluene, the electron-deficient aromatic ring of the perfluorotoluene undergoes nucleophilic aromatic substitution. The fluorine atoms on the perfluorotoluene ring serve as excellent leaving groups, facilitating the attack by the aryllithium species. This initiates a cascade of bond formations that ultimately fuse the biphenyl unit with the toluene derivative to form the rigid triphenylene core. The presence of the trifluoromethyl group is retained from the perfluorotoluene starting material, providing the specific electronic modulation required for the material's semiconducting properties.
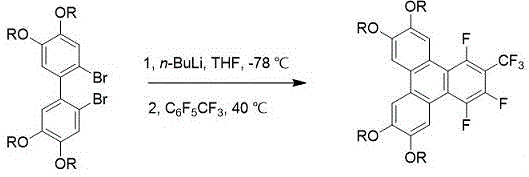
Controlling the stoichiometry and temperature is critical for managing the impurity profile and maximizing yield. The patent specifies a molar ratio of 1:4 for the biphenyl substrate to the alkyllithium reagent, ensuring complete conversion of both bromine sites to lithium species. Subsequent reaction with perfluorotoluene is carefully managed, with ratios varying between 1:3 and 1:1.5 depending on the specific base and solvent system used. The use of tetrahydrofuran (THF) versus diethyl ether significantly influences the aggregation state of the organolithium species, thereby affecting reaction kinetics. For instance, the method utilizing tert-butyllithium in diethyl ether allows the final cyclization to proceed at room temperature, whereas the n-butyllithium method in THF requires heating to 40 °C. This mechanistic flexibility allows process chemists to optimize the reaction based on available infrastructure and safety considerations, ensuring robust production of high-purity OLED material.
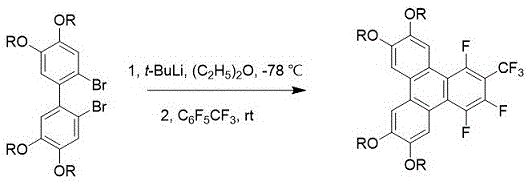
How to Synthesize Trifluoromethyl-Triphenylene Efficiently
The synthesis protocol detailed in the patent offers two distinct yet complementary pathways to access the target discotic liquid crystals, providing flexibility for different manufacturing environments. Both methods begin with the dissolution of the dibromo-biphenyl precursor in an anhydrous solvent under an inert nitrogen atmosphere to prevent moisture sensitivity issues inherent to organolithium chemistry. The reaction mixture is cooled to -78 °C to control the exothermic lithiation step before the base is introduced. Following the initial lithiation and warming period, the perfluorotoluene electrophile is added, and the mixture is allowed to react for an extended period to ensure complete ring closure. The workup involves standard aqueous extraction and drying, followed by purification via column chromatography and recrystallization to achieve the requisite electronic grade purity. Detailed standardized synthesis steps see the guide below.
- Dissolve 2,2'-dibromo-4,4',5,5'-tetraalkoxy-1,1'-biphenyl in anhydrous solvent (THF or Diethyl Ether) under nitrogen protection and cool to -78 °C.
- Add alkyllithium reagent (n-BuLi or t-BuLi) dropwise at low temperature, maintain stirring for 30 minutes, then warm to room temperature or 40 °C for initial reaction.
- Re-cool if necessary, inject perfluorotoluene (C6F5CF3), react at controlled temperatures (40 °C or RT) for 12 hours, then purify via column chromatography and recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible strategic benefits beyond mere technical performance. The elimination of transition metal catalysts removes a significant cost driver associated with precious metal recovery and residual metal testing, which is critical for electronic applications. Furthermore, the shortened synthetic sequence reduces the total processing time and equipment occupancy, effectively increasing throughput capacity without requiring additional capital investment. The use of commodity chemicals like perfluorotoluene and standard alkyllithium reagents ensures a stable and reliable supply chain, mitigating risks associated with specialized or scarce raw materials. This stability is crucial for maintaining continuous production schedules and meeting the demanding delivery timelines of the display and semiconductor industries.
- Cost Reduction in Manufacturing: The one-pot nature of this synthesis significantly lowers operational expenditures by consolidating multiple reaction steps into a single vessel. This consolidation reduces solvent consumption, energy usage for heating and cooling cycles, and labor hours required for intermediate handling and purification. By avoiding expensive palladium or nickel catalysts, the raw material cost profile is drastically improved, allowing for more competitive pricing structures in the final market. Additionally, the simplified workup procedure minimizes the volume of chemical waste generated, leading to substantial cost savings in waste treatment and environmental compliance management.
- Enhanced Supply Chain Reliability: The starting materials, specifically the alkoxy-substituted dibromobiphenyls and perfluorotoluene, are commercially available from multiple global suppliers, reducing dependency on single-source vendors. This diversification enhances supply security and provides leverage in price negotiations. The robustness of the reaction conditions, particularly the room temperature variant using tert-butyllithium, reduces the risk of batch failures due to equipment malfunction or temperature control issues. Consequently, this reliability translates into consistent lead times for high-purity electronic chemicals, enabling downstream manufacturers to plan their production cycles with greater confidence and accuracy.
- Scalability and Environmental Compliance: The mild reaction temperatures and absence of high-pressure requirements make this process inherently safer and easier to scale from laboratory to commercial production volumes. The use of common solvents like THF and diethyl ether simplifies solvent recovery and recycling operations, further enhancing the sustainability profile of the manufacturing process. The absence of heavy metals in the reaction mixture simplifies the regulatory approval process for new materials, as there is no need for extensive toxicological screening related to metal residues. This facilitates faster time-to-market for new electronic devices incorporating these advanced liquid crystal materials.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these trifluoromethyl-triphenylene derivatives. The answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, ensuring accuracy and relevance for industry stakeholders. Understanding these nuances is essential for evaluating the feasibility of integrating this technology into existing manufacturing workflows.
Q: What are the key advantages of the t-BuLi method over the n-BuLi method?
A: The t-BuLi method utilizes diethyl ether as a solvent and operates at room temperature for the final cyclization step, offering milder conditions and significantly higher yields compared to the n-BuLi method which requires THF and heating to 40 °C.
Q: What is the liquid crystal phase range for these compounds?
A: The synthesized compounds exhibit a wide columnar liquid crystal phase range. For example, the butoxy derivative shows a range of 62-223 °C, while the octyloxy derivative displays a range of -17-153 °C, making them suitable for various operating temperatures.
Q: Why is perfluorotoluene used in this synthesis?
A: Perfluorotoluene acts as the electrophilic coupling partner that introduces the trifluoromethyl group and completes the triphenylene core structure through nucleophilic aromatic substitution following the lithiation step.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoromethyl Triphenylene Supplier
As the demand for high-performance organic semiconductors continues to surge, partnering with an experienced CDMO is vital for successful commercialization. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our rigorous QC labs and adherence to stringent purity specifications guarantee that every batch of trifluoromethyl-triphenylene liquid crystals meets the exacting standards required for next-generation display and optoelectronic applications. We are committed to delivering materials that enable superior device performance while maintaining cost efficiency throughout the supply chain.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this catalytic-free methodology. Contact us today to obtain specific COA data and route feasibility assessments, and let us support your journey towards developing cutting-edge electronic materials with reliable quality and competitive pricing.
