Scaling Green Electrochemical C-H Activation for High-Purity Pharmaceutical Intermediates
Introduction to Patent CN111270260B: A Paradigm Shift in C-H Functionalization
The landscape of organic synthesis is undergoing a transformative shift towards sustainability, driven by the urgent need to reduce chemical waste and energy consumption in the manufacturing of complex molecules. Patent CN111270260B introduces a groundbreaking methodology for the ortho-alkenylation of aromatic amide compounds, leveraging electrochemical oxidation to drive Rhodium-catalyzed C-H activation. This innovation represents a significant departure from traditional stoichiometric approaches, replacing hazardous chemical oxidants with clean electrical current. For R&D directors and process chemists, this technology offers a robust pathway to access valuable Heck-type products with high regioselectivity and yield, while simultaneously addressing critical environmental and cost constraints inherent in conventional transition metal catalysis.
The core breakthrough lies in the seamless integration of electrochemistry with organometallic catalysis, enabling the efficient coupling of aromatic amides with non-activated olefins in an aqueous medium. By utilizing a weak current as the sole oxidant, the process not only minimizes the generation of toxic byproducts but also enhances the atom economy of the reaction. This patent details a comprehensive protocol that ensures high conversion rates and operational simplicity, making it an attractive candidate for the industrial scale-up of pharmaceutical intermediates and fine chemicals where purity and environmental compliance are paramount.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for directing group-assisted C-H alkenylation typically rely heavily on the use of stoichiometric amounts of inorganic or organic oxidants, such as silver salts, copper salts, or hypervalent iodine reagents. These conventional protocols often necessitate harsh reaction conditions and generate substantial quantities of metal-containing waste, posing significant challenges for waste treatment and environmental compliance in large-scale manufacturing. Furthermore, the reliance on expensive oxidants drastically increases the raw material costs, while the presence of residual metals in the product stream complicates downstream purification, often requiring additional scavenging steps that reduce overall process efficiency and yield.
The Novel Approach
In stark contrast, the novel electrochemical strategy described in the patent utilizes electricity as a traceless and renewable oxidant to regenerate the active Rh(III) catalyst from the reduced Rh(I) species formed after beta-hydride elimination. This approach effectively closes the catalytic cycle without the need for external chemical oxidants, thereby eliminating the associated waste streams and cost burdens. The reaction proceeds smoothly in a mixed aqueous solvent system, which provides high conductivity without the need for additional supporting electrolytes, further simplifying the reaction setup and workup procedures.
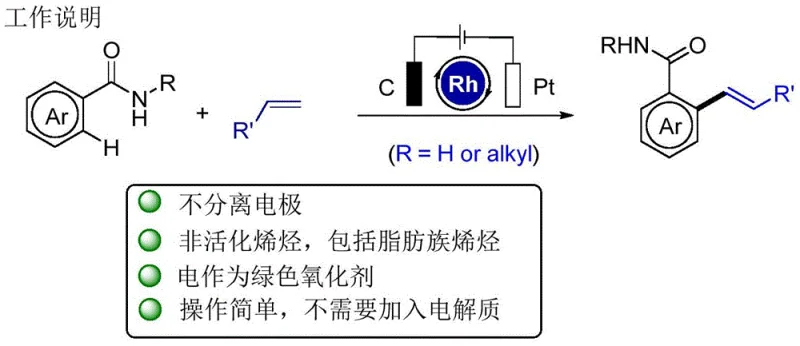
The versatility of this electrochemical protocol is demonstrated by its compatibility with a wide range of substrates, including those bearing sensitive functional groups that might be incompatible with strong chemical oxidants. The use of a graphite felt anode and a platinum cathode in an undivided cell setup ensures efficient electron transfer and stable reaction performance. This method not only achieves high yields and excellent regioselectivity but also aligns perfectly with the principles of green chemistry, offering a sustainable alternative for the synthesis of complex molecular scaffolds required in the pharmaceutical and agrochemical industries.
Mechanistic Insights into Electrochemical Rh(III)-Catalyzed C-H Activation
The mechanistic pathway of this transformation involves a sophisticated interplay between electrochemical oxidation and organometallic catalysis, centered around the Rhodium catalyst. Initially, the Rh(III) species coordinates with the directing group of the aromatic amide to facilitate C-H bond activation, forming a stable five-membered rhodacycle intermediate. This step is crucial for establishing the regioselectivity of the reaction, ensuring that the alkenylation occurs specifically at the ortho-position relative to the amide group. The presence of carboxylate additives, such as sodium pivalate, plays a vital role in assisting this C-H cleavage step by acting as a base and stabilizing the metal center.
Following C-H activation, the coordinated olefin undergoes migratory insertion into the Rh-C bond, extending the carbon skeleton and forming a new C-C bond. Subsequent beta-hydride elimination releases the desired alkenylated product and generates a Rh(I) species. In traditional thermal catalysis, this Rh(I) species would require a chemical oxidant to return to the active Rh(III) state. However, in this electrochemical system, the Rh(I) intermediate is efficiently re-oxidized to Rh(III) at the anode surface through the loss of electrons. This anodic oxidation step is the key to the catalytic turnover, allowing the cycle to continue with minimal catalyst loading and no chemical oxidant waste.
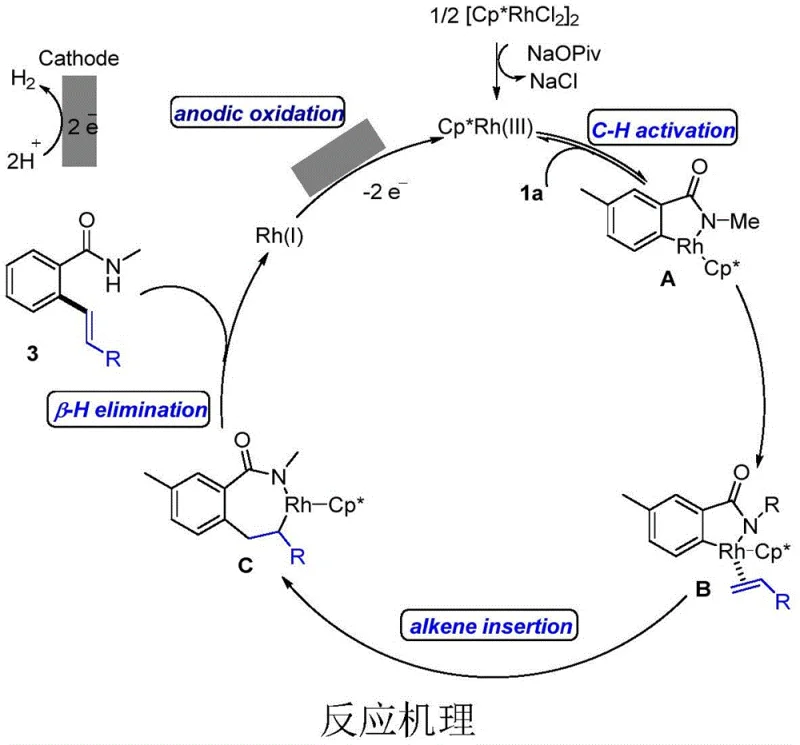
This mechanism highlights the elegance of using electricity to drive redox-neutral transformations that are otherwise dependent on stoichiometric reagents. The ability to control the oxidation potential via the applied current allows for fine-tuning of the reaction kinetics, preventing over-oxidation of sensitive substrates. Furthermore, the aqueous reaction medium enhances the stability of the ionic intermediates and facilitates proton management during the beta-hydride elimination and subsequent proton reduction at the cathode, where hydrogen gas is evolved as the only byproduct. This deep understanding of the catalytic cycle provides a solid foundation for optimizing reaction conditions and expanding the scope of applicable substrates.
How to Synthesize Ortho-Alkenylated Aromatic Amides Efficiently
The practical implementation of this electrochemical synthesis is straightforward and relies on standard laboratory equipment adapted for electrolysis. The general procedure involves dissolving the aromatic amide substrate and the olefin coupling partner in a mixture of tert-amyl alcohol and water, which serves as both the solvent and the conductive medium. Sodium pivalate is added as an essential additive to promote C-H activation, along with a catalytic amount of the dimeric rhodium precursor [Cp*RhCl2]2. The reaction is conducted in an undivided cell equipped with a graphite felt anode and a platinum sheet cathode, maintaining a constant current under heated conditions to ensure complete conversion.
- Dissolve the aromatic amide substrate, non-activated olefin, and NaOPiv additive in a t-AmOH/H2O solvent mixture within an undivided electrochemical cell equipped with a graphite felt anode and platinum cathode.
- Apply a constant current of approximately 4 mA while heating the reaction mixture to 100°C under air atmosphere for 12 to 24 hours to facilitate the Rh-catalyzed C-H activation and olefin insertion.
- Upon completion, remove solvents under vacuum, adsorb the residue onto silica gel, and purify the crude product via column chromatography using an EtOAc/n-hexane eluent system to isolate the target Heck-type product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this electrochemical technology presents compelling opportunities for cost optimization and risk mitigation. The elimination of stoichiometric chemical oxidants removes a significant variable cost component from the bill of materials, while also simplifying the supply chain by reducing the number of specialized reagents required. The use of earth-abundant solvents like water and tert-amyl alcohol further drives down material costs compared to exotic organic solvents, and the simplified workup procedure reduces the consumption of silica gel and eluents during purification, leading to substantial overall savings in manufacturing expenses.
- Cost Reduction in Manufacturing: The primary economic driver of this technology is the replacement of expensive silver or copper oxidants with electricity, which is significantly cheaper and more readily available. By minimizing the usage of precious rhodium catalyst through efficient electrochemical recycling, the process lowers the threshold for metal contamination in the final product, potentially reducing the cost and complexity of metal scavenging steps. Additionally, the high atom economy and reduced waste generation translate directly into lower waste disposal fees and a smaller environmental footprint, enhancing the overall profitability of the manufacturing process.
- Enhanced Supply Chain Reliability: The reliance on simple, commodity-grade reagents such as aromatic amides, styrenes, and sodium pivalate ensures a robust and resilient supply chain that is less susceptible to disruptions compared to processes requiring specialized or hazardous oxidants. The operational simplicity of the electrochemical setup, which does not require inert atmosphere techniques or complex pressure vessels, facilitates easier technology transfer and scale-up across different manufacturing sites. This reliability ensures consistent production schedules and reduces the lead time for delivering high-purity pharmaceutical intermediates to downstream customers.
- Scalability and Environmental Compliance: The aqueous nature of the reaction medium inherently improves safety profiles by reducing flammability risks associated with volatile organic solvents, making the process easier to scale from gram to kilogram and ton scales. The absence of heavy metal waste streams simplifies regulatory compliance and environmental permitting, as the effluent treatment becomes less burdensome. This alignment with green chemistry principles not only future-proofs the manufacturing process against tightening environmental regulations but also enhances the brand value of the final products in markets that prioritize sustainability and ethical sourcing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the electrochemical ortho-alkenylation process, providing clarity on its operational parameters and strategic benefits. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, ensuring accuracy and relevance for decision-makers evaluating this technology for adoption.
Q: What are the primary advantages of using electrochemical oxidation over traditional chemical oxidants in this C-H activation?
A: The electrochemical approach eliminates the need for stoichiometric amounts of expensive and waste-generating chemical oxidants such as silver or copper salts. By using electrons as the terminal oxidant, the process significantly reduces metal waste, lowers raw material costs, and simplifies the downstream purification process, resulting in a greener and more economically viable manufacturing route for pharmaceutical intermediates.
Q: Is the rhodium catalyst loading efficient for large-scale production?
A: Yes, the protocol utilizes a low catalyst loading of approximately 2.5 mol% of [Cp*RhCl2]2. The electrochemical regeneration of the active Rh(III) species from the Rh(I) intermediate allows for a sustained catalytic cycle, minimizing the total amount of precious metal required and reducing the burden of heavy metal removal in the final API product.
Q: Does this method support a broad range of substrate functional groups?
A: The method demonstrates excellent functional group tolerance, accommodating both electron-donating and electron-withdrawing substituents on the aromatic ring. It is compatible with various heterocycles and non-activated olefins, making it a versatile platform for synthesizing diverse libraries of ortho-alkenylated compounds essential for drug discovery and process development.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ortho-Alkenylated Aromatic Amide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of electrochemical C-H activation in modernizing the synthesis of complex pharmaceutical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like this are seamlessly translated into robust industrial processes. Our state-of-the-art facilities are equipped with advanced electrochemical reactors and rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of ortho-alkenylated aromatic amide delivered meets the highest quality standards required by global regulatory bodies.
We invite forward-thinking pharmaceutical companies and chemical manufacturers to collaborate with us to leverage this green synthesis technology for their specific pipeline needs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your target molecule, along with specific COA data and route feasibility assessments. Let us help you optimize your supply chain, reduce your environmental impact, and accelerate your time-to-market with our expert guidance and reliable manufacturing capabilities.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
