Scalable Enzymatic Production of High-Purity (S)-1-(4-Chlorophenyl)-1,3-Propanediol for Pharma
The pharmaceutical industry is constantly seeking more efficient and sustainable pathways for synthesizing critical chiral intermediates, particularly those used in liver-targeting antiviral therapies. Patent CN113816836A introduces a groundbreaking enzymatic production method for (S)-1-(4-chlorophenyl)-1,3-propanediol, a key building block in the synthesis of advanced prodrugs like tenofovir alafenamide. This technology leverages specific carbonyl reductase mutants to achieve exceptional stereoselectivity and operational safety, addressing long-standing bottlenecks in traditional chemical synthesis. By replacing harsh chemical reagents with biocatalysts, this route offers a compelling value proposition for R&D directors and procurement managers alike, ensuring high purity while mitigating supply chain risks associated with complex chemical handling. The strategic implementation of this enzymatic pathway represents a significant leap forward in the manufacturing of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
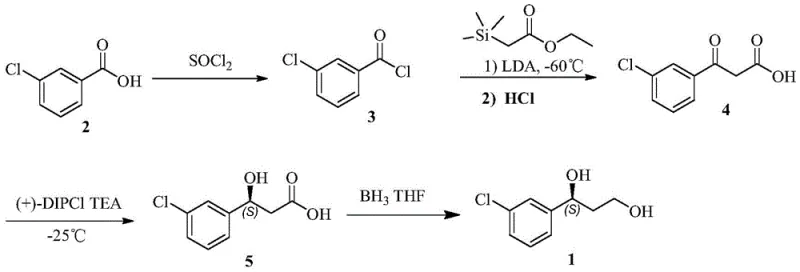
Traditional chemical synthesis routes for this diol intermediate, as documented in prior art such as US20030225277, rely heavily on cryogenic conditions and hazardous reagents that pose significant challenges for industrial scale-up. The conventional pathway typically necessitates ultra-low temperature operations, often reaching minus 60 degrees Celsius, which demands specialized and energy-intensive cooling infrastructure that drastically increases capital expenditure. Furthermore, these methods frequently employ expensive chiral catalysts like (+)-DIPCI and utilize borane for reduction steps, which introduces severe safety risks due to violent exothermic quenching reactions. The resulting chiral purity from these chemical routes often hovers around 98 percent, necessitating additional purification steps such as recrystallization to meet stringent regulatory standards for clinical applications. These cumulative factors create a high-cost, high-risk production environment that is increasingly unsustainable for modern supply chains.
The Novel Approach
In stark contrast, the novel enzymatic approach disclosed in the patent utilizes engineered carbonyl reductases to perform the critical chiral reduction under mild, ambient conditions. This biocatalytic strategy eliminates the need for cryogenic temperatures, operating efficiently between 30 to 35 degrees Celsius, which significantly reduces energy consumption and equipment complexity. By employing specific enzyme mutants, the process achieves a chiral purity exceeding 99.6 percent directly from the reaction, minimizing downstream processing requirements and waste generation. The substitution of hazardous borane reagents with benign enzymatic systems not only enhances workplace safety but also simplifies waste treatment protocols, aligning with green chemistry principles. This shift from chemocatalysis to biocatalysis provides a robust foundation for cost reduction in pharmaceutical intermediate manufacturing while ensuring consistent quality.
Mechanistic Insights into Carbonyl Reductase-Catalyzed Reduction
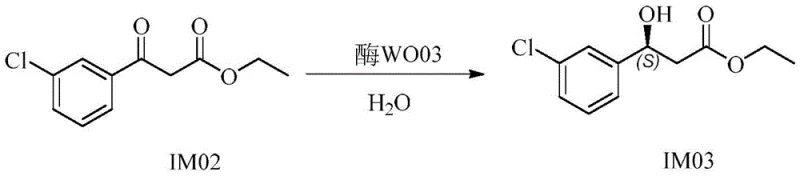
The core of this technological advancement lies in the precise engineering of carbonyl reductase enzymes, specifically the wild-type WO03 and its site-directed mutants. The patent details specific amino acid mutations, such as V45I, G141A, and I195L, which enhance the enzyme's active site geometry to favor the formation of the (S)-enantiomer with exceptional specificity. These mutations improve the binding affinity for the ketoester substrate, facilitating a highly efficient hydride transfer from the cofactor NADPH to the carbonyl group. The enzymatic cycle is sustained through a coenzyme regeneration system, often utilizing glucose dehydrogenase or formate dehydrogenase, which ensures continuous catalytic turnover without the need for stoichiometric amounts of expensive cofactors. This mechanistic efficiency allows for high substrate loading concentrations, up to 200 grams per liter, making the process viable for high-throughput industrial bioreactors.
Impurity control is inherently superior in this enzymatic system due to the high stereoselectivity of the biocatalyst, which effectively suppresses the formation of the unwanted (R)-enantiomer and other side products. The mild pH conditions, maintained between 6.5 and 7.5, prevent acid or base-catalyzed degradation of the sensitive ester and alcohol functionalities present in the intermediate. Furthermore, the aqueous nature of the biotransformation medium reduces the solubility of many organic impurities that typically plague organic solvent-based chemical reactions. The subsequent hydrolysis and reduction steps are designed to preserve this high purity, utilizing controlled alkaline hydrolysis and mild borohydride reduction conditions that do not compromise the chiral center. This comprehensive control over the reaction environment ensures that the final product meets rigorous quality specifications without extensive chromatographic purification.
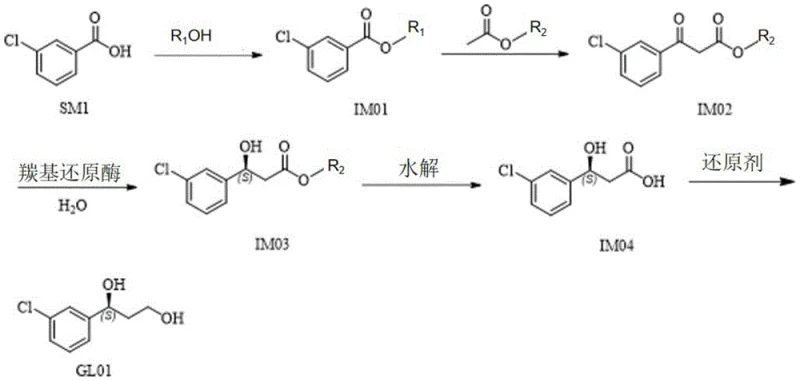
How to Synthesize (S)-1-(4-Chlorophenyl)-1,3-Propanediol Efficiently
The synthesis begins with the esterification of m-chlorobenzoic acid followed by a Claisen condensation to generate the ketoester substrate for the biocatalytic step. The critical enzymatic reduction is then performed using whole cells or crude enzyme preparations containing the engineered carbonyl reductase mutants. Following the biotransformation, the chiral ester undergoes hydrolysis to the corresponding acid, which is finally reduced to the target diol. The detailed standardized synthesis steps see the guide below.
- Perform esterification of m-chlorobenzoic acid with alcohol to form intermediate IM01.
- Conduct Claisen condensation with alkyl acetate to generate ketoester IM02.
- Execute key enzymatic reduction using carbonyl reductase mutants to obtain chiral alcohol IM03 with >99.6% ee.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, this enzymatic technology offers substantial strategic advantages by de-risking the supply of critical pharmaceutical intermediates. The elimination of ultra-low temperature requirements and hazardous reagents translates directly into lower operational costs and reduced dependency on specialized infrastructure, making the supply chain more resilient and flexible. The high yield and purity achieved in the enzymatic step minimize material loss and waste disposal costs, contributing to significant cost savings in manufacturing without compromising quality. Additionally, the use of robust enzyme mutants ensures consistent batch-to-batch performance, which is crucial for maintaining regulatory compliance and avoiding production delays. This reliability enhances supply chain continuity, allowing partners to plan long-term production schedules with greater confidence.
- Cost Reduction in Manufacturing: The transition to enzymatic catalysis removes the need for expensive chiral chemical catalysts and cryogenic cooling systems, leading to a drastic simplification of the production process. By avoiding the use of hazardous borane reagents in the key chiral step, the costs associated with safety management, specialized equipment, and waste treatment are significantly reduced. The high conversion rates and stereoselectivity of the enzyme mutants minimize raw material consumption and downstream purification needs, further driving down the overall cost of goods sold. These efficiencies create a more competitive pricing structure for the final intermediate, offering tangible value to downstream drug manufacturers.
- Enhanced Supply Chain Reliability: The mild reaction conditions and use of stable enzyme preparations reduce the risk of batch failures due to equipment malfunction or environmental fluctuations. Sourcing of raw materials is simplified as the process avoids specialized low-temperature reagents that may have limited availability or long lead times. The scalability of the fermentation-based enzyme production ensures a consistent supply of the biocatalyst, preventing bottlenecks that often occur with complex chemical catalysts. This stability allows for more predictable delivery schedules and strengthens the overall reliability of the supply chain for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The aqueous-based biotransformation process generates less hazardous waste compared to traditional organic synthesis, facilitating easier compliance with increasingly strict environmental regulations. The ability to operate at high substrate concentrations and ambient temperatures makes the process highly scalable from pilot plant to commercial production volumes without significant re-engineering. Reduced solvent usage and energy consumption align with sustainability goals, enhancing the corporate social responsibility profile of the manufacturing partner. This environmental compatibility ensures long-term operational viability and reduces the risk of regulatory shutdowns or fines.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this enzymatic synthesis route. These answers are derived directly from the patent data and practical manufacturing considerations to provide clarity for potential partners. Understanding these details is essential for evaluating the feasibility of integrating this technology into existing supply chains.
Q: How does the enzymatic route improve chiral purity compared to chemical synthesis?
A: The enzymatic route using carbonyl reductase mutants achieves over 99.6% ee, surpassing the 98% purity of conventional chemical methods, reducing the need for costly recrystallization.
Q: What are the safety advantages of this production method?
A: This method eliminates the need for hazardous borane reduction in the chiral step and avoids ultra-low temperature conditions (-60°C), significantly enhancing operational safety.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process operates at mild temperatures (30-35°C) and uses robust enzyme mutants, making it highly adaptable for industrial scale-up from kilograms to metric tons.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S)-1-(4-Chlorophenyl)-1,3-Propanediol Supplier
NINGBO INNO PHARMCHEM stands at the forefront of adopting such advanced enzymatic technologies to deliver superior pharmaceutical intermediates to the global market. Our CDMO experts possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative lab-scale routes are successfully translated into robust industrial processes. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of (S)-1-(4-chlorophenyl)-1,3-propanediol meets the highest quality standards required for clinical and commercial drug manufacturing. Our commitment to technical excellence ensures that your supply chain is supported by a partner who understands the critical nature of chiral intermediates.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs through the adoption of this efficient enzymatic route. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities can support your project timelines and quality goals. Let us engineer the solution for your production needs.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
