Revolutionizing Triazole Intermediate Production with Iron-Catalyzed Cyclization for Global Pharma Supply Chains
Revolutionizing Triazole Intermediate Production with Iron-Catalyzed Cyclization for Global Pharma Supply Chains
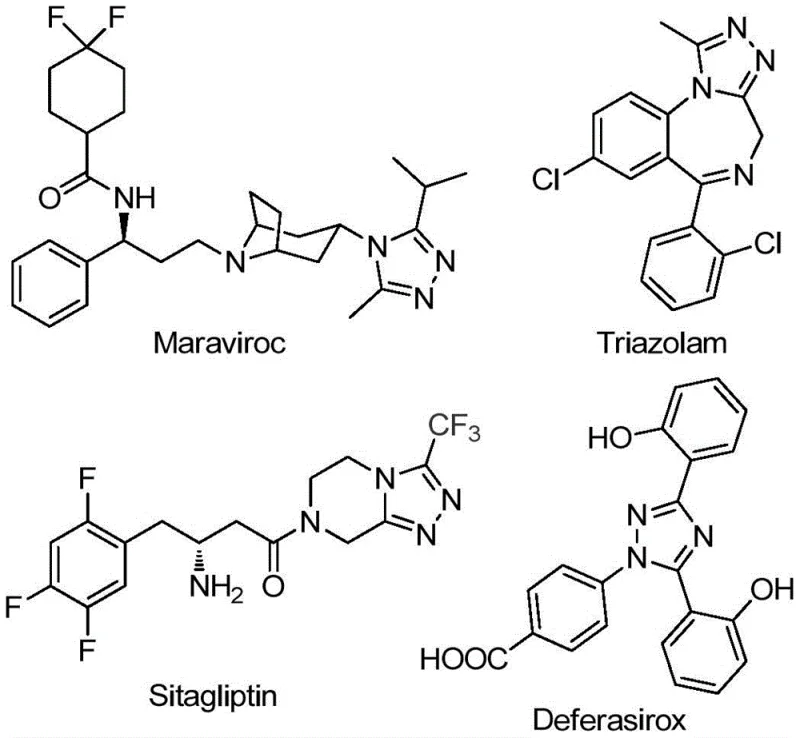
The pharmaceutical and agrochemical industries are constantly seeking robust, scalable, and cost-effective methodologies for constructing nitrogen-containing heterocycles, particularly those bearing trifluoromethyl groups which impart superior metabolic stability and lipophilicity to drug candidates. Patent CN111978265B, published in July 2022, introduces a groundbreaking preparation method for 5-trifluoromethyl substituted 1,2,4-triazole derivatives that addresses long-standing synthetic challenges. This technology leverages a dual-stage catalytic system involving sodium bicarbonate and ferric chloride to facilitate the cyclization of readily available hydrazides and trifluoroethylimide chlorides. For R&D directors and procurement managers alike, this innovation represents a pivotal shift away from complex, multi-step legacy processes toward a streamlined, operationally simple protocol that does not require stringent anhydrous or oxygen-free environments. The ability to access these privileged scaffolds efficiently is crucial for the development of next-generation therapeutics similar to Maraviroc and Sitagliptin.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of trifluoromethyl-substituted 1,2,4-triazoles has been plagued by significant technical hurdles that hinder efficient commercial manufacturing. Traditional literature methods often rely on the condensation of 3,5-ditrifluoromethyl-1,3,4-oxadiazoles with primary amines or the cyclization of trifluoromethyl hydrazides with amidines. These conventional pathways are frequently characterized by harsh reaction conditions, requiring extreme temperatures or pressures that pose safety risks and increase energy consumption. Furthermore, many existing protocols suffer from narrow substrate scopes, failing to accommodate alkyl hydrazones effectively, which limits the chemical diversity accessible to medicinal chemists. The necessity for lengthy reaction steps and the frequent occurrence of low reaction yields further exacerbate the cost burden, making these methods less attractive for large-scale API intermediate production where margin compression is a constant concern.
The Novel Approach
In stark contrast to these legacy techniques, the novel approach detailed in the patent utilizes a simple yet highly effective strategy starting from cheap and easily obtainable acyl hydrazides and trifluoroethylimide chlorides. This method employs ferric chloride as a promoter to drive the cyclization, operating under relatively mild conditions that do not demand specialized inert atmosphere equipment. The process is divided into two distinct thermal stages: an initial base-promoted reaction followed by a Lewis acid-promoted cyclization, ensuring high conversion rates. By avoiding the need for expensive transition metal catalysts like palladium or rhodium, and eliminating the requirement for rigorous drying of solvents, this new route drastically simplifies the operational workflow. The versatility of this approach allows for the facile synthesis of 3,4-disubstituted 1,2,4-triazole derivatives with various functional groups, providing a powerful tool for rapid library generation in drug discovery programs.
Mechanistic Insights into FeCl3-Catalyzed Cyclization
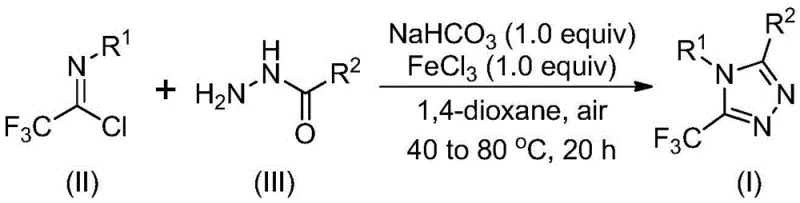
The mechanistic pathway of this transformation offers fascinating insights into how simple Lewis acids can orchestrate complex heterocycle formation with high fidelity. The reaction is hypothesized to proceed through an initial base-promoted intermolecular carbon-nitrogen bond formation between the trifluoroethylimide chloride and the hydrazide, generating a trifluoroacetamidine derivative intermediate. This step is critical as it sets the stage for the subsequent ring closure without generating excessive acidic byproducts that could degrade sensitive functional groups. Following this, the addition of ferric chloride acts as a potent Lewis acid promoter, facilitating an intramolecular dehydration condensation reaction. This second stage effectively drives the equilibrium towards the formation of the final 5-trifluoromethyl substituted 1,2,4-triazole core. The use of iron, an earth-abundant metal, ensures that the catalytic cycle is not only effective but also environmentally benign compared to heavy metal alternatives.
From an impurity control perspective, this mechanism provides distinct advantages for maintaining high product purity, a key metric for regulatory compliance in pharmaceutical manufacturing. The mild nature of the sodium bicarbonate base prevents the hydrolysis of the imidoyl chloride starting material, a common side reaction in more basic environments. Additionally, the specific activation by ferric chloride minimizes non-specific polymerization or decomposition of the trifluoromethyl group, which can be susceptible to defluorination under harsher conditions. The result is a cleaner reaction profile that simplifies downstream purification, often requiring only standard filtration and silica gel chromatography. This level of control over the reaction trajectory ensures that the final API intermediates meet stringent quality specifications, reducing the risk of batch failures and ensuring consistent supply for clinical and commercial needs.
How to Synthesize 5-Trifluoromethyl-1,2,4-Triazole Derivatives Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to the sequential addition of reagents and temperature control to maximize yield and selectivity. The process begins by dissolving the starting materials in a suitable aprotic solvent, with 1,4-dioxane identified as the preferred medium due to its ability to solubilize all reactants effectively while promoting high conversion. The initial heating phase allows for the formation of the key intermediate, after which the introduction of the iron catalyst triggers the final cyclization. Detailed standardized operating procedures regarding stoichiometry, specifically the molar ratios of hydrazide to imidoyl chloride, are critical for optimizing the economic efficiency of the process. For a comprehensive breakdown of the exact experimental parameters and workup procedures, please refer to the technical guide below.
- Mix sodium bicarbonate, trifluoroethylimide chloride, and hydrazide in an organic solvent like 1,4-dioxane.
- Heat the mixture to 30-50°C and react for 8-16 hours to form the intermediate.
- Add ferric chloride and increase temperature to 70-90°C for 6-10 hours to complete cyclization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology translates directly into tangible operational efficiencies and risk mitigation strategies. The shift away from exotic reagents and complex reaction conditions means that the barrier to entry for manufacturing these valuable intermediates is significantly lowered. By utilizing commodity chemicals like ferric chloride and sodium bicarbonate, manufacturers can decouple their supply chains from volatile markets for precious metals. This stability is crucial for maintaining continuous production schedules and meeting the just-in-time delivery demands of global pharmaceutical clients. Furthermore, the simplified post-processing requirements reduce the load on waste treatment facilities and lower the overall environmental footprint of the manufacturing site.
- Cost Reduction in Manufacturing: The elimination of expensive noble metal catalysts and the removal of strict anhydrous requirements lead to substantial cost savings in raw material procurement and facility operations. Traditional methods often necessitate glovebox operations or extensive solvent drying systems, which represent significant capital and operational expenditures. By contrast, this iron-catalyzed process can be run in standard reactors with minimal preprocessing of solvents, drastically reducing energy consumption and labor costs associated with setup and teardown. The high atom economy and improved yields further contribute to a lower cost of goods sold, allowing for more competitive pricing in the global marketplace.
- Enhanced Supply Chain Reliability: The reliance on widely available, commodity-grade starting materials ensures a robust and resilient supply chain that is less susceptible to geopolitical disruptions or shortages. Acyl chlorides and hydrazines are produced at massive scales for various industries, guaranteeing a steady flow of inputs for triazole synthesis. This availability allows manufacturers to maintain larger safety stocks without incurring prohibitive costs, thereby enhancing their ability to fulfill large volume orders on short notice. The robustness of the reaction conditions also means that production can be easily transferred between different manufacturing sites without the need for highly specialized equipment, adding another layer of security to the supply network.
- Scalability and Environmental Compliance: The demonstrated scalability of this method from milligram to gram scales indicates a clear path toward ton-level commercial production without encountering unforeseen engineering bottlenecks. The use of iron salts, which are generally regarded as safe and environmentally friendly, simplifies waste disposal and aligns with increasingly stringent green chemistry regulations. Unlike processes generating heavy metal waste streams that require costly remediation, the effluent from this process is easier to treat, reducing the environmental compliance burden. This alignment with sustainability goals not only lowers operational costs but also enhances the corporate social responsibility profile of the manufacturing organization.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. Understanding these details is essential for project managers evaluating the feasibility of integrating this route into their existing portfolios. The answers are derived directly from the experimental data and theoretical framework provided in the patent documentation, ensuring accuracy and reliability for decision-making purposes. These insights cover aspects ranging from substrate compatibility to purification strategies, providing a holistic view of the technology's capabilities.
Q: What are the key advantages of the FeCl3-catalyzed method over traditional oxadiazole routes?
A: The FeCl3-catalyzed method eliminates the need for harsh anhydrous and oxygen-free conditions required by traditional methods, significantly simplifying operational complexity and reducing equipment costs.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the patent explicitly demonstrates scalability to the gram level with simple post-processing steps like filtration and column chromatography, indicating strong potential for commercial scale-up.
Q: What is the substrate scope for R1 and R2 groups in this triazole synthesis?
A: The method exhibits broad substrate tolerance, accommodating substituted or unsubstituted aryl groups for R1 and alkyl, alkenyl, or aryl groups for R2, allowing for diverse derivative synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl-1,2,4-Triazole Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality heterocyclic intermediates play in the development of life-saving medications and advanced agrochemicals. Our team of expert chemists has extensively evaluated the FeCl3-catalyzed cyclization route described in CN111978265B and confirmed its potential for delivering high-purity products consistently. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition smoothly from benchtop discovery to full-scale manufacturing. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 5-trifluoromethyl-1,2,4-triazole derivative meets the exacting standards required by global regulatory bodies.
We invite you to collaborate with us to leverage this innovative technology for your next project. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. We encourage you to reach out today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for custom synthesis projects. Let us help you optimize your supply chain and accelerate your time to market with our reliable, cost-effective, and scalable chemical solutions.
