Advanced Spiroindane-Derived Rhodium Catalysts for High-Value Pharmaceutical Intermediates
Introduction to Next-Generation Chiral Catalysts
The landscape of asymmetric catalysis is continuously evolving, driven by the demand for higher enantioselectivity and more robust catalytic systems in the synthesis of complex pharmaceutical intermediates. Patent CN115093313A introduces a significant breakthrough in this domain by disclosing a novel class of multi-methyl substituted spiroindane derived cyclopentadienes and their corresponding rhodium complexes. This technology addresses critical limitations in existing ligand libraries by providing a rigid, chiral scaffold that ensures single stereoselectivity upon metallization. The core innovation lies in the unique spiroindane backbone, which creates a highly defined chiral environment around the metal center, thereby facilitating superior control in asymmetric C-H bond functionalization reactions. For R&D directors seeking reliable solutions for challenging synthetic transformations, this patent represents a pivotal advancement in organometallic chemistry.
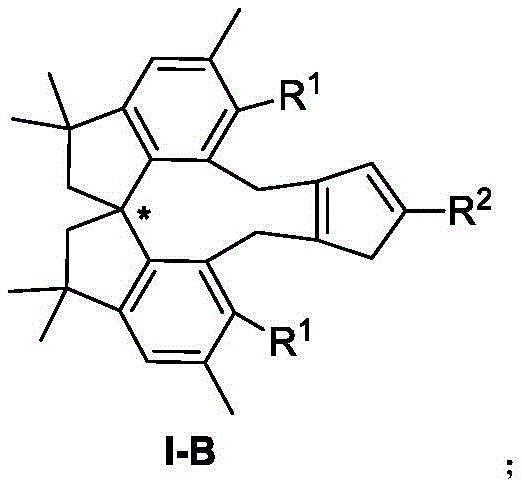
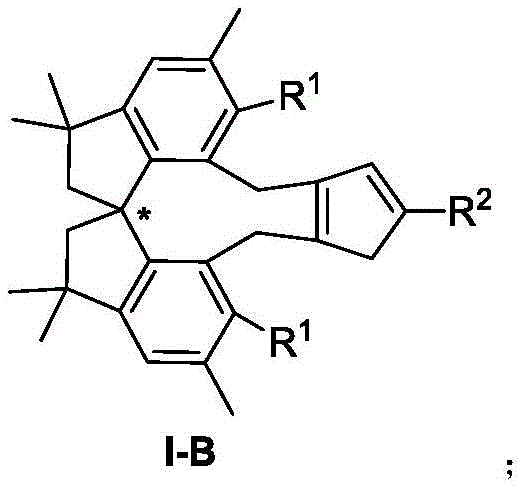
The compounds disclosed, represented generally by formula I-B, feature a spiro chirality that can be configured in either R or S forms, offering versatility for different synthetic pathways. The substituents R1 and R2 are broadly defined, encompassing alkoxy radicals, aryl radicals, and various alkyl groups, which allows for precise tuning of the ligand's steric and electronic properties. This tunability is essential for optimizing catalytic performance across a wide range of substrates. By expanding the structural diversity of cyclopentadienyl ligands beyond traditional scaffolds, this invention opens new avenues for the efficient production of high-purity chiral molecules required in the fine chemical and pharmaceutical industries.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of chiral cyclopentadienyl ligands has faced several persistent challenges that hinder their widespread industrial adoption. As noted in the background of the patent, while ligands based on mannitol derivatization or binaphthyl skeletons have shown promise, they often suffer from relatively limited structural types and complicated synthesis steps. Many existing routes require harsh reaction conditions and lengthy sequences to achieve the desired chirality, which increases production costs and reduces overall process efficiency. Furthermore, a significant drawback of many current ligands is the potential for generating isomers during metallization, which complicates purification and lowers the effective enantiomeric excess of the final catalytic system. The lack of diverse substituents on the cyclopentadiene ring itself in prior art limits the ability to fine-tune the catalyst for specific steric or electronic requirements, often resulting in suboptimal activity or selectivity for challenging substrates.
The Novel Approach
The novel approach presented in CN115093313A overcomes these hurdles by utilizing a polymethyl-substituted spiroindane framework that inherently possesses high rigidity and a well-defined chiral pocket. This structural integrity ensures that upon coordination with rhodium, the complex maintains a single stereoisomeric form, eliminating the issue of isomer generation that plagues other ligand classes. The synthesis strategy allows for the introduction of various functional groups at the R1 and R2 positions, enabling chemists to systematically optimize the catalyst for specific reactions. For instance, the ability to incorporate methoxy, isopropoxy, or phenyl groups provides a powerful toolkit for modulating the electronic density around the metal center. This method not only simplifies the ligand design process but also enhances the catalytic performance in terms of both yield and enantioselectivity, making it a superior choice for cost reduction in fine chemical manufacturing where efficiency is paramount.
Mechanistic Insights into Rh-Catalyzed Asymmetric C-H Functionalization
The efficacy of these rhodium complexes stems from their ability to facilitate asymmetric C-H bond activation with exceptional precision. The spiroindane-derived cyclopentadienyl ligand coordinates strongly with the rhodium center, creating a stable yet reactive species capable of activating inert C-H bonds. The rigid backbone of the ligand restricts the conformational freedom of the substrate during the transition state, effectively directing the stereochemical outcome of the reaction. This mechanism is particularly advantageous in [3+2] cyclization dearomatization reactions, where the formation of multiple stereocenters requires strict spatial control. The presence of the multi-methyl substitution pattern further enhances the steric bulk around the active site, preventing unwanted side reactions and promoting the formation of the desired chiral product.
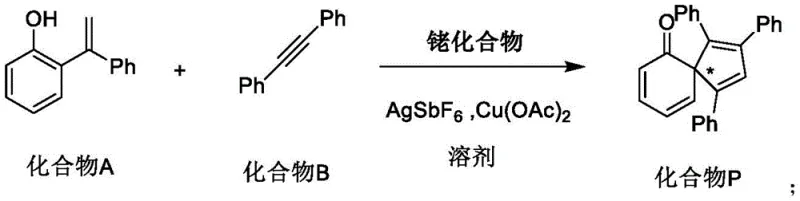
In practical applications, these complexes demonstrate superior performance in intermolecular cyclization reactions. The catalytic cycle typically involves the coordination of the substrate to the rhodium center, followed by C-H activation and subsequent insertion or coupling steps. The unique chiral environment provided by the spiro scaffold ensures that the prochiral faces of the substrate are differentiated effectively, leading to high enantiomeric ratios. This level of control is critical for the synthesis of biologically active compounds where the wrong enantiomer can be inactive or even toxic. The patent data indicates that these catalysts can operate under relatively mild conditions, utilizing standard additives like AgSbF6 and Cu(OAc)2, which simplifies the operational complexity for process chemists aiming for commercial scale-up of complex pharmaceutical intermediates.
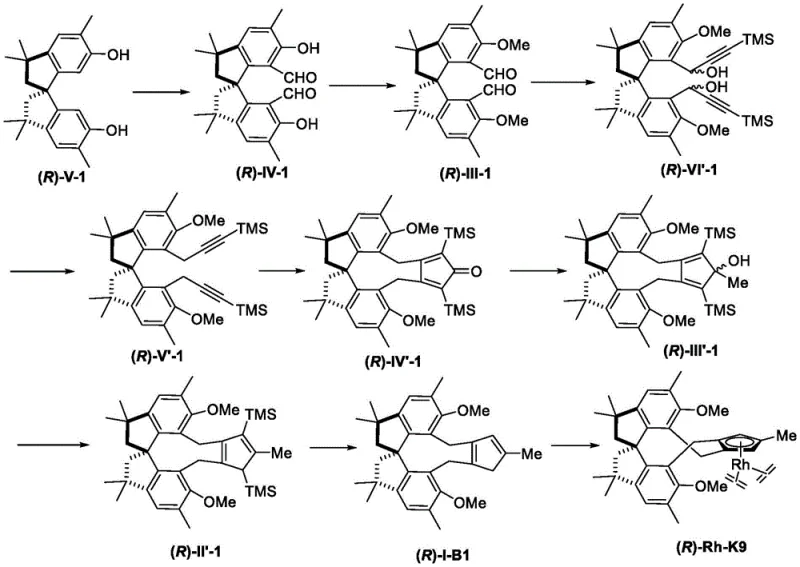
How to Synthesize Multi-Methyl Substituted Spiroindane Derived Rhodium Complex Efficiently
The synthesis of these high-value catalysts follows a logical and scalable sequence of organic transformations that are well-suited for industrial production. The process begins with the functionalization of the spiroindane core, typically involving formylation to introduce aldehyde handles which are subsequently converted into the cyclopentadiene ring system. Key steps include the use of Grignard reagents for alkynylation and cobalt-mediated cyclization to construct the five-membered ring. The final metallization step involves deprotonation of the cyclopentadiene ligand followed by transmetallation with a rhodium precursor. The detailed standardized synthesis steps below outline the specific conditions and reagents required to achieve high purity and yield, ensuring reproducibility for reliable supply chain partners.
- Perform Duff formylation on the spiroindane precursor using hexamethylenetetramine and trifluoroacetic acid to introduce aldehyde groups.
- Execute alkylation or etherification on the phenolic hydroxyl groups using methyl iodide or isopropyl iodide to tune steric properties.
- Conduct alkynylation with trimethylsilylacetylene followed by cobalt-mediated [2+2+1] cyclization to construct the cyclopentadienone core.
- Reduce the cyclopentadienone to the corresponding cyclopentadiene ligand using lithium aluminum hydride and aluminum trichloride.
- Complete the metallization by reacting the ligand with thallium ethoxide and a rhodium precursor such as [Rh(cod)Cl]2 to form the final catalyst.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology offers tangible benefits related to process robustness and material availability. The synthetic routes described in the patent utilize commercially available starting materials and standard reagents such as sodium hydride, thionyl chloride, and common Grignard reagents. This reliance on commodity chemicals significantly mitigates supply chain risks associated with exotic or hard-to-source precursors. Furthermore, the reactions are performed in common solvents like tetrahydrofuran, dichloromethane, and toluene, which simplifies solvent recovery and waste management protocols. The ability to produce these catalysts without requiring specialized equipment or extreme conditions translates directly into lower capital expenditure and operational costs for manufacturing facilities.
- Cost Reduction in Manufacturing: The streamlined synthesis pathway eliminates the need for complex resolution steps often required to separate ligand isomers in traditional methods. Since the spiroindane scaffold ensures single stereoselectivity upon metallization, the downstream purification burden is drastically reduced. This efficiency gain leads to substantial cost savings by minimizing material loss and reducing the consumption of chromatography media and solvents. Additionally, the high catalytic activity allows for lower catalyst loading in downstream applications, further decreasing the cost per kilogram of the final pharmaceutical intermediate produced.
- Enhanced Supply Chain Reliability: The robustness of the synthetic method ensures consistent quality and batch-to-batch reproducibility, which is critical for maintaining uninterrupted production schedules. The use of stable intermediates and standard reaction conditions means that the manufacturing process is less susceptible to variations in raw material quality or environmental factors. This reliability allows supply chain planners to forecast lead times more accurately and maintain optimal inventory levels without the fear of unexpected production delays caused by finicky chemistry.
- Scalability and Environmental Compliance: The patent examples demonstrate successful synthesis on gram scales with clear pathways for expansion to multi-kilogram production. The workup procedures involve standard techniques like extraction, washing, and crystallization, which are easily adaptable to large-scale reactors. Moreover, the avoidance of highly toxic heavy metals in the ligand synthesis phase (prior to rhodium introduction) and the potential for catalyst recycling contribute to a greener manufacturing profile, helping companies meet increasingly stringent environmental regulations and sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this catalytic technology. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for decision-makers evaluating this solution for their specific applications.
Q: What are the structural advantages of spiroindane-derived cyclopentadienyl ligands over traditional binaphthyl ligands?
A: According to patent CN115093313A, spiroindane-derived ligands possess a more rigid structure and a defined chiral environment due to the spiro chirality. Unlike monosubstituted ligands, they exhibit single stereoselectivity after metallization, preventing the generation of isomers which facilitates better chiral control in asymmetric catalysis.
Q: What specific reactions can these rhodium complexes catalyze effectively?
A: These complexes are specifically designed for rhodium-catalyzed asymmetric C-H bond functionalization reactions. The patent highlights their efficacy in intermolecular [3+2] cyclization dearomatization reactions, achieving high yields and enantioselectivity compared to prior art catalysts.
Q: How tunable are the substituents on the cyclopentadiene ring in this new ligand class?
A: The invention allows for significant tunability. The R1 groups on the indane skeleton can be hydrogen, alkoxy (like methoxy or isopropoxy), or aryl groups. Furthermore, the R2 group on the cyclopentadiene ring itself can be hydrogen or alkyl groups like methyl, allowing chemists to fine-tune steric hindrance and electronic properties for specific substrate compatibility.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Spiroindane Cyclopentadiene Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like those described in CN115093313A for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to industrial manufacturing is seamless. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, guaranteeing that every batch of spiroindane-derived rhodium complex or downstream intermediate meets the highest standards of quality required by global regulatory bodies.
We invite you to leverage our technical expertise to optimize your synthetic routes and achieve significant process improvements. By collaborating with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific project needs. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate how our capabilities can support your supply chain and accelerate your time to market.
