Advanced Solvent-Free Synthesis of 3,6-Dimethoxy-2,7-Dimethyl-4-Octenedial for Carotenoid Production
Advanced Solvent-Free Synthesis of 3,6-Dimethoxy-2,7-Dimethyl-4-Octenedial for Carotenoid Production
The chemical synthesis landscape for carotenoid precursors is undergoing a significant transformation driven by the need for higher purity and greener processing standards. Patent CN113773185B introduces a groundbreaking preparation method for 3,6-dimethoxy-2,7-dimethyl-4-octenedial, a pivotal intermediate in the production of C10 dialdehyde, which serves as the backbone for synthesizing beta-carotene, canthaxanthin, astaxanthin, and lycopene. This innovation addresses long-standing challenges in the industry, specifically the issues of high viscosity, poor thermal stability, and excessive impurity profiles associated with traditional acidolysis routes. By implementing a controlled steam stripping technique coupled with precise intermediate monitoring, this process achieves raw material conversion rates exceeding 99% and selectivity greater than 96%, setting a new benchmark for efficiency in fine chemical manufacturing.
For research and development directors overseeing complex synthesis pipelines, the ability to produce intermediates with consistent rheological properties is paramount. The traditional reliance on sulfuric acid catalysis often results in products with deteriorated fluidity and high viscosity, complicating downstream processing and storage. The novel approach detailed in this patent utilizes mild organic acids like oxalic acid in a solvent-free environment, drastically reducing the formation of polymeric by-products. This shift not only enhances the chemical integrity of the 3,6-dimethoxy-2,7-dimethyl-4-octenedial but also ensures that the material maintains a room temperature viscosity of less than 50cP, facilitating seamless integration into subsequent Wittig or Grignard reactions required for vitamin and pigment synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical methods for preparing 3,6-dimethoxy-2,7-dimethyl-4-octenedial have predominantly relied on the use of strong mineral acids, such as sulfuric acid, to catalyze the acetal cleavage of 1,1,3,6,8,8-hexamethoxy-2,7-dimethyl-4-octene. While chemically feasible, these conventional processes suffer from severe drawbacks that impact both product quality and operational safety. The aggressive nature of sulfuric acid promotes uncontrolled side reactions, leading to the formation of complex impurity profiles that significantly increase the viscosity of the final product. High viscosity creates substantial handling difficulties, requiring heated transfer lines and specialized pumping equipment, which increases capital expenditure and operational complexity. Furthermore, the presence of residual acid and polymeric impurities compromises the thermal stability of the dialdehyde, leading to degradation during storage and necessitating frequent quality control interventions to ensure batch consistency.
The Novel Approach
The innovative methodology presented in patent CN113773185B circumvents these limitations through a sophisticated control of reaction kinetics and thermodynamics. Instead of relying on harsh mineral acids in bulk solvent, this process employs catalytic amounts of organic acids, such as oxalic acid, in a neat reaction system. The core breakthrough lies in the introduction of water vapor for gas stripping, which serves a dual purpose: it acts as a heat transfer medium to maintain uniform reaction temperatures between 80°C and 100°C, and it continuously removes methanol, a by-product of the acetal cleavage, from the reaction equilibrium. This continuous removal drives the reaction forward according to Le Chatelier's principle without the need for excessive temperatures that would trigger decomposition. The result is a cleaner reaction profile with minimal waste generation, aligning perfectly with modern green chemistry principles and reducing the burden on wastewater treatment facilities.
Mechanistic Insights into Controlled Acidolysis and Intermediate Management
Understanding the mechanistic pathway of this acidolysis reaction is crucial for optimizing yield and purity. The transformation of 1,1,3,6,8,8-hexamethoxy-2,7-dimethyl-4-octene into the target dialdehyde proceeds through a stepwise hydrolysis mechanism. Initially, the acid catalyst protonates the methoxy groups, facilitating the elimination of methanol to form a hemiacetal intermediate, often referred to as the half-acidolysis product. This intermediate is unstable and must be carefully managed; if allowed to accumulate or persist for too long under acidic conditions, it undergoes further dehydration and conjugation reactions to form unwanted by-products, such as the highly conjugated acidolysis by-product shown in the reaction scheme. These by-products are responsible for the dark coloration and high viscosity observed in inferior batches.
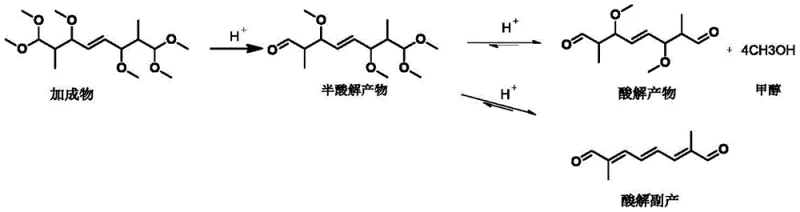
The patent reveals a critical discovery: the mass content of this half-acidolysis intermediate must be strictly controlled within a window of 1% to 20% during the reaction process. When the intermediate concentration exceeds this upper limit, the rate of side reactions accelerates, leading to the polymerization and conjugation issues mentioned previously. Conversely, if the intermediate concentration is too low, it may indicate over-reaction or insufficient catalyst activity, potentially leading to different impurity profiles. By utilizing real-time monitoring, likely via HPLC as described in the patent examples, operators can determine the precise endpoint of the reaction. The steam stripping technique is instrumental here, as the flow rate of water vapor (optimized between 100 mL/h and 500 mL/h) directly influences the rate of methanol removal and thus the concentration of the reactive intermediate, allowing for fine-tuned control over the product's molecular architecture.
How to Synthesize 3,6-Dimethoxy-2,7-Dimethyl-4-Octenedial Efficiently
Implementing this synthesis route requires precise adherence to the specified reaction parameters to ensure the benefits of low viscosity and high stability are realized. The process begins with charging a reaction vessel with the hexamethoxy adduct and a catalytic amount of oxalic acid, typically ranging from 1% to 5% by weight. The system is then heated to the target range of 80°C to 100°C under a slight vacuum or atmospheric pressure, depending on the specific setup. Crucially, water vapor is introduced at the bottom of the reactor through a gas distributor to ensure uniform dispersion throughout the liquid phase. This vapor stream carries the evolved methanol out of the reactor and into a rectification column for separation, preventing the reverse reaction and maintaining the driving force for hydrolysis. Detailed standardized synthetic steps see the guide below.
- Charge the reactor with 1,1,3,6,8,8-hexamethoxy-2,7-dimethyl-4-octene and an acid catalyst such as oxalic acid.
- Heat the mixture to 80-100°C under reduced pressure while introducing water vapor for gas stripping.
- Monitor the reaction to ensure the half-acidolysis intermediate content remains between 1% and 20% before stopping the reaction.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this novel synthesis route offers compelling strategic advantages beyond mere chemical elegance. The elimination of organic solvents represents a significant reduction in raw material costs and logistical complexity. Solvent-free processes remove the need for large-scale solvent recovery systems, reduce fire hazards, and minimize the volume of hazardous waste requiring disposal. This translates directly into a leaner cost structure and a more robust supply chain that is less vulnerable to fluctuations in solvent markets. Furthermore, the improved physical properties of the product—specifically its low viscosity and high thermal stability—simplify storage and transportation. Unlike traditional high-viscosity intermediates that may require heated tanks or specialized drumming procedures, this improved dialdehyde flows freely at room temperature, reducing energy consumption in warehouses and minimizing the risk of product solidification during transit in colder climates.
- Cost Reduction in Manufacturing: The transition to a solvent-free system using inexpensive organic acid catalysts like oxalic acid drastically lowers the bill of materials. By avoiding the procurement, storage, and recycling of volatile organic compounds, manufacturers can achieve substantial operational savings. Additionally, the high selectivity of the reaction means that less raw material is wasted on forming unusable by-products, maximizing the yield per kilogram of input. The simplified downstream processing, which no longer requires extensive neutralization and washing steps to remove strong mineral acids, further reduces utility costs and labor hours, contributing to a more competitive pricing model for the final carotenoid ingredients.
- Enhanced Supply Chain Reliability: The robustness of this process enhances supply continuity by reducing the frequency of batch failures due to quality deviations. Traditional methods often struggle with batch-to-batch variability in viscosity and color, leading to rejected lots and supply disruptions. The precise control offered by the steam stripping method ensures consistent product specifications, allowing for reliable inventory planning. Moreover, the use of readily available and stable reagents like oxalic acid and deionized water mitigates the risk of supply bottlenecks associated with specialized or hazardous catalysts, ensuring that production schedules can be maintained even during periods of market volatility.
- Scalability and Environmental Compliance: From a regulatory and sustainability perspective, this process is exceptionally well-suited for scale-up. The absence of organic solvents simplifies the permitting process for new production lines and reduces the facility's overall environmental footprint. The "three wastes" (wastewater, waste gas, and solid waste) are significantly minimized, aligning with increasingly stringent global environmental regulations. The ability to recycle condensed water vapor back into the system for stripping further demonstrates a commitment to resource efficiency. This green manufacturing profile not only future-proofs the supply chain against tightening environmental laws but also appeals to end-consumers and brand owners who prioritize sustainably sourced ingredients in their nutraceutical and cosmetic products.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this advanced acidolysis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering a clear picture of why this method represents a superior choice for modern chemical manufacturing. Understanding these nuances helps stakeholders make informed decisions about integrating this intermediate into their broader production workflows.
Q: Why is controlling the half-acidolysis intermediate content critical?
A: Maintaining the intermediate content between 1% and 20% prevents excessive side reactions that lead to high-viscosity impurities and poor thermal stability in the final product.
Q: What are the advantages of using steam stripping over traditional solvent methods?
A: Steam stripping eliminates the need for organic solvents, reduces three-waste generation, and continuously removes methanol by-products to drive the equilibrium forward without degrading product quality.
Q: How does this process improve the physical properties of the dialdehyde?
A: By minimizing impurity formation, the resulting product exhibits low viscosity (less than 50cP at room temperature) and superior fluidity, making it easier to store and transport compared to conventional methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,6-Dimethoxy-2,7-Dimethyl-4-Octenedial Supplier
At NINGBO INNO PHARMCHEM, we recognize that the quality of the final carotenoid product is inextricably linked to the purity and consistency of its precursors. Our technical team has extensively analyzed the pathway described in CN113773185B and possesses the expertise to replicate and optimize this solvent-free acidolysis process on an industrial scale. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to manufacturing plant is seamless and efficient. Our rigorous QC labs enforce stringent purity specifications, guaranteeing that every batch of 3,6-dimethoxy-2,7-dimethyl-4-octenedial meets the low viscosity and high stability criteria essential for your downstream synthesis of beta-carotene and other high-value pigments.
We invite you to collaborate with us to leverage this technological advancement for your supply chain. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out for specific COA data and route feasibility assessments to verify how our optimized manufacturing capabilities can enhance your production efficiency. Let us help you secure a stable, high-quality supply of this critical intermediate, enabling you to focus on innovation and market expansion with confidence.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
