Scalable Synthesis of 6'-CN Modified Nucleosides for Next-Gen Oligonucleotide Therapeutics
Scalable Synthesis of 6'-CN Modified Nucleosides for Next-Gen Oligonucleotide Therapeutics
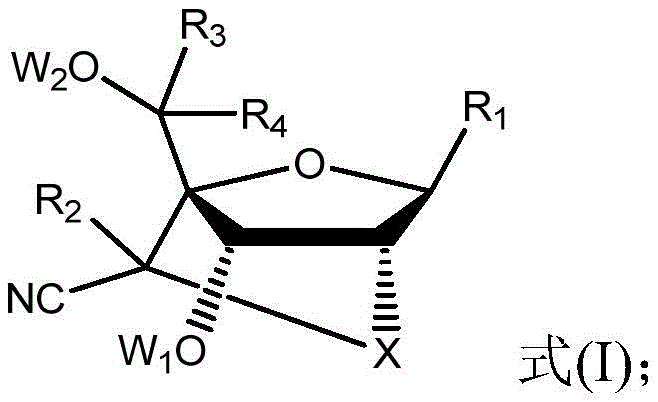
The rapid evolution of oligonucleotide therapeutics demands chemical modifications that balance stability with high binding affinity. Patent CN110590886B introduces a groundbreaking class of modified nucleosides featuring a nitrile group at the 6' position of the bridge ring. This structural innovation addresses the critical limitations of natural oligonucleotides, such as poor in vivo stability and low bioavailability, by significantly enhancing ribozyme tolerance. For pharmaceutical developers, this represents a pivotal advancement in designing second-generation antisense gapmers with reduced hepatotoxicity and superior target specificity. The technology provides a robust foundation for creating high-purity intermediates essential for next-generation RNA-targeting medicines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional oligonucleotide therapies often struggle with rapid degradation by endogenous nucleases, necessitating frequent dosing or high concentrations that can lead to toxicity. While earlier modifications like phosphorothioates or 2'-OMe improved stability, they often compromised binding affinity or introduced significant off-target effects. Furthermore, existing Locked Nucleic Acid (LNA) technologies, although effective, can sometimes exhibit excessive binding strength leading to non-specific interactions or limited solubility profiles. The industry urgently requires a modification strategy that offers a precise balance between metabolic stability and hybridization thermodynamics without complicating the manufacturing supply chain with rare or hazardous reagents.
The Novel Approach
The methodology described in the patent overcomes these hurdles by introducing a polar cyano group (-CN) at the C6' position of the nucleoside scaffold. This specific substitution increases steric hindrance around the methylene bridge, effectively shielding the phosphodiester backbone from enzymatic hydrolysis. Unlike bulky hydrophobic groups, the polar nature of the nitrile moiety enhances hydration, which improves tissue distribution and antisense activity. This approach allows for shorter antisense sequences while maintaining potent gene silencing efficacy, thereby simplifying the overall drug design and potentially reducing the metabolic burden on the liver. The result is a highly selective therapeutic candidate with a markedly improved safety profile.
Mechanistic Insights into 6'-Nitrile Substitution and Cyclization
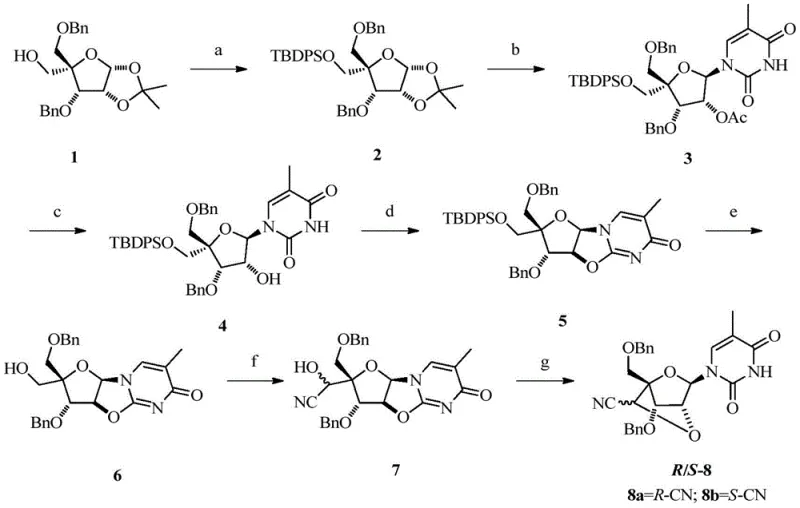
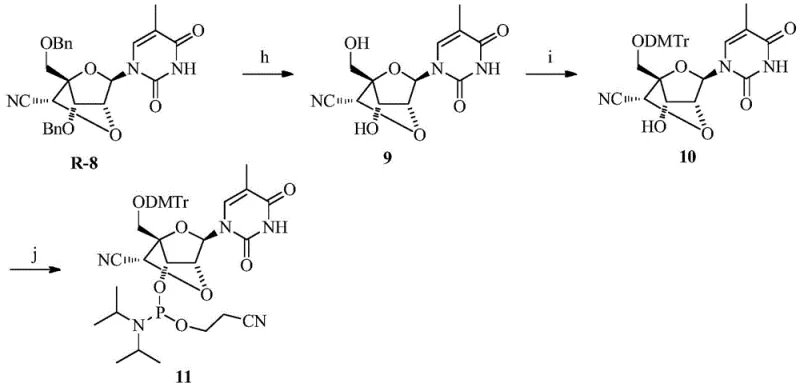
The core of this technology lies in the precise construction of the bridged bicyclic structure followed by the regioselective introduction of the nitrile group. The synthesis begins with the protection of the 5'-hydroxyl group, typically using bulky silyl groups like TBDPS to ensure orthogonality during subsequent steps. The formation of the 2',4'-bridge is achieved through an intramolecular cyclization reaction, often catalyzed by Lewis acids or activated sulfonates, which locks the sugar into the C3'-endo conformation required for high-affinity RNA binding. This conformational locking is crucial as it pre-organizes the nucleotide for hybridization, reducing the entropic penalty upon binding to the target mRNA strand.
Following the bridge formation, the 6'-hydroxyl group is oxidized to an aldehyde intermediate using mild oxidants such as IBX (2-iodoxybenzoic acid). This aldehyde serves as the electrophilic center for the subsequent nucleophilic attack by a cyanide source, such as trimethylsilyl cyanide (TMSCN), in the presence of a Lewis acid catalyst like aluminum chloride. This step is critical as it installs the functional group responsible for the enhanced nuclease resistance. The reaction conditions are carefully controlled to prevent epimerization, ensuring the production of the desired stereoisomer which exhibits the optimal biological activity. The final deprotection and phosphitylation steps yield the phosphoramidite building blocks ready for automated oligonucleotide synthesis.
How to Synthesize 6'-CN Modified Nucleosides Efficiently
The preparation of these advanced intermediates follows a logical sequence of protection, coupling, cyclization, and functionalization. The process starts with commercially available sugar derivatives, making the raw material supply chain robust and cost-effective. Key steps involve the use of standard protecting group strategies compatible with solid-phase synthesis, ensuring that the final phosphoramidites can be seamlessly integrated into existing manufacturing workflows. The oxidation and cyanation steps are performed under anhydrous conditions to maximize yield and purity, minimizing the formation of diastereomers that could complicate downstream purification. For a detailed breakdown of the specific reaction parameters and stoichiometry, refer to the standardized guide below.
- Protect the 5'-hydroxyl group of the starting sugar substrate using TBDPSCl and imidazole in DMF to form the silyl ether.
- Perform nucleobase coupling with thymine using BSA and TMSOTf, followed by intramolecular cyclization with DMAP and Tf2O to form the bridged structure.
- Oxidize the 6'-hydroxyl to an aldehyde using IBX, then perform nucleophilic substitution with TMSCN and AlCl3 to introduce the nitrile group.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the synthetic route outlined in the patent utilizes widely available reagents and avoids the need for precious metal catalysts or cryogenic conditions that often drive up costs in fine chemical manufacturing. The reliance on standard silyl protecting groups and common oxidants means that sourcing risks are minimized, ensuring a stable supply of critical intermediates. Furthermore, the high yields reported in the experimental examples suggest a material-efficient process that reduces waste generation and lowers the overall cost of goods sold. This efficiency is particularly valuable for long-term commercial contracts where price stability and volume scalability are paramount concerns for supply chain directors.
- Cost Reduction in Manufacturing: The elimination of complex transition metal catalysts and the use of straightforward workup procedures significantly lower the operational expenditure associated with purification. By avoiding expensive chromatography steps where possible and utilizing crystallization for intermediate isolation, the process achieves substantial cost savings. The high atom economy of the cyanation step further contributes to reducing raw material consumption, making the production of these high-value intermediates more economically viable for large-scale applications.
- Enhanced Supply Chain Reliability: The starting materials, such as protected ribose derivatives and thymine, are commodity chemicals with established global supply chains. This reduces the risk of bottlenecks that often plague the production of exotic nucleoside analogues. Additionally, the robustness of the synthetic route allows for flexible manufacturing schedules, enabling suppliers to respond quickly to fluctuating demand from pharmaceutical clients without compromising on quality or delivery timelines.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing solvents and reagents that are manageable in large reactor systems. The waste streams generated are primarily organic and can be treated using standard industrial effluent protocols, ensuring compliance with stringent environmental regulations. This eco-friendly profile not only mitigates regulatory risk but also aligns with the sustainability goals of modern pharmaceutical companies seeking green chemistry solutions for their drug pipelines.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this 6'-CN modification technology. These insights are derived directly from the patent specifications and are intended to clarify the feasibility and benefits of adopting this synthetic pathway for oligonucleotide drug development. Understanding these nuances is essential for project managers evaluating the integration of these novel building blocks into their existing discovery and development programs.
Q: What is the primary advantage of the 6'-CN modification over standard LNA?
A: The introduction of a polar nitrile group at the 6' position significantly enhances nuclease resistance and improves binding affinity to target RNA while maintaining high selectivity, reducing off-target effects compared to traditional LNA modifications.
Q: Is the synthetic route suitable for large-scale manufacturing?
A: Yes, the process utilizes standard reagents like TMSCN and IBX under controlled conditions, avoiding exotic catalysts, which facilitates scale-up from laboratory to commercial production volumes.
Q: How does this modification impact the safety profile of oligonucleotide drugs?
A: The 6'-CN modification reduces hepatotoxicity and immunostimulation risks by improving the specificity of RNaseH cleavage and shortening the required antisense gapmer sequence length.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 6'-CN Modified Nucleoside Supplier
NINGBO INNO PHARMCHEM stands at the forefront of nucleoside chemistry, offering extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the sensitive chemistry required for 6'-CN modified nucleosides, ensuring stringent purity specifications are met for every batch. With rigorous QC labs and a dedicated team of process chemists, we guarantee the consistency and quality necessary for clinical and commercial supply of oligonucleotide intermediates. We understand the critical nature of these materials in the drug development timeline and are committed to delivering excellence.
We invite you to collaborate with us to optimize your supply chain for next-generation antisense therapeutics. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific project needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can accelerate your path to market while reducing overall production costs.
