Advanced Synthesis of Chiral Ferrocene Homoallylamine Derivatives for Scalable Pharmaceutical Production
The chemical industry is constantly seeking more efficient pathways to access high-value chiral building blocks, and patent CN112010910B represents a significant leap forward in this domain by disclosing a robust method for synthesizing chiral ferrocene homoallylamine derivatives. This technology addresses the critical need for scalable production of chiral amine structural units, which are essential scaffolds in the development of next-generation antidepressant and antitumor medications. By leveraging a sophisticated iridium-catalyzed allylation and 2-aza-Cope rearrangement tandem reaction, the process bypasses the limitations of classical resolution techniques, offering a direct route to high-purity intermediates. For R&D directors and procurement specialists alike, this innovation signals a shift towards more sustainable and cost-effective manufacturing paradigms for complex organometallic compounds. The ability to generate these versatile precursors with exceptional stereocontrol opens new avenues for optimizing supply chains in the fine chemical sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral ferrocenyl ethanol and related Ugi amines has been plagued by inefficiencies inherent in traditional resolution strategies. The conventional splitting method, which involves resolving racemates using chiral tartaric acid, is notoriously resource-intensive, often resulting in a theoretical maximum yield of only 50% for the desired enantiomer. Furthermore, achieving high optical purity frequently necessitates multiple rounds of recrystallization, which drastically increases solvent consumption, processing time, and overall production costs. Alternatively, enzymatic catalysis methods, while greener in principle, often struggle with substrate specificity and require expensive enzyme reagents that can degrade process economics. These legacy approaches create bottlenecks in the supply chain, leading to longer lead times and inconsistent batch-to-batch quality, which are unacceptable for the rigorous standards of modern pharmaceutical manufacturing.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a chiral iridium complex to catalyze a tandem allylation and 2-aza-Cope rearrangement, fundamentally transforming the synthesis landscape. This method allows for the direct construction of the chiral center with remarkable precision, achieving yields between 90% and 96% and enantiomeric excess values exceeding 97%. By employing readily available substrates such as cinnamyl methyl carbonate derivatives, the process eliminates the need for costly resolving agents and reduces the number of synthetic steps significantly. The reaction conditions are notably mild, operating effectively at temperatures between 25°C and 110°C, which enhances safety and reduces energy consumption. This streamlined methodology not only accelerates the timeline from bench to pilot plant but also ensures a more reliable supply of high-purity intermediates for downstream drug synthesis.
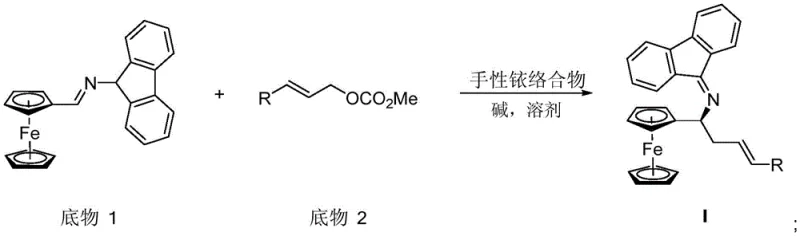
Mechanistic Insights into Iridium-Catalyzed Asymmetric Allylation
The core of this technological breakthrough lies in the precise orchestration of the chiral iridium complex, which acts as a highly selective molecular machine to direct the stereochemical outcome of the reaction. The catalyst, formed in situ from precursors like [Ir(COD)Cl]2 and specific chiral ligands such as (S,S,S)-L1, creates a rigid chiral environment around the metal center. During the reaction, the iridium species activates the allylic carbonate substrate, facilitating the nucleophilic attack by the ferrocenyl imine in a highly controlled manner. This is followed by a spontaneous 2-aza-Cope rearrangement that locks in the chirality, ensuring that the resulting alpha-ferrocene homoallylamine possesses the desired configuration. The mechanistic elegance of this tandem process minimizes the formation of by-products, as the transition state is tightly regulated by the steric bulk of the ligand system.
From an impurity control perspective, the high stereoselectivity of the iridium catalyst is paramount in reducing the burden on downstream purification processes. In traditional syntheses, the presence of unwanted enantiomers or regioisomers often requires complex chromatographic separations that are difficult to scale. However, with enantiomeric excess values consistently above 97%, the crude product from this iridium-catalyzed reaction is of sufficient quality for many subsequent transformations without extensive purification. This inherent purity is a critical advantage for pharmaceutical applications, where impurity profiles are strictly regulated. The robustness of the catalytic cycle also means that the process is less sensitive to minor fluctuations in reaction parameters, providing a stable and reproducible manufacturing protocol that aligns with Good Manufacturing Practice (GMP) requirements.
How to Synthesize Chiral Ferrocene Homoallylamine Efficiently
The synthesis of these valuable chiral intermediates follows a logical progression starting from the tandem catalytic reaction to form the protected amine, followed by deprotection and functionalization steps. The initial step involves dissolving the ferrocenyl imine substrate and the allylic carbonate in a solvent like tetrahydrofuran under an inert nitrogen atmosphere. A chiral iridium complex, prepared separately or in situ, is added along with a base such as DBU to initiate the transformation. Following the formation of the key intermediate, various downstream modifications can be performed, including acid hydrolysis to reveal the free amine or reductive amination to introduce dimethylamino groups. These steps allow for the diversification of the scaffold into a library of potential ligands or drug candidates. Detailed standardized synthetic steps for this process are provided in the guide below.
- Perform an allylation/2-aza-Cope rearrangement tandem reaction between substrate 1 and substrate 2 using a chiral iridium complex catalyst in a solvent like tetrahydrofuran with a base such as DBU.
- Convert the resulting compound I into chiral alpha-ferrocene homoallylamine derivative II via acid hydrolysis or amine exchange reactions using hydroxylamine acetate.
- Further functionalize derivative II through catalytic hydrogenation to form derivative III, or via reductive amination to produce derivative IV for ligand synthesis.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this iridium-catalyzed technology translates into tangible strategic benefits that extend beyond simple chemistry. The elimination of resolution steps and the high atom economy of the tandem reaction significantly reduce the raw material intensity of the process. By avoiding the 50% yield loss inherent in racemic splitting, the effective throughput of the manufacturing facility is nearly doubled without requiring additional capital investment in reactor volume. Furthermore, the use of common organic solvents and commercially available catalyst precursors mitigates supply risk, ensuring that production schedules are not disrupted by the scarcity of exotic reagents. This reliability is crucial for maintaining continuous operations in a competitive global market.
- Cost Reduction in Manufacturing: The economic impact of this process is profound, primarily driven by the drastic reduction in waste generation and the high yield of the target product. Since the chiral information is introduced directly during the bond-forming step, there is no need to purchase expensive chiral acids or enzymes for resolution, which are often single-use consumables. Additionally, the low catalyst loading, typically around 3 mol%, ensures that the cost contribution of the precious metal iridium remains manageable, especially when considering the high value of the final pharmaceutical intermediates. The simplified workup procedures, often requiring only filtration or simple crystallization, further lower the operational expenditures associated with labor and utilities.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the versatility of the substrate scope, which tolerates a wide range of substituents including aryl, heterocyclic, and alkyl groups. This flexibility allows manufacturers to source alternative starting materials if specific supply lines are constrained, without needing to revalidate the entire process. The robust nature of the reaction conditions, which do not require cryogenic temperatures or ultra-high pressures, means that the process can be executed in standard multipurpose reactors found in most fine chemical plants. This compatibility with existing infrastructure accelerates the technology transfer from lab to commercial scale, reducing the time to market for new drug candidates.
- Scalability and Environmental Compliance: Scaling this process to industrial levels is facilitated by the homogeneous nature of the catalysis and the absence of hazardous by-products. The reaction generates minimal waste compared to stoichiometric chiral auxiliary methods, aligning with increasingly stringent environmental regulations and corporate sustainability goals. The ability to recycle solvents and the potential for catalyst recovery further enhance the green chemistry profile of the manufacture. For large-scale production, the process demonstrates excellent heat transfer characteristics and safety margins, making it suitable for ton-scale campaigns required to support the global demand for antidepressant and antitumor therapies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral ferrocene synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear picture of the process capabilities. Understanding these details helps stakeholders make informed decisions about integrating this methodology into their existing production portfolios. The answers reflect the practical realities of running asymmetric catalysis in a commercial setting.
Q: What are the advantages of the iridium-catalyzed method over traditional resolution methods?
A: The iridium-catalyzed method described in patent CN112010910B offers significantly higher efficiency and chiral purity compared to traditional splitting methods which suffer from low yields and require repeated recrystallization. This catalytic approach achieves yields of 90-96% with enantiomeric excess greater than 97%, eliminating the high costs associated with chiral resolving agents.
Q: Can these ferrocene derivatives be used for applications beyond pharmaceutical intermediates?
A: Yes, beyond serving as intermediates for antidepressant and antitumor drugs, these chiral ferrocene compounds are pivotal precursors for synthesizing chiral ferrocene phosphorane and diphosphine ligands. These ligands possess wide application prospects in the field of asymmetric catalysis, enhancing the utility of the synthetic route for broader chemical manufacturing.
Q: What are the typical reaction conditions for the key allylation step?
A: The key allylation/2-aza-Cope rearrangement tandem reaction typically proceeds under mild conditions, reacting for 12-24 hours at temperatures ranging from 25-110°C. The process utilizes a chiral iridium complex at low loading (preferably 3 mol%) in solvents such as tetrahydrofuran, ensuring operational simplicity and safety.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Ferrocene Homoallylamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the chiral ferrocene homoallylamine derivatives described in patent CN112010910B for the pharmaceutical and fine chemical industries. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from gram-scale discovery to industrial reality. Our facilities are equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, including chiral HPLC analysis to guarantee enantiomeric excess levels that meet or exceed the >97% benchmark established by the patent. We are committed to delivering high-purity pharmaceutical intermediates that empower your drug development programs.
We invite you to collaborate with our technical team to explore how this advanced iridium-catalyzed route can optimize your specific supply chain needs. By engaging with us, you can request a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this high-yield methodology for your specific targets. Our experts are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate the viability of this technology for your portfolio. Contact our technical procurement team today to discuss how we can support your journey towards more efficient and sustainable chiral synthesis.
