Advanced Chiral Quinoline Oxazoline Ligands for High-Performance Asymmetric Catalysis
The landscape of asymmetric catalysis is continuously evolving, driven by the demand for more efficient and cost-effective methods to produce chiral intermediates. Patent CN113880822A introduces a significant breakthrough with the disclosure of a novel class of chiral imine-containing quinoline oxazoline compounds. These compounds, characterized by their high optical purity and unique structural framework, serve as exceptional ligands for transition metal catalysis. The patent details a robust synthetic methodology that achieves a remarkable two-step total yield of 85%, addressing common inefficiencies in ligand production. By integrating an imine functionality at the 2-position and an oxazoline ring at the 8-position of the quinoline scaffold, these ligands offer superior coordination properties. This innovation is particularly relevant for the pharmaceutical industry, where the need for reliable pharmaceutical intermediate suppliers who can deliver high-enantiomeric excess products is critical. The ability of these ligands to form stable complexes with abundant transition metals like Cobalt and Iron represents a paradigm shift away from reliance on scarce precious metals.
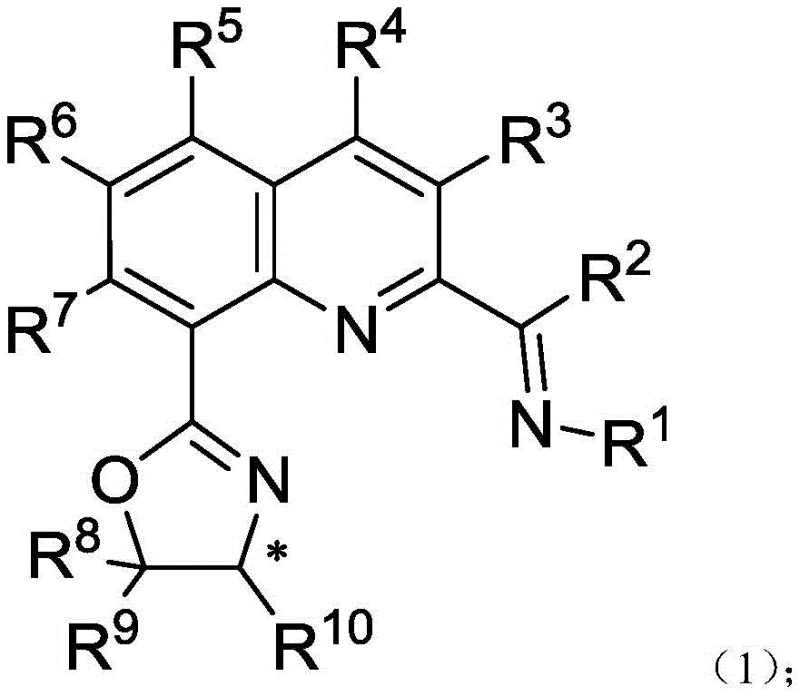
Traditional methods for synthesizing chiral amines and alcohols often rely on classical bisoxazoline ligands or pyridine diimines, which, while effective, can present limitations in terms of substrate scope and catalyst cost. Conventional processes frequently require expensive noble metals such as Ruthenium or Rhodium to achieve acceptable levels of stereoselectivity, creating bottlenecks in cost reduction in pharmaceutical manufacturing. Furthermore, the synthesis of complex ligand architectures often involves multi-step sequences with poor atom economy, leading to significant waste generation and extended lead times. In contrast, the novel approach outlined in the patent utilizes a streamlined two-step strategy. The initial condensation of 2-acyl-8-bromoquinoline with various amines followed by a palladium-catalyzed coupling with chiral oxazolines provides direct access to the target ligands. This method not only simplifies the supply chain for high-purity intermediates but also enhances the overall sustainability of the production process by minimizing solvent usage and purification steps.
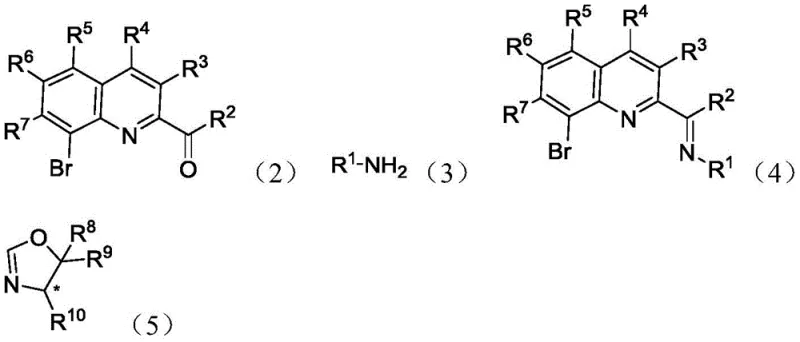
The mechanistic efficacy of these catalysts stems from their ability to create a rigid, chiral environment around the central metal atom. When complexed with metals such as Cobalt (as seen in Formula 6), the tridentate coordination mode involving the quinoline nitrogen, the imine nitrogen, and the oxazoline nitrogen locks the metal into a specific geometry. This precise spatial arrangement is crucial for differentiating between the enantiotopic faces of the substrate during the catalytic cycle. For instance, in the asymmetric hydrosilylation of aryl ketimines, the catalyst facilitates the transfer of a hydride species from the silane to the imine carbon with high regioselectivity. The steric bulk provided by substituents on the oxazoline ring (such as tert-butyl or isopropyl groups) further shields one face of the reactive center, thereby enforcing high optical selectivity. This level of control is essential for reducing lead time for high-purity intermediates, as it minimizes the need for downstream resolution steps to separate racemic mixtures.
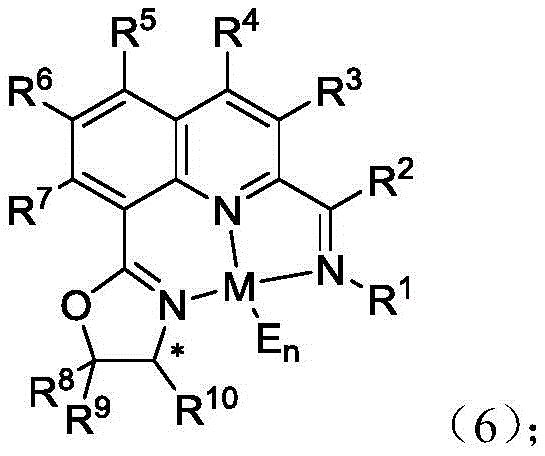
Understanding the catalytic cycle is vital for optimizing reaction conditions in an industrial setting. The metal complexes function by activating the silicon-hydrogen or boron-hydrogen bond, generating a reactive metal-hydride species. This species then undergoes migratory insertion into the carbon-nitrogen or carbon-oxygen double bond of the substrate. The subsequent reductive elimination releases the chiral product and regenerates the active catalyst. The patent demonstrates that these complexes are effective even at low loadings (0.1 to 5 mol%), which significantly lowers the residual metal content in the final product—a key concern for regulatory compliance in API synthesis. Moreover, the versatility of the ligand system allows for fine-tuning of electronic and steric properties by modifying the R groups, enabling chemists to tailor the catalyst for specific challenging substrates. This adaptability ensures consistent quality and performance across different batches, a hallmark of a trusted specialty chemical partner.
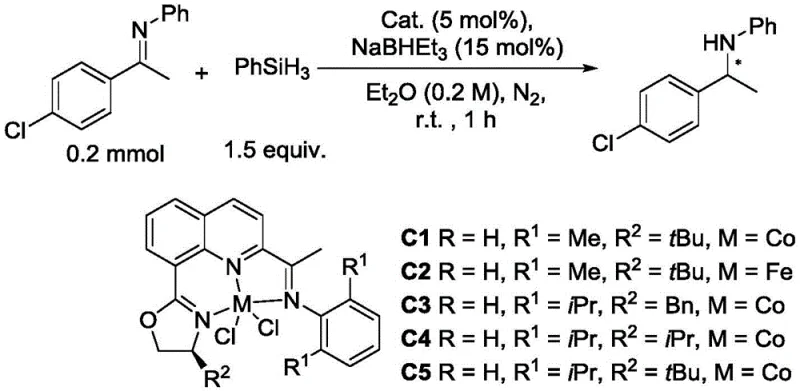
How to Synthesize Chiral Imine-Containing Quinoline Oxazoline Efficiently
The synthesis of these advanced ligands is designed for operational simplicity and high reproducibility, making it suitable for both laboratory research and commercial scale-up of complex polymer additives or pharmaceutical precursors. The process begins with the preparation of the key intermediate, 8-bromo-2-iminoquinoline, via a dehydration condensation reaction. This step typically employs p-toluenesulfonic acid as a catalyst in toluene, utilizing a Dean-Stark apparatus to remove water and drive the equilibrium towards product formation. Following isolation, the intermediate undergoes a cross-coupling reaction with a chiral oxazoline derivative. This critical step utilizes a palladium catalyst system, specifically palladium acetate combined with a bidentate phosphine ligand like 1,2-bis(diphenylphosphino)ethane, in the presence of a strong base such as lithium tert-butoxide. The reaction is conducted under strictly anhydrous and anaerobic conditions to prevent catalyst deactivation. Detailed standardized synthesis steps are provided in the guide below.
- Condense 2-acyl-8-bromoquinoline with an amine compound using p-toluenesulfonic acid in toluene under reflux with water separation.
- Perform a coupling reaction between the resulting iminoquinoline and an oxazoline compound using palladium acetate, a phosphine ligand, and lithium tert-butoxide in dioxane.
- Purify the final chiral quinoline oxazoline product via column chromatography using petroleum ether and ethyl acetate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this technology offers substantial strategic benefits beyond mere technical performance. The shift towards using earth-abundant metals like Cobalt and Iron, as enabled by these robust ligands, directly addresses the volatility associated with precious metal pricing. By eliminating the dependency on Rhodium or Iridium for certain asymmetric transformations, manufacturers can achieve significant cost savings in raw material procurement. Additionally, the high efficiency of the synthetic route, with yields reaching up to 94% for specific ligand variants, ensures better material throughput and reduced waste disposal costs. This efficiency translates into a more resilient supply chain, capable of meeting fluctuating market demands without the risk of raw material shortages. The simplified purification protocols, often requiring only standard column chromatography or recrystallization, further reduce the operational complexity and energy consumption associated with production.
- Cost Reduction in Manufacturing: The utilization of inexpensive transition metal salts such as Cobalt Chloride instead of precious metals drastically lowers the catalyst cost per kilogram of product. This economic advantage is compounded by the high turnover numbers observed in the catalytic reactions, meaning less catalyst is needed to produce the same amount of product. Furthermore, the ligand synthesis itself avoids exotic reagents, relying on commercially available starting materials like substituted anilines and acetylquinolines. This accessibility ensures stable pricing and reduces the risk of supply disruptions caused by geopolitical factors affecting rare metal mining. The overall process design prioritizes atom economy, minimizing the generation of hazardous byproducts and lowering the environmental compliance costs associated with waste treatment.
- Enhanced Supply Chain Reliability: The robustness of the synthetic protocol allows for flexible manufacturing schedules. Since the reactions can be performed at room temperature or mild reflux conditions, there is no need for specialized cryogenic equipment or extreme high-pressure reactors, which are often bottlenecks in chemical plants. The stability of the metal complexes also implies a longer shelf life for the catalyst inventory, reducing the frequency of replenishment orders. This reliability is crucial for maintaining continuous production lines for critical pharmaceutical intermediates. By securing a source of these ligands, companies can mitigate the risks associated with single-source suppliers of proprietary catalysts, thereby diversifying their supply base and enhancing overall operational security.
- Scalability and Environmental Compliance: The described methods have been validated on gram scales with consistent results, indicating a clear path towards kilogram and ton-scale production. The use of common organic solvents like toluene, dioxane, and diethyl ether simplifies solvent recovery and recycling processes, aligning with green chemistry principles. The high selectivity of the catalysts reduces the formation of unwanted isomers, which simplifies downstream purification and reduces the volume of solvent required for chromatography. This reduction in solvent intensity not only lowers costs but also decreases the carbon footprint of the manufacturing process. Compliance with increasingly stringent environmental regulations is easier to achieve when the process inherently generates less waste and utilizes safer, more abundant materials.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this catalytic technology. These insights are derived directly from the experimental data and embodiments provided in the patent documentation, ensuring accuracy and relevance for process development teams. Understanding these nuances helps in making informed decisions about integrating this chemistry into existing production workflows. The answers cover aspects ranging from catalyst loading to substrate compatibility, providing a comprehensive overview of the technology's capabilities.
Q: What are the primary advantages of these quinoline oxazoline ligands over traditional bisoxazoline ligands?
A: These novel ligands feature a unique hybrid structure combining an imine group at the C2 position and an oxazoline ring at the C8 position of the quinoline core. This specific architecture allows for stable complexation with inexpensive transition metals like Cobalt and Iron, offering a cost-effective alternative to precious metal catalysts while maintaining high enantioselectivity (up to >85% ee) in asymmetric reductions.
Q: Can these metal complexes be used for large-scale industrial applications?
A: Yes, the synthetic route described in the patent is highly efficient with a reported two-step total yield reaching 85%. The use of robust transition metals and standard organic solvents facilitates scalability. Furthermore, the catalysts operate effectively at room temperature for hydrosilylation and hydroboration reactions, reducing energy consumption and simplifying process control for commercial manufacturing.
Q: What types of chemical transformations do these catalysts support?
A: The metal complexes derived from these ligands are versatile catalysts primarily designed for asymmetric hydrosilylation and hydroboration reactions. They are particularly effective in reducing carbon-heteroatom double bonds, such as converting aryl ketimines to chiral amines and aryl ketones to chiral alcohols, which are critical building blocks in pharmaceutical and agrochemical synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Quinoline Oxazoline Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the chiral quinoline oxazoline technology disclosed in CN113880822A for the synthesis of high-value pharmaceutical intermediates. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to full-scale manufacturing is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including enantiomeric excess analysis via chiral HPLC, guaranteeing that every batch meets the highest international standards. We understand that consistency is key in the pharmaceutical supply chain, and our dedicated process chemistry team is ready to optimize these catalytic routes for your specific API needs.
We invite you to collaborate with us to leverage these advanced catalytic systems for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your current manufacturing challenges. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our expertise in asymmetric catalysis can accelerate your development timelines and enhance your product's market competitiveness. Let us be your partner in turning innovative chemical concepts into commercial reality.
