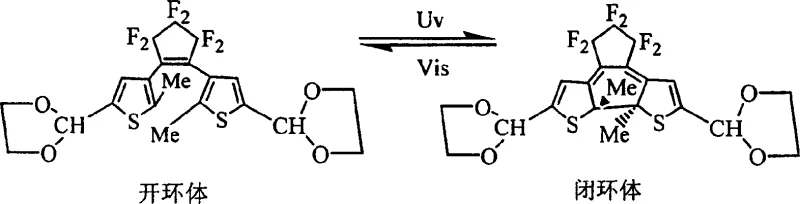
Scalable Production of High-Performance Diarylethene Photochromic Compounds for Advanced Optical Storage
The rapid evolution of information technology has created an insatiable demand for digital storage solutions that offer unprecedented density, speed, and longevity. Traditional magnetic recording media are approaching their physical limits, prompting a strategic shift towards optical storage technologies capable of non-contact read/write operations and superior carrier-to-noise ratios. Within this landscape, organic photochromic materials have emerged as a critical enabler for ultra-high-density optical discs, overcoming the resolution limits inherent in inorganic magneto-optical media. Patent CN1206308C discloses a novel class of diarylethene photochromic compounds that address the historical shortcomings of earlier organic dyes, such as poor thermal stability and low fatigue resistance. These compounds, characterized by a perfluorocyclopentene bridge connecting thiophene rings, demonstrate reversible photoisomerization with high quantum yields, making them ideal candidates for next-generation rewritable optical information storage and optical switching applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of optical storage media relied heavily on inorganic materials like phase-change alloys or organic dyes such as cyanine and phthalocyanine. While these materials served early generations of optical discs, they possess intrinsic defects that hinder their application in ultra-high-density storage systems. Inorganic media often suffer from irregular thermal properties that distort the shape of recorded micro-spots, creating a hard ceiling on storage resolution. Similarly, conventional organic dyes like spiropyrans and fulgides frequently exhibit poor thermal stability in their colored states and degrade rapidly after repeated cycling, a phenomenon known as fatigue. This degradation leads to signal loss and limits the rewrite capability of the storage medium, rendering them unsuitable for archival-quality data retention or high-frequency rewriting scenarios required by modern enterprise storage architectures.
The Novel Approach
The methodology outlined in patent CN1206308C introduces a robust synthetic pathway to diarylethene derivatives that fundamentally resolves these stability issues. By utilizing a perfluorocyclopentene backbone flanked by methyl-substituted thiophene rings, the resulting molecules achieve a planar conformation in the closed-ring state that is thermally irreversible, ensuring data retention without spontaneous erasure. The synthesis strategically employs regioselective bromination and subsequent lithiation to functionalize the thiophene cores, allowing for the precise attachment of various side chains such as dioxolane or nitrobenzyl groups. This modular approach not only enhances the solubility and processability of the final material for thin-film coating but also tunes the absorption characteristics to match specific laser wavelengths, thereby optimizing the signal-to-noise ratio during the read/write process.
Mechanistic Insights into Photochromic 6π-Electrocyclization
The core functionality of these diarylethene compounds relies on a reversible 6π-electrocyclization reaction triggered by electromagnetic radiation. Upon irradiation with ultraviolet light, the molecule undergoes a conrotatory ring-closing reaction, transforming from a colorless open-ring isomer to a highly colored closed-ring isomer. This structural transformation is accompanied by a significant extension of the π-conjugation system across the two thiophene rings and the central cyclopentene bridge, resulting in a bathochromic shift of the absorption maximum into the visible region. Crucially, the presence of fluorine atoms on the central ring and methyl groups at the reactive carbons prevents parallel reactions and stabilizes the closed form against thermal reversion, a common failure mode in earlier photochromic families.
Furthermore, the impurity profile of the final product is tightly controlled through the specific sequence of intermediate purification steps described in the patent. The initial bromination step is conducted under ice-bath conditions to minimize poly-bromination, while the subsequent formation of the boronic acid intermediate utilizes strict temperature control at -78°C to prevent side reactions such as metal-halogen exchange at unintended positions. This rigorous control over reaction parameters ensures that the final diarylethene product possesses high isomeric purity, which is essential for maintaining consistent optical performance in thin-film applications. Any residual impurities could act as quenchers or scatterers, degrading the contrast ratio of the stored data, thus the emphasis on chromatographic purification at multiple stages is a critical quality attribute for commercial viability.
How to Synthesize Diarylethene Photochromic Compounds Efficiently
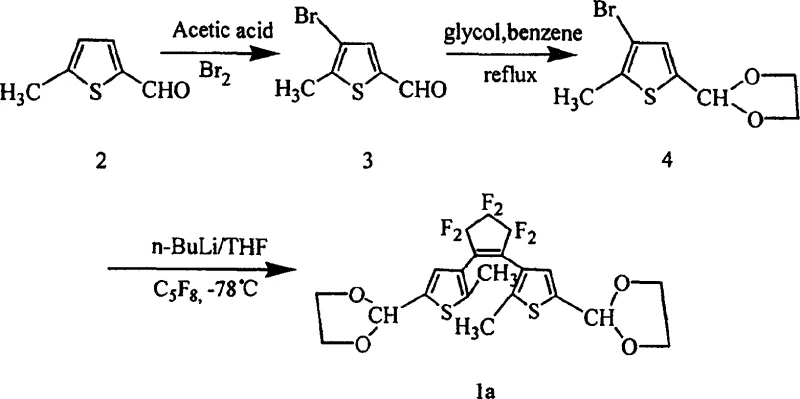
The synthesis of these high-performance materials follows a logical four-step progression designed to maximize yield while minimizing hazardous waste. The process begins with the functionalization of readily available 2-methylthiophene, followed by the construction of the biaryl framework via palladium-catalyzed coupling, and concludes with the formation of the photochromic core using perfluorocyclopentene. This route avoids the use of exotic reagents, relying instead on standard fine chemical building blocks that are accessible through global supply chains. The detailed operational parameters, including solvent choices like THF and benzene, and specific stoichiometric ratios, are optimized to ensure reproducibility at scale.
- Bromination of 2-methylthiophene derivatives under controlled ice-bath conditions to ensure regioselectivity.
- Conversion of the bromo-intermediate to a boronic acid derivative using tributyl borate and n-BuLi at low temperatures.
- Palladium-catalyzed coupling reaction to attach benzyl or heterocyclic groups to the thiophene ring.
- Final cyclization with perfluorocyclopentene using lithiation at -78°C to form the target diarylethene core.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers distinct strategic advantages over sourcing legacy photochromic materials. The reliance on commodity chemicals such as 2-methylthiophene, ethylene glycol, and perfluorocyclopentene reduces exposure to volatile raw material markets and ensures a stable supply base. Furthermore, the elimination of complex chiral catalysts or rare earth metals simplifies the waste treatment process, significantly lowering the environmental compliance burden associated with manufacturing. The robustness of the reaction conditions, particularly the tolerance of the coupling steps to standard workup procedures, facilitates a streamlined production workflow that minimizes batch-to-batch variability.
- Cost Reduction in Manufacturing: The synthetic pathway eliminates the need for expensive transition metal catalysts in the final cyclization step, relying instead on organolithium reagents which are cost-effective at industrial scales. By avoiding the use of precious metals like platinum or rhodium in the core ring-closing reaction, the overall bill of materials is drastically reduced. Additionally, the high regioselectivity of the initial bromination step minimizes the formation of difficult-to-separate isomers, reducing the load on downstream purification units and lowering solvent consumption costs significantly.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, including thiophene derivatives and perfluorocyclopentene, are produced by multiple established chemical manufacturers globally, mitigating the risk of single-source supply disruptions. The process does not depend on proprietary enzymes or biologically derived reagents that might suffer from shelf-life issues or cold-chain logistics requirements. This chemical stability allows for bulk purchasing and long-term inventory storage, providing procurement teams with greater flexibility in managing cash flow and hedging against market price fluctuations.
- Scalability and Environmental Compliance: The reaction steps utilize common organic solvents such as tetrahydrofuran, ether, and benzene, which are easily recovered and recycled using standard distillation infrastructure found in most fine chemical plants. The absence of heavy metal residues in the final product simplifies the disposal of aqueous waste streams, aligning with increasingly stringent environmental regulations regarding heavy metal discharge. This ease of waste management translates to lower operational expenditures for effluent treatment and reduces the regulatory hurdles for expanding production capacity to meet growing market demand.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photochromic technology in optical storage devices. These answers are derived directly from the experimental data and performance metrics disclosed in the patent documentation, providing a factual basis for feasibility assessments.
Q: What are the key stability advantages of these diarylethene compounds compared to traditional dyes?
A: Unlike azo or spiropyran dyes which suffer from thermal instability and fatigue, these diarylethene derivatives exhibit exceptional thermal stability in both open and closed ring states, along with remarkable fatigue resistance suitable for over 10,000 write/erase cycles.
Q: Can this synthesis route be scaled for industrial production of optical storage media?
A: Yes, the process utilizes standard organic synthesis techniques such as bromination, Suzuki coupling, and lithiation which are well-established in fine chemical manufacturing, allowing for reliable scale-up from laboratory to commercial tonnage.
Q: What is the absorption range of the colored state for these materials?
A: The colored closed-ring state exhibits an absorption spectrum located between 400nm and 600nm, which aligns perfectly with standard 532nm laser diodes used in optical recording devices.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diarylethene Photochromic Compound Supplier
As the demand for high-density optical storage and smart optical switches continues to grow, securing a supply of high-purity photochromic intermediates is paramount for product success. NINGBO INNO PHARMCHEM leverages extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to deliver these complex molecules with consistent quality. Our facility is equipped with rigorous QC labs and stringent purity specifications to ensure that every batch meets the exacting standards required for electronic grade materials, guaranteeing optimal performance in your final optical devices.
We invite you to engage with our technical procurement team to discuss how we can optimize your supply chain for these advanced materials. By requesting a Customized Cost-Saving Analysis, you can gain insights into potential process improvements and volume-based pricing structures tailored to your specific production needs. We encourage you to contact us today to obtain specific COA data and route feasibility assessments that will accelerate your product development timeline.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
