Advanced Synthesis of Pyrimido-Triazole Hydrazine Compounds for Commercial Scale-Up of Complex Anticancer Intermediates
Advanced Synthesis of Pyrimido-Triazole Hydrazine Compounds for Commercial Scale-Up of Complex Anticancer Intermediates
The landscape of oncology drug discovery is constantly evolving, with epigenetic regulators emerging as pivotal targets for next-generation therapeutics. Patent CN109293664B introduces a novel class of pyrimido[1,2,4]-triazole hydrazine compounds that exhibit significant potential as Lysine-Specific Demethylase 1 (LSD1) inhibitors. These compounds are characterized by a unique fused heterocyclic scaffold that allows for precise modulation of gene expression in tumor cells. As a leading entity in fine chemical manufacturing, we recognize the critical importance of securing a reliable supply chain for such high-value pharmaceutical intermediates. The structural versatility offered by the variable R1 and R2 groups in the general formula enables extensive Structure-Activity Relationship (SAR) studies, facilitating the rapid optimization of potency and selectivity for clinical candidates.
![General structural formulas I and II of pyrimido[1,2,4]-triazole hydrazine compounds showing variable R1 and R2 substituents](/insights/img/pyrimido-triazole-hydrazine-lsd1-inhibitor-intermediate-supplier-20260312001316-01.webp)
The significance of this technology extends beyond mere molecular novelty; it represents a robust platform for developing treatments against aggressive malignancies such as gastric cancer, lung cancer, and leukemia. LSD1 is known to be abnormally highly expressed in these tumor types, where it promotes proliferation and metastasis by regulating chromatin dynamics. By effectively inhibiting LSD1, these pyrimido-triazole derivatives can restore normal gene function and induce apoptosis in cancer cells. For procurement managers and R&D directors alike, accessing a stable source of these intermediates is paramount to accelerating preclinical and clinical development timelines without compromising on quality or regulatory compliance standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for constructing complex fused triazole systems often suffer from significant drawbacks that hinder their industrial applicability. Many conventional methods rely on harsh reaction conditions, such as extremely high temperatures or the use of hazardous reagents that pose safety risks and environmental challenges. Furthermore, older methodologies frequently involve multiple protection and deprotection steps, which drastically reduce the overall atom economy and increase the generation of chemical waste. In the context of LSD1 inhibitor synthesis, legacy routes may struggle with regioselectivity issues, leading to difficult-to-separate impurities that compromise the purity profile required for pharmaceutical applications. These inefficiencies translate directly into higher production costs and longer lead times, creating bottlenecks for supply chain heads who need to ensure continuous material flow for drug development programs.
The Novel Approach
The synthesis method disclosed in patent CN109293664B offers a transformative solution by streamlining the construction of the pyrimido[1,2,4]-triazole core through a highly efficient sequence. This novel approach leverages a strategic cyclization followed by a selective chlorination and subsequent nucleophilic substitution, avoiding the need for complex transition metal catalysts that often require expensive removal processes. The use of common industrial solvents like ethanol and glacial acetic acid simplifies the workup procedures and facilitates solvent recovery, contributing to substantial cost savings in pharmaceutical intermediate manufacturing. Moreover, the reaction conditions are mild enough to preserve sensitive functional groups while ensuring high conversion rates, as evidenced by the reported total yields exceeding 56 percent. This feasibility makes the route exceptionally attractive for commercial scale-up, allowing manufacturers to produce high-purity intermediates with consistent quality batch after batch.
Mechanistic Insights into Hydrazine Substitution and Heterocyclic Formation
The core of this synthetic strategy lies in the precise formation of the pyrimido-triazole ring system and the subsequent installation of the hydrazine moiety. The process begins with the S-alkylation of 2-amino-5-mercapto-1,2,4-triazole, which serves as a versatile building block for introducing diverse side chains via the sulfur atom. This intermediate then undergoes a condensation reaction with a beta-keto acid ester, driven by thermal energy in an acidic medium, to close the pyrimidine ring. The resulting bicyclic structure possesses a reactive carbonyl group that is subsequently activated for substitution. The critical step involves the conversion of the carbonyl or hydroxyl functionality into a good leaving group, typically a chlorine atom, using phosphorus oxychloride. This activation is essential for enabling the final nucleophilic attack by hydrazine.
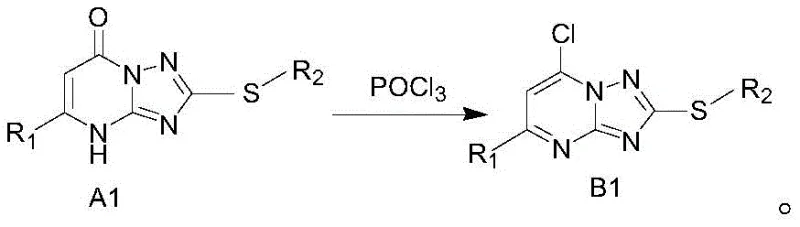
Impurity control is meticulously managed throughout this mechanistic pathway, particularly during the final substitution step where hydrazine hydrate reacts with the chlorinated intermediate. The reaction is conducted at normal temperature in ethanol, which minimizes the risk of over-reaction or decomposition of the sensitive hydrazine group. By carefully monitoring the reaction progress via Thin Layer Chromatography (TLC), operators can ensure complete conversion of the starting material while preventing the formation of bis-substituted byproducts. The high selectivity of this nucleophilic aromatic substitution ensures that the final product maintains the integrity of the triazole ring, which is crucial for its biological activity as an LSD1 inhibitor. This level of mechanistic control is vital for R&D teams aiming to produce reference standards and clinical trial materials with defined impurity profiles.
How to Synthesize Pyrimido-Triazole Hydrazine Efficiently
The synthesis of these high-value intermediates follows a logical four-step progression that balances reactivity with operational simplicity. Starting from commercially available 2-amino-5-mercapto-1,2,4-triazole, the process builds molecular complexity in a controlled manner. The initial alkylation sets the stage for diversity, while the subsequent cyclization establishes the core pharmacophore. The chlorination step activates the molecule for the final functionalization, which introduces the key hydrazine group responsible for target engagement. Detailed standard operating procedures for each step, including specific molar ratios, temperature controls, and purification techniques, are essential for reproducing the high yields reported in the patent literature.
- Perform S-alkylation of 2-amino-5-mercapto-1,2,4-triazole with alkyl chloride under alkaline conditions to form the thioether intermediate.
- React the thioether intermediate with a beta-keto acid ester in glacial acetic acid at elevated temperatures to achieve cyclization and form the pyrimido-triazole core.
- Execute chlorination using phosphorus oxychloride followed by nucleophilic substitution with hydrazine hydrate to yield the final hydrazine compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented synthesis route offers distinct strategic advantages that go beyond simple chemical transformation. The elimination of expensive transition metal catalysts and the reliance on commodity chemicals significantly lowers the raw material cost base, allowing for more competitive pricing in the long term. Additionally, the robustness of the reaction conditions means that the process is less susceptible to variations in ambient conditions or minor fluctuations in reagent quality, thereby enhancing supply chain reliability. This stability is crucial for maintaining uninterrupted production schedules, especially when scaling up from laboratory grams to commercial metric tons. The simplified purification steps also reduce the consumption of chromatography media and solvents, further driving down the operational expenditure associated with manufacturing these complex intermediates.
- Cost Reduction in Manufacturing: The synthetic route is designed to maximize atom economy and minimize waste generation, which directly translates to lower disposal costs and reduced raw material consumption. By avoiding the use of precious metal catalysts, the process eliminates the need for costly metal scavenging steps and rigorous testing for residual metals, which are often required for regulatory compliance in pharmaceutical production. Furthermore, the high yields achieved at each step reduce the amount of starting material needed per kilogram of final product, optimizing the overall cost structure. This efficiency allows manufacturers to offer significant cost savings to their clients without compromising on the stringent quality standards required for oncology drug intermediates.
- Enhanced Supply Chain Reliability: The reliance on widely available and stable starting materials ensures that the supply chain is resilient against market volatility and geopolitical disruptions. Unlike routes that depend on exotic or single-source reagents, this method utilizes commodity chemicals that can be sourced from multiple suppliers globally. This diversification of the supply base mitigates the risk of shortages and ensures consistent availability of the intermediate. Moreover, the scalability of the process means that production capacity can be rapidly expanded to meet surging demand from clinical trials or commercial launch phases, providing peace of mind to supply chain heads who are tasked with securing long-term material availability.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to work effectively from small laboratory scales to larger pilot batches without significant loss in efficiency. The use of standard reactor equipment and common solvents facilitates easy technology transfer between different manufacturing sites. From an environmental perspective, the reduced generation of hazardous waste and the avoidance of heavy metals align with modern green chemistry principles and increasingly strict environmental regulations. This compliance not only reduces the regulatory burden but also enhances the sustainability profile of the supply chain, which is becoming a key criterion for selection by major pharmaceutical companies seeking responsible partners.
Frequently Asked Questions (FAQ)
Understanding the technical nuances and commercial implications of this technology is essential for stakeholders involved in drug development. The following questions address common inquiries regarding the synthesis, application, and supply of these pyrimido-triazole hydrazine compounds. The answers are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for decision-makers.
Q: What is the primary therapeutic target of these pyrimido-triazole compounds?
A: These compounds are designed as potent inhibitors of Lysine-Specific Demethylase 1 (LSD1), an epigenetic regulator highly expressed in various tumor cells such as gastric and lung cancer.
Q: How does this synthesis route improve upon conventional methods?
A: The patented route utilizes readily available starting materials and avoids complex transition metal catalysis, resulting in a feasible process with a total yield exceeding 56% and simplified purification.
Q: Are these intermediates suitable for large-scale production?
A: Yes, the synthesis employs robust reaction conditions such as reflux in common solvents like ethanol and acetic acid, making it highly amenable to commercial scale-up from kilograms to metric tons.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrimido-Triazole Hydrazine Supplier
At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from discovery to market. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications required for oncology intermediates. We understand the critical nature of LSD1 inhibitor development and are committed to providing a reliable supply of high-quality pyrimido-triazole hydrazine compounds that adhere to the highest industry standards. Our team of expert chemists is ready to assist with process optimization and custom synthesis to meet your specific project requirements.
We invite you to contact our technical procurement team to request specific COA data and route feasibility assessments tailored to your needs. By partnering with us, you gain access to a Customized Cost-Saving Analysis that highlights how our optimized manufacturing processes can reduce your overall development budget. Let us help you accelerate your drug discovery timeline with our dependable supply chain and technical expertise.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
