Advanced Manufacturing of Esomeprazole Magnesium: A Cost-Effective Route for Global Supply Chains
The global demand for proton pump inhibitors (PPIs) remains robust, driven by the increasing prevalence of gastroesophageal reflux disease (GORD) and peptic ulcers. At the forefront of this therapeutic class is esomeprazole, the S-isomer of omeprazole, which offers superior pharmacokinetic profiles compared to its racemic counterpart. A pivotal advancement in the manufacturing landscape is detailed in patent CN103709143A, which discloses a highly efficient preparation method for esomeprazole and its magnesium salt. This technical disclosure outlines a streamlined synthetic pathway that begins with the readily available 4-methoxy-2-hydroxymethyl-3,5-dimethyl pyridine. By optimizing reaction conditions across bromination, nucleophilic substitution, and crucial asymmetric oxidation steps, this methodology addresses long-standing challenges in impurity control and cost efficiency. For pharmaceutical manufacturers, adopting such refined processes is not merely a technical upgrade but a strategic imperative to ensure supply continuity and margin protection in a competitive generic market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for esomeprazole often suffer from significant drawbacks that hinder large-scale commercial viability. Many legacy processes rely on expensive chiral resolving agents or complex enzymatic resolutions that drive up the cost of goods sold (COGS) substantially. Furthermore, conventional oxidation steps frequently utilize harsh oxidants that lack selectivity, leading to the formation of difficult-to-remove sulfone impurities and racemization of the chiral center. These side reactions necessitate extensive downstream purification, such as multiple recrystallizations or chromatographic separations, which drastically reduce overall yield and increase solvent consumption. Additionally, older methods may involve unstable intermediates that require cryogenic conditions throughout the entire synthesis, imposing heavy energy burdens on production facilities and complicating safety protocols regarding thermal runaway risks.
The Novel Approach
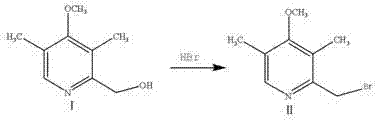
The methodology presented in the referenced patent introduces a robust alternative that mitigates these inefficiencies through a rational design of the synthetic sequence. By initiating the synthesis with 4-methoxy-2-hydroxymethyl-3,5-dimethyl pyridine, the process leverages a stable and cost-effective starting material that eliminates the need for complex upstream protection-deprotection strategies. The subsequent conversion to the bromo-intermediate is achieved under controlled heating, ensuring high conversion rates without excessive degradation. Crucially, the formation of the thioether linkage is conducted under mild conditions (0-30°C) in a methanol medium, which simplifies the reaction setup and allows for straightforward isolation of the intermediate. This approach not only enhances the safety profile of the manufacturing process but also significantly reduces the environmental footprint by minimizing waste generation and solvent usage compared to more archaic techniques.
Mechanistic Insights into Kagan-Modified Asymmetric Oxidation
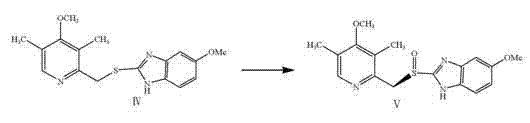
The cornerstone of this synthesis is the enantioselective oxidation of the sulfide intermediate to the corresponding sulfoxide, a transformation that dictates the optical purity of the final API. The patent employs a modified Sharpless-Kagan oxidation system, utilizing titanium tetraisopropoxide (or tetrabutyl titanate as specified) coordinated with D-diethyl tartrate (DET) as the chiral ligand. In this catalytic cycle, the titanium center acts as a Lewis acid, coordinating with both the chiral tartrate and the sulfide substrate to create a rigid chiral environment. When the oxidant, typically an organic peroxide like cumene hydroperoxide derivatives, is introduced, the oxygen transfer occurs preferentially to one face of the sulfur atom. This stereochemical control is vital, as the biological activity of esomeprazole is exclusively associated with the (S)-enantiomer. The precise stoichiometry of the titanium catalyst, chiral ligand, and water content is critical; the patent specifies molar ratios that optimize the formation of the active catalytic species while suppressing non-selective background oxidation.
Impurity control in this oxidation step is paramount for meeting regulatory specifications. The primary impurities of concern are the achiral sulfone (over-oxidation product) and the (R)-enantiomer. The described process manages these risks through strict temperature programming. The reaction is initiated at moderate temperatures (40-70°C) to facilitate the formation of the titanium-tartrate complex, then cooled significantly to -20 to 0°C prior to oxidant addition. This low-temperature regime slows down the reaction kinetics just enough to favor the lower-energy transition state leading to the desired (S)-sulfoxide, thereby maximizing enantiomeric excess (ee). Furthermore, the choice of oxidant and the addition rate are tuned to prevent local exotherms that could trigger racemization. Post-reaction, the protocol includes a specific workup involving organic bases and filtration, which effectively removes titanium residues and tartrate byproducts, ensuring the crude esomeprazole obtained is of sufficient quality for final recrystallization.
How to Synthesize Esomeprazole Magnesium Efficiently
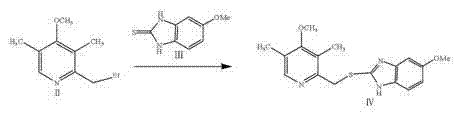
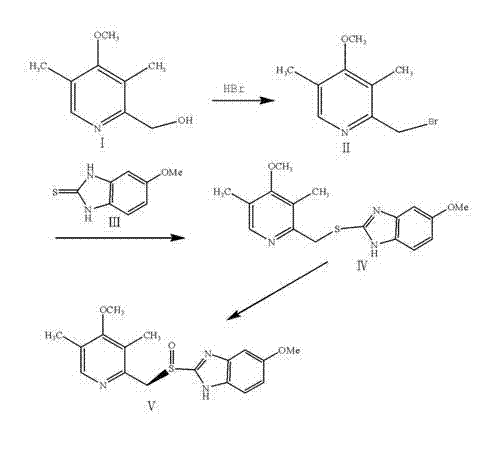
The synthesis of esomeprazole magnesium via this patented route involves a logical progression of four distinct chemical transformations, each optimized for scalability and yield. The process begins with the activation of the pyridine methanol group, followed by coupling with the benzimidazole core, chiral oxidation, and finally salt formation. This sequence is designed to minimize unit operations and maximize throughput in a multi-purpose reactor setup. The detailed standard operating procedures (SOPs) for temperature ramps, addition rates, and crystallization parameters are critical for reproducing the high purity described in the patent documentation. For process chemists looking to implement this technology, understanding the nuances of the workup phases—particularly the solvent swaps and drying conditions—is essential to preventing product degradation.
- Bromination of 4-methoxy-2-hydroxymethyl-3,5-dimethyl pyridine with hydrobromic acid at 80-130°C to form the bromo-intermediate.
- Nucleophilic substitution reaction with 5-methoxy-2-mercaptobenzimidazole in methanol at 0-30°C to generate the thioether compound.
- Asymmetric oxidation using D-diethyl tartrate, tetrabutyl titanate, and an organic peroxide oxidant at controlled low temperatures (-20 to 10°C).
- Salt formation by reacting the purified esomeprazole with magnesium methoxide solution followed by crystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis route offers tangible strategic benefits beyond mere technical feasibility. The primary advantage lies in the significant reduction of production costs driven by the optimization of raw material utilization and process efficiency. By utilizing 4-methoxy-2-hydroxymethyl-3,5-dimethyl pyridine as a starting point, manufacturers can bypass expensive chiral pool precursors, relying instead on a resolution-free asymmetric synthesis that delivers high optical purity directly. This shift eliminates the 50% theoretical yield loss inherent in classical resolution methods, effectively doubling the output per kilogram of starting material and drastically lowering the cost basis for the active pharmaceutical ingredient (API).
- Cost Reduction in Manufacturing: The economic impact of this process is profound due to the elimination of costly separation steps and the use of commodity chemicals. The bromination and substitution steps utilize hydrobromic acid and sodium hydroxide, which are inexpensive and widely available bulk chemicals. Furthermore, the simplified post-reaction treatment, which often involves simple filtration rather than complex extractions or chromatography, reduces labor hours and solvent disposal costs. The ability to recycle solvents like methanol and dichloromethane further enhances the margin profile, making the final esomeprazole magnesium highly competitive in price-sensitive markets.
- Enhanced Supply Chain Reliability: Supply continuity is bolstered by the reliance on robust, non-proprietary reagents. Unlike processes dependent on specialized enzymes or scarce chiral auxiliaries that may face supply bottlenecks, this method uses standard industrial chemicals with multiple global suppliers. The reaction conditions are also forgiving enough to allow for flexible scheduling; the intermediate thioether is stable enough to be stored or transported if necessary, decoupling the oxidation step from the upstream synthesis. This modularity allows supply chain planners to build inventory buffers at the intermediate stage, mitigating the risk of production stoppages due to equipment maintenance or raw material delays.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this route is superior for large-scale manufacturing. The avoidance of heavy metal catalysts (other than recoverable titanium) and the use of milder oxidants reduce the toxicity of the waste stream. The process operates at pressures close to atmospheric, removing the need for high-pressure autoclaves and reducing capital expenditure requirements for new production lines. The simplified crystallization protocols for the final magnesium salt ensure consistent particle size distribution, which is critical for downstream formulation, while the high purity achieved reduces the risk of batch rejection during quality control testing.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation of this synthesis pathway. These insights are derived directly from the experimental data and claims within the patent specification, providing clarity on critical process parameters. Understanding these details is essential for technology transfer teams evaluating the feasibility of integrating this route into existing manufacturing infrastructure.
Q: What are the critical temperature controls for the asymmetric oxidation step?
A: The oxidation step requires precise thermal management, initially stirring at 40-70°C for complexation, then cooling to -20 to 0°C before adding the oxidant, and finally warming to 5-10°C for the reaction completion to ensure high enantiomeric purity.
Q: Which oxidants are suitable for this specific esomeprazole synthesis route?
A: The patent specifies the use of organic peroxides such as tert-amyl hydroperoxide, 3,3'-bis-(tert-pentyl peroxide) ethyl butyrate, or peroxydisuccinic acid to achieve efficient sulfoxidation.
Q: How is the final magnesium salt purified to meet pharmaceutical standards?
A: The crude esomeprazole magnesium is recrystallized using a methanol and acetone solvent system, specifically adding acetone to a methanol solution at 0-5°C, followed by vacuum drying at 60°C to remove residual solvents and ensure stability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Esomeprazole Magnesium Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory patent to commercial reality requires more than just chemical knowledge; it demands engineering excellence and rigorous quality assurance. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the intricate temperature controls and stoichiometric balances required for the asymmetric oxidation of esomeprazole are maintained with precision at every scale. We operate stringent purity specifications and utilize rigorous QC labs equipped with advanced chiral HPLC and NMR capabilities to guarantee that every batch of esomeprazole magnesium meets the highest international pharmacopeial standards for enantiomeric excess and impurity profiles.
We invite global partners to collaborate with us to leverage this cost-effective manufacturing technology for their supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage potential clients to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that optimize both the quality and economics of your proton pump inhibitor portfolio.
