Advanced Microbial Dehydrogenation Technology for Scalable Hormone Intermediate Production
Advanced Microbial Dehydrogenation Technology for Scalable Hormone Intermediate Production
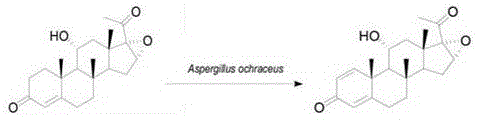
The pharmaceutical industry continuously seeks robust and scalable methodologies for synthesizing complex steroidal intermediates, particularly those serving as precursors for critical hormone therapies. A significant breakthrough in this domain is detailed in patent CN106520890B, which discloses a highly efficient preparation method for 11 Alpha-hydroxy-16 α, 17 α-epoxy progesterone dehydrogen substance. This innovation leverages a specialized two-phase fermentation system utilizing sterile water and toluene, coupled with a toluene-tolerant strain of Aspergillus ochraceus. By fundamentally altering the solvation environment of the substrate, this technology overcomes the historical bottleneck of poor substrate solubility in aqueous media, enabling a dramatic increase in the C1,2-dehydrogenation reaction efficiency. For R&D directors and process chemists, this represents a pivotal shift from cumbersome chemical oxidation or low-yield single-phase fermentation towards a streamlined, high-conversion biocatalytic process that is inherently safer and more environmentally compliant.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the microbial dehydrogenation of steroidal compounds such as 11 Alpha-hydroxy-16 α, 17 α-epoxy progesterone has been plagued by intrinsic physicochemical limitations. In traditional single-phase aqueous fermentation systems, the substrate exhibits extremely low solubility, typically ranging between 1 × 10-5 mol/L and 1 × 10-6 mol/L. This insolubility creates a massive mass transfer barrier, preventing the steroid molecules from effectively contacting the intracellular biological enzymes responsible for the C1,2-dehydrogenation. To mitigate this, prior art has attempted various physical and chemical modifications, including cyclodextrin embedding, substrate particle size reduction, ultrasonication, and microemulsification. However, these methods often introduce significant complexity and cost; for instance, cyclodextrins are expensive inclusion agents that complicate downstream purification, while ultrasonication is difficult to scale industrially. Consequently, these conventional approaches frequently result in prolonged conversion times, inconsistent yields, and a final product profile that struggles to meet the stringent purity requirements of modern API manufacturing.
The Novel Approach
The methodology described in the patent introduces a paradigm shift by employing a biphasic solvent system comprising sterile water as the continuous phase and toluene as the organic phase. In this novel configuration, the hydrophobic steroid substrate is dissolved directly into the toluene phase at a molecular level, creating a high-concentration reservoir of reactant in immediate proximity to the aqueous-microbial interface. This setup utilizes a specifically screened strain of Aspergillus ochraceus that possesses tolerance to toluene, allowing the microorganisms to thrive and function effectively at the phase boundary. The result is a substantial increase in the effective contact area between the substrate and the biological enzyme, which drastically shortens the conversion time. Unlike the scattering of solid particles in water, the molecular dissolution in toluene ensures that the reaction kinetics are driven by concentration gradients rather than diffusion limits, leading to conversion ratios that can reach as high as 99.1% within a concise 20 to 30-hour fermentation window.
Mechanistic Insights into Aspergillus Ochraceus-Catalyzed Dehydrogenation
The core mechanism driving this transformation is the interfacial biocatalysis facilitated by the unique physiology of the toluene-tolerant Aspergillus ochraceus. In standard aqueous environments, the cell membrane acts as a barrier to hydrophobic steroids, but in the presence of a water-immiscible organic solvent like toluene, the cellular machinery adapts to utilize the organic-aqueous interface as the primary reaction zone. The enzyme systems within the fungus, likely involving flavin-dependent oxidoreductases, abstract hydrogen atoms from the C1 and C2 positions of the steroid A-ring. The presence of toluene not only solubilizes the substrate but may also influence the permeability of the cell membrane, facilitating the transport of the hydrophobic substrate into the enzymatic active sites or allowing the enzyme to act at the cell surface. This mechanistic advantage is evidenced by the rapid formation of the conjugated enone system (the Δ1,4-3-ketone structure), which is thermodynamically stable and crucial for the biological activity of downstream corticosteroid drugs. The specificity of the microbial catalyst ensures that dehydrogenation occurs exclusively at the C1,2-position without affecting other sensitive functional groups like the 11α-hydroxyl or the 16α,17α-epoxy ring.
Furthermore, the impurity control mechanism in this two-phase system is superior to chemical alternatives. Chemical dehydrogenation often requires harsh reagents like selenium dioxide or DDQ, which can lead to over-oxidation or rearrangement of the steroid skeleton. In contrast, the biological system operates under mild conditions (25°C to 48°C) and neutral pH, preserving the stereochemical integrity of the molecule. The patent data indicates that the process yields a crude product with high selectivity, which is further purified through a targeted recrystallization process using chloroform and ethanol. This solvent pair is selected based on the differential solubility of the dehydrogenated product versus the unreacted starting material. The dehydrogenated substance precipitates with high efficiency, while the unreacted 11 Alpha-hydroxy-16 α, 17 α-epoxy progesterone remains largely in the mother liquor, allowing for its recovery and recycling. This dual outcome of high product purity (>98%) and significant substrate recovery (>5-10%) minimizes waste generation and maximizes atom economy, a key metric for green chemistry in pharmaceutical synthesis.
How to Synthesize 11 Alpha-hydroxy-16 alpha, 17 alpha-epoxy Progesterone Dehydrogen Substance Efficiently
Implementing this biocatalytic route requires precise control over strain preparation and phase ratios to ensure reproducibility at scale. The process begins with the activation of the Aspergillus ochraceus spores on potato slope culture media, followed by expansion in a liquid nutrient medium to generate a robust mycelial biomass capable of tolerating organic solvents. The critical step involves the creation of the two-phase fermentation liquid, where the substrate-loaded toluene is emulsified into the aqueous microbial suspension under mechanical stirring. This agitation is vital to maintain a high surface-area-to-volume ratio, ensuring that the toluene droplets containing the steroid are constantly renewed at the interface with the fungal cells. Following the incubation period, the separation of the product relies on filtration and selective crystallization, a unit operation that is well-understood in industrial settings.
- Prepare the toluene-tolerant Aspergillus ochraceus strain by culturing on potato slope and yeast extract media, followed by suspension in sterile water.
- Dissolve the substrate (11 Alpha-hydroxy-16 alpha, 17 alpha-epoxy progesterone) in toluene to form an organic phase, then mix with the aqueous microbial suspension to create a two-phase fermentation liquid.
- Incubate the mixture at 25-48°C with mechanical stirring for 20-30 hours, then filter and recrystallize the product using chloroform/ethanol to isolate the high-purity dehydrogenated substance.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this two-phase microbial fermentation technology offers compelling economic and operational benefits that extend beyond simple yield improvements. The elimination of expensive solubilizing agents like cyclodextrins directly translates to a reduction in raw material costs, as the process relies on commodity solvents like toluene and standard fermentation nutrients. Additionally, the simplified downstream processing—avoiding complex extraction protocols associated with emulsions or solid dispersions—reduces the consumption of energy and auxiliary chemicals. The high conversion efficiency means that less starting material is required per kilogram of finished product, effectively lowering the cost of goods sold (COGS) for this critical intermediate. From a supply chain perspective, the robustness of the toluene-tolerant strain ensures consistent batch-to-batch performance, reducing the risk of production delays caused by failed fermentations or low titers.
- Cost Reduction in Manufacturing: The process significantly lowers manufacturing expenses by removing the need for costly inclusion complexes and specialized physical treatment equipment like ultrasonic processors. The use of a recyclable organic solvent system allows for the recovery of unreacted starting material, which can be fed back into the process, thereby maximizing the utility of every gram of expensive steroid precursor purchased. Furthermore, the mild reaction conditions reduce energy consumption for heating or cooling compared to exothermic chemical oxidation reactions, contributing to a leaner operational budget.
- Enhanced Supply Chain Reliability: By utilizing a fermentation-based approach with high conversion rates (consistently above 98%), manufacturers can secure a more predictable supply of high-purity intermediates. The ability to recover and recycle unreacted substrate acts as a buffer against fluctuations in raw material availability, ensuring that production targets are met even if initial substrate quality varies slightly. This reliability is crucial for maintaining the continuity of supply for downstream API synthesis, preventing bottlenecks in the production of life-saving hormone medications.
- Scalability and Environmental Compliance: The technology is designed for industrial scalability, utilizing standard stirred-tank reactors that are common in existing fermentation facilities. The avoidance of heavy metal catalysts and toxic oxidants aligns with increasingly strict environmental regulations regarding wastewater discharge and hazardous waste disposal. The solvent system, primarily consisting of toluene and alcohols, can be managed through established distillation and recovery units, minimizing the environmental footprint and simplifying the permitting process for large-scale commercial production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented biocatalytic technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a clear understanding of the operational parameters and expected outcomes. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for their own manufacturing pipelines.
Q: Why is a two-phase toluene-water system superior to traditional aqueous fermentation for this reaction?
A: Traditional aqueous fermentation suffers from the extremely low solubility of steroidal substrates (approx. 1x10^-5 to 1x10^-6 mol/L), which limits contact with intracellular enzymes. The two-phase system dissolves the substrate in toluene, drastically increasing the interfacial contact area with the microorganism, thereby accelerating the C1,2-dehydrogenation rate and improving overall conversion efficiency.
Q: What are the critical parameters for maintaining high conversion rates in this biocatalytic process?
A: Critical parameters include maintaining the inoculum concentration of the toluene-tolerant Aspergillus ochraceus between 7% and 10% of the total fermentation volume, controlling the substrate concentration in the range of 1 g/100ml to 2 g/100ml, and ensuring mechanical stirring at temperatures between 25°C and 48°C to facilitate mass transfer between the organic and aqueous phases.
Q: How does this method address the impurity profile compared to chemical dehydrogenation?
A: Microbial catalysis using specific strains like Aspergillus ochraceus offers high regioselectivity for the C1,2-position, minimizing side reactions common in chemical dehydrogenation. Furthermore, the subsequent recrystallization using chloroform/ethanol mixed solvents effectively separates the product from unreacted starting material, achieving purity levels greater than 98% without the need for complex chromatographic purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 11 Alpha-hydroxy-16 alpha, 17 alpha-epoxy Progesterone Dehydrogen Substance Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the synthesis of advanced hormonal therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 11 Alpha-hydroxy-16 α, 17 α-epoxy progesterone dehydrogen substance meets the exacting standards required by global regulatory bodies. Our commitment to technological excellence allows us to offer a reliable pharmaceutical intermediate supplier partnership that prioritizes both quality and consistency.
We invite you to collaborate with us to leverage this advanced biocatalytic technology for your specific project needs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your current production volumes. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized processes can enhance your supply chain resilience and drive down your overall manufacturing costs.
