Advanced Synthesis of Alkylporphyrin Photosensitizers for Commercial Scale-Up
Advanced Synthesis of Alkylporphyrin Photosensitizers for Commercial Scale-Up
The landscape of photodynamic therapy (PDT) has evolved significantly with the demand for second-generation photosensitizers that offer defined chemical structures and superior pharmacokinetic profiles compared to first-generation mixtures like Photofrin II. Patent CN101591341A introduces a robust synthetic methodology for producing a novel class of alkylporphyrin compounds, specifically designed to overcome the solubility and selectivity limitations of earlier derivatives. These compounds, characterized by amino-substituted alkyl chains at the meso-positions of the porphyrin ring, demonstrate exceptional potential as both antitumor agents and photoactivated insecticides. For R&D directors and procurement specialists in the pharmaceutical sector, this technology represents a critical opportunity to access high-purity intermediates with streamlined manufacturing protocols. The synthesis leverages accessible raw materials and mild reaction conditions, ensuring that the transition from laboratory discovery to commercial production is both technically feasible and economically viable for global supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for porphyrin-based photosensitizers often rely on harsh acidic conditions using liquid Lewis acids such as boron trifluoride etherate or strong mineral acids, which pose significant challenges for large-scale manufacturing. These conventional methods frequently result in complex reaction mixtures that require extensive and costly purification processes to remove residual metals and acidic byproducts, thereby inflating the overall production cost. Furthermore, many legacy processes struggle to control the regioselectivity of substitution, leading to isomeric impurities that can compromise the biological efficacy and safety profile of the final drug substance. The environmental burden associated with disposing of large volumes of corrosive liquid acid waste also conflicts with modern green chemistry principles, creating regulatory hurdles for sustainable production facilities.
The Novel Approach
The innovative methodology described in the patent utilizes Montmorillonite K10, a solid clay-based Lewis acid, to catalyze the condensation of pyrrole and 6-chloro-1-hexanal, offering a cleaner and more manageable alternative to liquid catalysts. This solid acid approach not only simplifies the reaction workup by allowing for easy filtration of the catalyst but also minimizes the generation of hazardous liquid waste, aligning with stringent environmental compliance standards. Following the initial condensation, the protocol employs chloranil (tetrachloro-1,4-benzoquinone) as a mild oxidizing agent to aromatize the porphyrinogen intermediate efficiently. This two-step sequence ensures high structural fidelity and reproducibility, providing a reliable foundation for the subsequent functionalization with various amine groups to tune the physicochemical properties of the final photosensitizer.
Mechanistic Insights into Lewis Acid-Catalyzed Porphyrin Formation
The core of this synthetic strategy lies in the acid-catalyzed condensation mechanism where the Lewis acid activates the carbonyl group of 6-chloro-1-hexanal, facilitating nucleophilic attack by the pyrrole nitrogen. This interaction promotes the formation of dipyrromethane intermediates which subsequently oligomerize to form the macrocyclic porphyrinogen structure. The use of Montmorillonite K10 is particularly advantageous as its layered silicate structure provides a unique microenvironment that can stabilize transition states and enhance reaction rates without the aggressive degradation often seen with stronger liquid acids. The subsequent oxidation step is critical, as it converts the non-aromatic porphyrinogen into the fully conjugated, aromatic porphine system responsible for the compound's light-absorbing capabilities. This precise control over the oxidation state ensures that the final product possesses the necessary electronic properties for effective singlet oxygen generation upon light irradiation.
Following the formation of the chloro-substituted porphyrin intermediate, the synthesis proceeds via a nucleophilic substitution reaction where the terminal chlorine atoms are displaced by secondary amines such as morpholine or pyrrolidine. This transformation is facilitated by the presence of potassium carbonate and potassium iodide in acetonitrile, where the iodide acts as a catalyst to enhance the leaving group ability of the chloride. The resulting amino-alkyl side chains introduce basic nitrogen centers that can be protonated at physiological pH, thereby imparting water solubility to the otherwise hydrophobic porphyrin core. This amphiphilic nature is crucial for biological applications, as it allows the molecule to traverse lipid membranes while remaining soluble in the aqueous environment of the bloodstream, ensuring efficient delivery to target tumor tissues.
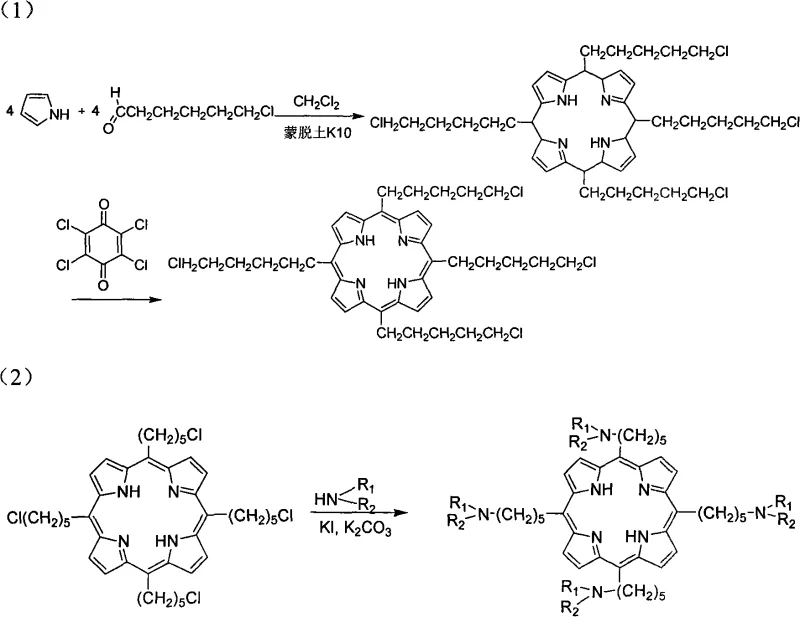
How to Synthesize 5,10,15,20-Tetrakis(5-aminopentyl)-Porphines Efficiently
The preparation of these high-value alkylporphyrin compounds follows a logical and scalable two-stage process that begins with the construction of the porphyrin macrocycle and concludes with the installation of solubilizing side chains. The initial stage involves the reflux of pyrrole and 6-chloro-1-hexanal in dichloromethane with activated Montmorillonite K10, followed by oxidation to yield the key 5,10,15,20-tetrakis(5-chloropentyl)-porphine intermediate. This intermediate serves as a versatile platform for further derivatization, allowing manufacturers to produce a library of analogues by simply varying the amine reactant in the second stage. The detailed standardized synthesis steps, including specific molar ratios, temperature controls, and purification parameters required to achieve pharmaceutical-grade purity, are outlined in the comprehensive guide below.
- Condense pyrrole and 6-chloro-1-hexanal using Montmorillonite K10 as a Lewis acid catalyst in dichloromethane, followed by oxidation with chloranil to form the chloro-intermediate.
- Perform a nucleophilic substitution reaction on the intermediate using specific amines (e.g., morpholine, pyrrolidine) in the presence of potassium carbonate and potassium iodide.
- Purify the final crude product via silica gel column chromatography using a solvent system of dichloromethane and methanol to isolate the high-purity alkylporphyrin.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers distinct strategic advantages regarding cost stability and raw material security. The reliance on commodity chemicals such as pyrrole and simple halo-aldehydes ensures a robust supply base that is less susceptible to the volatility often associated with exotic reagents. Moreover, the elimination of expensive transition metal catalysts and the use of a recyclable solid acid catalyst significantly reduce the raw material costs per kilogram of finished product. The simplified purification workflow, which avoids complex metal scavenging steps, further contributes to operational efficiency by reducing processing time and consumable usage, ultimately translating to a more competitive pricing structure for bulk purchasers.
- Cost Reduction in Manufacturing: The substitution of traditional liquid Lewis acids with Montmorillonite K10 eliminates the need for specialized corrosion-resistant equipment and complex neutralization protocols, leading to substantial capital expenditure savings. Additionally, the high selectivity of the reaction minimizes the formation of byproducts, which reduces the load on downstream purification units and lowers the consumption of chromatography media and solvents. This streamlined process flow ensures that the cost of goods sold (COGS) remains optimized even when scaling up to multi-ton production volumes.
- Enhanced Supply Chain Reliability: By utilizing widely available starting materials and avoiding reagents with long lead times or restrictive export controls, manufacturers can maintain consistent production schedules and meet tight delivery deadlines. The robustness of the reaction conditions, which tolerate minor variations in temperature and stoichiometry without significant yield loss, further enhances process reliability and reduces the risk of batch failures. This stability is critical for maintaining uninterrupted supply to pharmaceutical partners who depend on just-in-time inventory models for their clinical and commercial programs.
- Scalability and Environmental Compliance: The use of solid catalysts and standard organic solvents like dichloromethane and acetonitrile facilitates straightforward scale-up from pilot plant to commercial manufacturing without requiring fundamental process redesigns. The reduced generation of hazardous acidic waste aligns with increasingly strict environmental regulations, minimizing disposal costs and mitigating regulatory risks associated with chemical manufacturing. This eco-friendly profile not only supports corporate sustainability goals but also simplifies the permitting process for new production facilities in regulated markets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these alkylporphyrin derivatives, based on the specific data and embodiments provided in the patent literature. Understanding these details is essential for stakeholders evaluating the feasibility of integrating this technology into their existing portfolios or supply networks. The answers reflect the proven capabilities of the described synthetic method to deliver high-performance materials suitable for rigorous biomedical and agrochemical applications.
Q: What are the advantages of using Montmorillonite K10 over traditional liquid Lewis acids?
A: Montmorillonite K10 is a solid acid catalyst that simplifies the workup process significantly compared to liquid acids like BF3·Et2O. It eliminates the need for complex neutralization steps and reduces corrosive waste, making the process more environmentally friendly and easier to scale for industrial production.
Q: What specific medical applications do these alkylporphyrin compounds target?
A: These compounds function as second-generation photosensitizers designed for Photodynamic Therapy (PDT). They exhibit high selective absorption in tumor cells, such as colon cancer (SW480) and sarcoma (S180), while minimizing systemic skin photosensitivity, making them ideal candidates for oncology treatments.
Q: How does the side-chain modification affect the solubility profile?
A: By introducing amino-substituted alkyl chains at the 5, 10, 15, and 20 positions of the porphyrin ring, the compounds achieve a balanced lipophilic and hydrophilic character. This dual solubility facilitates transport through the bloodstream and enhances penetration across cell membranes, improving therapeutic efficacy.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alkylporphyrin Supplier
As a premier CDMO partner, NINGBO INNO PHARMCHEM possesses the technical expertise and infrastructure necessary to translate this patented alkylporphyrin synthesis into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with unwavering consistency and quality. We operate stringent purity specifications and utilize rigorous QC labs to verify that every batch of photosensitizer intermediate meets the exacting standards required for pharmaceutical development, providing you with the confidence to advance your projects through clinical trials and into the market.
We invite you to engage with our technical procurement team to discuss how we can tailor this synthesis to your specific volume and purity requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this greener, more efficient manufacturing route. Contact us today to obtain specific COA data and route feasibility assessments that will demonstrate our commitment to being your trusted partner in the development of next-generation photodynamic therapies.
